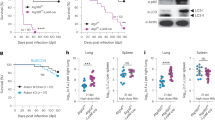
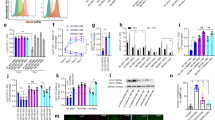
Abstract
Mycobacterium tuberculosis (Mtb) establishes a persistent infection, despite inducing antigen-specific T-cell responses. Although T cells arrive at the site of infection, they do not provide sterilizing immunity. The molecular basis of how Mtb impairs T-cell function is not clear. Mtb has been reported to block major histocompatibility complex class II (MHC-II) antigen presentation; however, no bacterial effector or host-cell target mediating this effect has been identified. We recently found that Mtb EsxH, which is secreted by the Esx-3 type VII secretion system, directly inhibits the endosomal sorting complex required for transport (ESCRT) machinery. Here, we showed that ESCRT is required for optimal antigen processing; correspondingly, overexpression and loss-of-function studies demonstrated that EsxH inhibited the ability of macrophages and dendritic cells to activate Mtb antigen-specific CD4+ T cells. Compared with the wild-type strain, the esxH-deficient strain induced fivefold more antigen-specific CD4+ T-cell proliferation in the mediastinal lymph nodes of mice. We also found that EsxH undermined the ability of effector CD4+ T cells to recognize infected macrophages and clear Mtb. These results provide a molecular explanation for how Mtb impairs the adaptive immune response.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Change history
14 July 2017
In the PDF version of this article previously published, the year of publication provided in the footer of each page and in the 'How to cite' section was erroneously given as 2017, it should have been 2016. This error has now been corrected. The HTML version of the article was not affected.
References
Wolf, A. J. et al. Initiation of the adaptive immune response to Mycobacterium tuberculosis depends on antigen production in the local lymph node, not the lungs. J. Exp. Med. 205, 105–115 (2008).
MacMicking, J. D. Interferon-inducible effector mechanisms in cell-autonomous immunity. Nat. Rev. Immunol. 12, 367–382 (2012).
Fortune, S. M. et al. Mycobacterium tuberculosis inhibits macrophage responses to IFN-γ through myeloid differentiation factor 88-dependent and -independent mechanisms. J. Immunol. 172, 6272–6280 (2004).
Noss, E. H. et al. Toll-like receptor 2-dependent inhibition of macrophage class II MHC expression and antigen processing by 19-kDa lipoprotein of Mycobacterium tuberculosis. J. Immunol. 167, 910–918 (2001).
Pennini, M. E., Pai, R. K., Schultz, D. C., Boom, W. H. & Harding, C. V. Mycobacterium tuberculosis 19-kDa lipoprotein inhibits IFN-γ-induced chromatin remodeling of MHC2TA by TLR2 and MAPK signaling. J. Immunol. 176, 4323–4330 (2006).
Kincaid, E. Z. et al. Codominance of TLR2-dependent and TLR2-independent modulation of MHC class II in Mycobacterium tuberculosis infection in vivo. J. Immunol. 179, 3187–3195 (2007).
Blander, J. M. & Medzhitov, R. On regulation of phagosome maturation and antigen presentation. Nat. Immunol. 7, 1029–1035 (2006).
Ramachandra, L., Noss, E., Boom, W. H. & Harding, C. V. Processing of Mycobacterium tuberculosis antigen 85B involves intraphagosomal formation of peptide-major histocompatibility complex II complexes and is inhibited by live bacilli that decrease phagosome maturation. J. Exp. Med. 194, 1421–1432 (2001).
Jagannath, C. et al. Autophagy enhances the efficacy of BCG vaccine by increasing peptide presentation in mouse dendritic cells. Nat. Med. 15, 267–276 (2009).
Johansen, P. et al. Relief from Zmp1-mediated arrest of phagosome maturation is associated with facilitated presentation and enhanced immunogenicity of mycobacterial antigens. Clin. Vaccine. Immunol. 18, 907–913 (2011).
Majlessi, L. et al. Inhibition of phagosome maturation by mycobacteria does not interfere with presentation of mycobacterial antigens by MHC molecules. J. Immunol. 179, 1825–1833 (2007).
Hanekom, W. A. et al. Mycobacterium tuberculosis inhibits maturation of human monocyte-derived dendritic cells in vitro. J. Infect. Dis. 188, 257–266 (2003).
Henderson, R. A., Watkins, S. C. & Flynn, J. L. Activation of human dendritic cells following infection with Mycobacterium tuberculosis. J. Immunol. 159, 635–643 (1997).
Giacomini, E. et al. Infection of human macrophages and dendritic cells with Mycobacterium tuberculosis induces a differential cytokine gene expression that modulates T cell response. J. Immunol. 166, 7033–7041 (2001).
Wolf, A. J. et al. Mycobacterium tuberculosis infects dendritic cells with high frequency and impairs their function in vivo. J. Immunol. 179, 2509–2519 (2007).
Grace, P. S. & Ernst, J. D. Suboptimal antigen presentation contributes to virulence of Mycobacterium tuberculosis in vivo. J. Immunol. 196, 357–364 (2016).
