Abstract
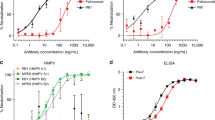
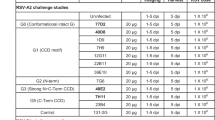
Respiratory syncytial virus (RSV) remains a major human pathogen, infecting the majority of infants before age two and causing re-infection throughout life. Despite decades of RSV research, there is no licensed RSV vaccine. Most candidate vaccines studied to date have incorporated the RSV fusion (F) surface glycoprotein, because the sequence of F is highly conserved among strains of RSV. To better define the human B cell response to RSV F, we isolated from a single donor 13 new neutralizing human monoclonal antibodies (mAbs) that recognize the RSV F protein in the pre-fusion conformation. Epitope binning studies showed that the majority of neutralizing mAbs targeted a new antigenic site on the globular head domain of F, designated here antigenic site VIII, which occupies an intermediate position between the previously defined major antigenic sites II and site Ø. Antibodies to site VIII competed for binding with antibodies to both of those adjacent neutralizing sites. The new mAbs exhibited unusual breadth for pre-fusion F-specific antibodies, cross-reacting with F proteins from both RSV subgroups A and B viruses. We solved the X-ray crystal structure of one site VIII mAb, hRSV90, in complex with pre-fusion RSV F protein. The structure revealed a large footprint of interaction for hRSV90 on RSV F, in which the heavy chain and light chain both have specific interactions mediating binding to site VIII, the heavy chain overlaps with site Ø, and the light chain interacts partially with site II.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
The IMpact-RSV Study Group. Palivizumab, a humanized respiratory syncytial virus monoclonal antibody, reduces hospitalization from respiratory syncytial virus infection in high-risk infants. Pediatrics 102, 531–537 (1998).
Smith, E. C., Popa, A., Chang, A., Masante, C. & Dutch, R. E. Viral entry mechanisms: the increasing diversity of paramyxovirus entry. FEBS J. 276, 7217–7227 (2009).
McLellan, J. S., Yang, Y., Graham, B. S. & Kwong, P. D. Structure of respiratory syncytial virus fusion glycoprotein in the postfusion conformation reveals preservation of neutralizing epitopes. J. Virol. 85, 7788–7796 (2011).
McLellan, J. S. et al. Structure of RSV fusion glycoprotein trimer bound to a prefusion-specific neutralizing antibody. Science 340, 1113–1117 (2013).
McLellan, J. S. et al. Structure-based design of a fusion glycoprotein vaccine for respiratory syncytial virus. Science 342, 592–598 (2013).
Krarup, A. et al. A highly stable prefusion RSV F vaccine derived from structural analysis of the fusion mechanism. Nat. Commun. 6, 8143 (2015).
Anderson, L. J. et al. Antigenic characterization of respiratory syncytial virus strains with monoclonal antibodies. J. Infect. Dis. 151, 626–633 (1985).
Wu, H. et al. Development of motavizumab, an ultra-potent antibody for the prevention of respiratory syncytial virus infection in the upper and lower respiratory tract. J. Mol. Biol. 368, 652–665 (2007).
Wu, S. J. et al. Characterization of the epitope for anti-human respiratory syncytial virus F protein monoclonal antibody 101F using synthetic peptides and genetic approaches. J. Gen. Virol. 88, 2719–2723 (2007).
Lopez, J. A. et al. Antigenic structure of human respiratory syncytial virus fusion glycoprotein. J. Virol. 72, 6922–6928 (1998).
Gilman, M. S. A. et al. Characterization of a prefusion-specific antibody that recognizes a quaternary, cleavage-dependent epitope on the RSV fusion glycoprotein. PLoS Pathogens 11, e1005035 (2015).
Corti, D. et al. Cross-neutralization of four paramyxoviruses by a human monoclonal antibody. Nature 501, 439–443 (2013).
