Abstract
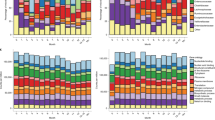
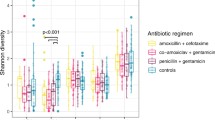
Development of the preterm infant gut microbiota is emerging as a critical research priority1. Since preterm infants almost universally receive early and often extended antibiotic therapy2, it is important to understand how these interventions alter gut microbiota development3–6. Analysis of 401 stools from 84 longitudinally sampled preterm infants demonstrates that meropenem, cefotaxime and ticarcillin–clavulanate are associated with significantly reduced species richness. In contrast, vancomycin and gentamicin, the antibiotics most commonly administered to preterm infants, have non-uniform effects on species richness, but these can be predicted with 85% accuracy based on the relative abundance of only two bacterial species and two antibiotic resistance (AR) genes at treatment initiation. To investigate resistome development, we functionally selected resistance to 16 antibiotics from 21 faecal metagenomic expression libraries. Of the 794 AR genes identified, 79% had not previously been classified as AR genes. Combined with deep shotgun sequencing of all stools, we find that multidrug-resistant members of the genera Escherichia, Klebsiella and Enterobacter, genera commonly associated with nosocomial infections, dominate the preterm infant gut microbiota. AR genes that are enriched following specific antibiotic treatments are generally unique to the specific treatment and are highly correlated with the abundance of a single species. The most notable exceptions include ticarcillin–clavulanate and ampicillin, both of which enrich for a large number of overlapping AR genes, and are correlated with Klebsiella pneumoniae. We find that all antibiotic treatments are associated with widespread collateral microbiome impact by enrichment of AR genes that have no known activity against the specific antibiotic driver.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Groer, M. W. et al. Development of the preterm infant gut microbiome: a research priority. Microbiome 2, 38 (2014).
Clark, R. H., Bloom, B. T., Spitzer, A. R. & Gerstmann, D. R. Reported medication use in the neonatal intensive care unit: data from a large national data set. Pediatrics 117, 1979–1987 (2006).
La Rosa, P. S. et al. Patterned progression of bacterial populations in the premature infant gut. Proc. Natl Acad. Sci. USA 111, 12522–12527 (2014).
Vangay, P., Ward, T., Gerber, J. S. & Knights, D. Antibiotics, pediatric dysbiosis, and disease. Cell Host Microbe 17, 553–564 (2015).
Greenwood, C. et al. Early empiric antibiotic use in preterm infants is associated with lower bacterial diversity and higher relative abundance of Enterobacter. J. Pediatr. 165, 23–29 (2014).
Arboleya, S. et al. Intestinal microbiota development in preterm neonates and effect of perinatal antibiotics. J. Pediatr. 166, 538–544 (2015).
Yatsunenko, T. et al. Human gut microbiome viewed across age and geography. Nature 486, 222–227 (2012).
Azad, M. B. et al. Gut microbiota of healthy Canadian infants: profiles by mode of delivery and infant diet at 4 months. Can. Med. Assoc. J. 185, 385–394 (2013).
Gronlund, M. M., Lehtonen, O. P., Eerola, E. & Kero, P. Fecal microflora in healthy infants born by different methods of delivery: permanent changes in intestinal flora after cesarean delivery. J. Pediatr. Gastroenterol. Nutr. 28, 19–25 (1999).
Backhed, F. et al. Dynamics and stabilization of the human gut microbiome during the first year of life. Cell Host Microbe 17, 690–703 (2015).
Rosenthal, V. D. et al. International Nosocomial Infection Control Consortium (INICC) report, data summary of 36 countries, for 2004–2009. Am. J. Infect. Control 40, 396–407 (2012).
Richards, M. J., Edwards, J. R., Culver, D. H. & Gaynes, R. P. Nosocomial infections in pediatric intensive care units in the United States. National Nosocomial Infections Surveillance System. Pediatrics 103, e39 (1999).
Dicksved, J. et al. Molecular analysis of the gut microbiota of identical twins with Crohn's disease. ISME J. 2, 716–727 (2008).
Abrahamsson, T. R. et al. Low gut microbiota diversity in early infancy precedes asthma at school age. Clin. Exp. Allergy 44, 842–850 (2014).