McLellan, J. S. Neutralizing epitopes on the respiratory syncytial virus fusion glycoprotein. Curr. Opin. Virol. 11, 70–75 (2015).
Mousa, J. J. et al. Structural basis for nonneutralizing antibody competition at antigenic site II of the respiratory syncytial virus fusion protein. Proc. Natl Acad. Sci. USA 113, E6849–E6858 (2016).
Ngwuta, J. O. et al. Prefusion F-specific antibodies determine the magnitude of RSV neutralizing activity in human sera. Sci. Transl. Med. 7, 309ra162 (2015).
Smith, S. A. & Crowe, J. E. Use of human hybridoma technology to isolate human monoclonal antibodies. Microbiol. Spectr. 3, 1–12 (2015).
Strong, M. et al. Toward the structural genomics of complexes: crystal structure of a PE/PPE protein complex from Mycobacterium tuberculosis. Proc. Natl Acad. Sci. USA 103, 8060–8065 (2006).
Brochet, X., Lefranc, M. P. & Giudicelli, V. IMGT/V-QUEST: the highly customized and integrated system for IG and TR standardized V-J and V-D-J sequence analysis. Nucleic Acids Res. 36, 503–508 (2008).
Yu, X., McGraw, P. A., House, F. S. & Crowe, J. E. An optimized electrofusion-based protocol for generating virus-specific human monoclonal antibodies. J. Immunol. Methods 336, 142–151 (2008).
Kabsch, W. XDS. Acta Crystallogr. 66, 125–132 (2010).
Adams, P. D. et al. PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. 66, 213–221 (2010).
Emsley, P. & Cowtan, K. COOT: model-building tools for molecular graphics. Acta Crystallogr. 60, 2126–2132 (2004).
Acknowledgements
The authors thank members of the Crowe Laboratory, especially R. Bombardi, N. Murphy and G. Sapparapu, for technical assistance. A portion of the experiments described here used the Vanderbilt robotic crystallization facility, which was supported by NIH grant S10 RR026915. J.J.M. was supported by T32 AI 07474, NIAID and National Institutes of Health (NIH). The described project was supported by CTSA award no. UL1TR000445 from the National Center for Advancing Translational Sciences. Its contents are solely the responsibility of the authors and do not necessarily represent official views of the National Center for Advancing Translational Sciences or the NIH.
Author information
Authors and Affiliations
Contributions
J.J.M. and J.E.C. designed the experiments and interpreted the data. J.J.M. expressed, purified, crystallized, collected data and solved the crystal structure of the hRSV-90-RSV F SC-TM complex. J.J.M. and N.K. generated hybridomas by electrofusion. P.M. expanded hybridoma cell cultures. J.J.M. and P.M. performed neutralization experiments and J.J.M. performed competition-binding and ELISA binding studies. P.G. performed self-reactivity assays. J.J.M. and J.E.C. wrote the manuscript. All authors reviewed and edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures 1–9, Supplementary Table 1. (PDF 6535 kb)
Rights and permissions
About this article
Cite this article
Mousa, J., Kose, N., Matta, P. et al. A novel pre-fusion conformation-specific neutralizing epitope on the respiratory syncytial virus fusion protein. Nat Microbiol 2, 16271 (2017). https://doi.org/10.1038/nmicrobiol.2016.271
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/nmicrobiol.2016.271
This article is cited by
-
Design of hepadnavirus core protein-based chimeric virus-like particles carrying epitopes from respiratory syncytial virus
npj Vaccines (2024)
-
Antibody-based protection against respiratory syncytial virus in mice and their offspring through vectored immunoprophylaxis
Gene Therapy (2023)
-
Profiling of hMPV F-specific antibodies isolated from human memory B cells
Nature Communications (2022)
-
In silico virtual screening of lead compounds for major antigenic sites in respiratory syncytial virus fusion protein
Emergent Materials (2022)
-
Respiratory syncytial, parainfluenza and influenza virus infection in young children with acute lower respiratory infection in rural Gambia
Scientific Reports (2019)