Fujimura, K. E., Slusher, N. A., Cabana, M. D. & Lynch, S. V. Role of the gut microbiota in defining human health. Expert Rev. Anti-infect. Ther. 8, 435–454 (2010).
Wang, Y. et al. 16S rRNA gene-based analysis of fecal microbiota from preterm infants with and without necrotizing enterocolitis. ISME J 3, 944–954 (2009).
Forsberg, K. J. et al. The shared antibiotic resistome of soil bacteria and human pathogens. Science 337, 1107–1111 (2012).
Pehrsson, E. C., Forsberg, K. J., Gibson, M. K., Ahmadi, S. & Dantas, G. Novel resistance functions uncovered using functional metagenomic investigations of resistance reservoirs. Front. Microbiol. 4, 145 (2013).
Forsberg, K. J. et al. Bacterial phylogeny structures soil resistomes across habitats. Nature 509, 612–616 (2014).
Moore, A. M. et al. Gut resistome development in healthy twin pairs in the first year of life. Microbiome 3, 27 (2015).
Moore, A. M. et al. Pediatric fecal microbiota harbor diverse and novel antibiotic resistance genes. PLoS ONE 8, e78822 (2013).
Sommer, M. O., Dantas, G. & Church, G. M. Functional characterization of the antibiotic resistance reservoir in the human microflora. Science 325, 1128–1131 (2009).
Riesenfeld, C. S., Goodman, R. M. & Handelsman, J. Uncultured soil bacteria are a reservoir of new antibiotic resistance genes. Environ. Microbiol. 6, 981–989 (2004).
Handelsman, J. Metagenomics: application of genomics to uncultured microorganisms. Microbiol. Mol. Biol. Rev. 68, 669–685 (2004).
Chai, G. et al. Trends of outpatient prescription drug utilization in US children, 2002–2010. Pediatrics 130, 23–31 (2012).
Gibson, M. K., Forsberg, K. J. & Dantas, G. Improved annotation of antibiotic resistance determinants reveals microbial resistomes cluster by ecology. ISME J. 9, 207–216. (2015).
McArthur, A. G. et al. The comprehensive antibiotic resistance database. Antimicrob. Agents Chemother. 57, 3348–3357 (2013).
Boucher, H. W. et al. Bad bugs, no drugs: no ESKAPE! An update from the Infectious Diseases Society of America. Clin. Infect. Dis. 48, 1–12 (2009).
Peterson, L. R. Bad bugs, no drugs: no ESCAPE revisited. Clin. Infect. Dis. 49, 992–993 (2009).
Brooks, B. et al. Microbes in the neonatal intensive care unit resemble those found in the gut of premature infants. Microbiome 2, 1 (2014).
Kaminski, J. et al. High-specificity targeted functional profiling in microbial communities with ShortBRED. PLoS Comput. Biol. 11, e1004557 (2015).
Wiener, A. L. a. M. Classification and Regression by randomForest. R News 2, 18–22 (2002).
Mahoney, T. F. & Silhavy, T. J. The Cpx stress response confers resistance to some, but not all, bactericidal antibiotics. J. Bacteriol. 195, 1869–1874 (2013).
Caporaso, J. G. et al. Ultra-high-throughput microbial community analysis on the Illumina HiSeq and MiSeq platforms. ISME J. 6, 1621–1624 (2012).
Edgar, R. C. UPARSE: highly accurate OTU sequences from microbial amplicon reads. Nature Methods 10, 996–998 (2013).
Caporaso, J. G. et al. QIIME allows analysis of high-throughput community sequencing data. Nature Methods 7, 335–336 (2010).
Bolger, A. M., Lohse, M. & Usadel, B. Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics 30, 2114–2120 (2014).
Schmieder, R. & Edwards, R. Fast identification and removal of sequence contamination from genomic and metagenomic datasets. PLoS ONE 6, e17288 (2011).
Segata, N. et al. Metagenomic microbial community profiling using unique clade-specific marker genes. Nature Methods 9, 811–814 (2012).
Markowitz, V. M. et al. IMG: the Integrated Microbial Genomes database and comparative analysis system. Nucleic Acids Res. 40, D115–D122 (2012).
Zhu, W., Lomsadze, A. & Borodovsky, M. Ab initio gene identification in metagenomic sequences. Nucleic Acids Res. 38, e132 (2010).
Finn, R. D., Clements, J. & Eddy, S. R. HMMER web server: interactive sequence similarity searching. Nucleic Acids Res. 39, W29–W37 (2011).
Li, W. & Godzik, A. Cd-hit: a fast program for clustering and comparing large sets of protein or nucleotide sequences. Bioinformatics 22, 1658–1659 (2006).
Tita, A. T. & Andrews, W. W. Diagnosis and management of clinical chorioamnionitis. Clin. Perinatol. 37, 339–354 (2010).
Avila, C. et al. Usefulness of two clinical chorioamnionitis definitions in predicting neonatal infectious outcomes: a systematic review. Am. J. Perinatol. 32, 1001–1009 (2015).
Acknowledgements
We thank A. Moore for initial study conception and clinical insight, C. Hall-Moore for assistance in access to cohort samples, M. Wallace for cultured strain characterization, and members of the Dantas lab for discussions of the results and analyses. We also thank families and clinical staff in the Saint Louis Children's Hospital Neonatal Intensive Care Unit for their cooperation with study, and Laura Linneman and Julie Hoffmann for their efforts in enrolment and data accrual. This work is supported in part by awards to G.D. through the Children's Discovery Institute (MD-II-2011-117 and 127), the March of Dimes Foundation (6-FY12-394), and the National Institute of General Medical Sciences of the National Institutes of Health (R01-GM099538). This work is also supported in part by an award to Washington University School of Medicine through a Clinical and Translational Science Award (CTSA) Grant (UL1 TR000448). This collection was also supported by the US National Institutes of Health Grants UH3AI083265 and P30DK052574 (Biobank Core), along with funding from the Eunice Kennedy Shriver National Institute of Child Health and Human Development, and Foundation for the National Institutes of Health (made possible by support from the Gerber Foundation). M.K.G. is a Mr. and Mrs. Spencer T. Olin Fellow at Washington University and a National Science Foundation (NSF) graduate research fellow (DGE-1143954). The content is solely the responsibility of the authors and does not necessarily represent the official views of the funding agencies.
Author information
Authors and Affiliations
Contributions
P.I.T. and B.B.W. assembled the cohort and developed protocols and infrastructure to obtain the biologics and clinical metadata from the cohort. M.K.G. and G.D. designed and conceived the study. M.K.G. performed 16S rRNA gene sequencing for comparison to term infants. B.W. prepared shotgun metagenomic sequencing libraries. S.A. created metagenomic libraries, performed functional selections, and prepared sequencing libraries. M.K.G. performed functional metagenomic data analysis, shotgun metagenomic data analysis, statistical modelling. M.K.G. wrote the manuscript with contributions from C.A.B., P.I.T., B.B.W., and G.D.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures 1-9 (PDF 646 kb)
Supplementary Table 1
Metadata for 401 stool samples and 84 individuals. (XLSX 192 kb)
Supplementary Table 2
Functional metagenomic selections of 21 fecal metagenomic expression libraries to 16 antibiotics. (XLSX 39 kb)
Supplementary Table 3
VelvetOptimiser assembly stats for 312 assembled preterm infant meatgenomes. (XLSX 61 kb)
Rights and permissions
About this article
Cite this article
Gibson, M., Wang, B., Ahmadi, S. et al. Developmental dynamics of the preterm infant gut microbiota and antibiotic resistome. Nat Microbiol 1, 16024 (2016). https://doi.org/10.1038/nmicrobiol.2016.24
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/nmicrobiol.2016.24
This article is cited by
-
The role of gut microbiota in human metabolism and inflammatory diseases: a focus on elderly individuals
Annals of Microbiology (2024)
-
Longitudinal dynamics of farmer and livestock nasal and faecal microbiomes and resistomes
Nature Microbiology (2024)
-
Strain-resolved metagenomic analysis of the gut as a reservoir for bloodstream infection pathogens among premature infants in Singapore
Gut Pathogens (2023)
-
Intravenous antibiotics in preterm infants have a negative effect upon microbiome development throughout preterm life
Gut Pathogens (2023)
-
Improved eukaryotic detection compatible with large-scale automated analysis of metagenomes
Microbiome (2023)