Samstein, M. et al. Essential yet limited role for CCR2+ inflammatory monocytes during Mycobacterium tuberculosis-specific T cell priming. eLife 2, e01086 (2013).
Srivastava, S. & Ernst, J. D. Cell-to-cell transfer of M. tuberculosis antigens optimizes CD4T cell priming. Cell Host Microbe 15, 741–752 (2014).
Srivastava, S., Grace, P. S. & Ernst, J. D. Antigen export reduces antigen presentation and limits T cell control of M. tuberculosis. Cell Host Microbe 19, 44–54 (2016).
Mehra, A. et al. Mycobacterium tuberculosis type VII secreted effector EsxH targets host ESCRT to impair trafficking. PLoS Pathog. 9, e1003734 (2013).
Serafini, A., Pisu, D., Palù, G., Rodriguez, G. M. & Manganelli, R. The ESX-3 secretion system is necessary for iron and zinc homeostasis in Mycobacterium tuberculosis. PLoS ONE 8, e78351 (2013).
Serafini, A., Boldrin, F., Palù, G. & Manganelli, R. Characterization of a Mycobacterium tuberculosis ESX-3 conditional mutant: essentiality and rescue by iron and zinc. J. Bacteriol. 191, 6340–6344 (2009).
Siegrist, M. S. et al. Mycobacterial Esx-3 requires multiple components for iron acquisition. mBio 5, e01073–14 (2014).
Siegrist, M. S. et al. Mycobacterial Esx-3 is required for mycobactin-mediated iron acquisition. Proc. Natl Acad. Sci. USA 106, 18792–18797 (2009).
Tinaztepe, E. et al. Role of metal-dependent regulation of ESX-3 secretion in intracellular survival of Mycobacterium tuberculosis. Infect. Immun. 84, 2255–2263 (2016).
Tufariello, J. M. et al. Separable roles for Mycobacterium tuberculosis ESX-3 effectors in iron acquisition and virulence. Proc. Natl Acad. Sci. USA 113, E348–E357 (2016).
Sweeney, K. A. et al. A recombinant Mycobacterium smegmatis induces potent bactericidal immunity against Mycobacterium tuberculosis. Nat. Med. 17, 1261–1268 (2011).
Raiborg, C. & Stenmark, H. The ESCRT machinery in endosomal sorting of ubiquitylated membrane proteins. Nature 458, 445–452 (2009).
Philips, J. A., Porto, M. C., Wang, H., Rubin, E. J. & Perrimon, N. ESCRT factors restrict mycobacterial growth. Proc. Natl Acad. Sci. USA 105, 3070–3075 (2008).
Tamura, T. et al. The role of antigenic peptide in CD4+ T helper phenotype development in a T cell receptor transgenic model. Int. Immunol. 16, 1691–1699 (2004).
Baravalle, G. et al. Ubiquitination of CD86 is a key mechanism in regulating antigen presentation by dendritic cells. J. Immunol. 187, 2966–2973 (2011).
Ohmura-Hoshino, M. et al. Cutting edge: requirement of MARCH-I-mediated MHC II ubiquitination for the maintenance of conventional dendritic cells. J. Immunol. 183, 6893–6897 (2009).
Via, L. E. et al. Arrest of mycobacterial phagosome maturation is caused by a block in vesicle fusion between stages controlled by rab5 and rab7. J. Biol. Chem. 272, 13326–13331 (1997).
Gutierrez, M. G. et al. Autophagy is a defense mechanism inhibiting BCG and Mycobacterium tuberculosis survival in infected macrophages. Cell 119, 753–766 (2004).
Sakowski, E. T. et al. Ubiquilin 1 promotes IFN-γ-induced xenophagy of Mycobacterium tuberculosis. PLoS Pathogens 11, e1005076 (2015).
Srivastava, S. & Ernst, J. D. Cutting edge: direct recognition of infected cells by CD4T cells is required for control of intracellular Mycobacterium tuberculosis in vivo. J. Immunol. 191, 1016–1020 (2013).
Ten Broeke, T., Wubbolts, R. & Stoorvogel, W. MHC class II antigen presentation by dendritic cells regulated through endosomal sorting. Cold Spring Harb. Perspect. Biol. 5, a016873 (2013).
Zwart, W. et al. Spatial separation of HLA-DM/HLA-DR interactions within MIIC and phagosome-induced immune escape. Immunity 22, 221–233 (2005).
Bosch, B. et al. Disruption of multivesicular body vesicles does not affect major histocompatibility complex (MHC) class II-peptide complex formation and antigen presentation by dendritic cells. J. Biol. Chem. 288, 24286–24292 (2013).
Singh, C. R. et al. Processing and presentation of a mycobacterial antigen 85B epitope by murine macrophages is dependent on the phagosomal acquisition of vacuolar proton ATPase and in situ activation of cathepsin D. J. Immunol. 177, 3250–3259 (2006).
Hinchey, J. et al. Enhanced priming of adaptive immunity by a proapoptotic mutant of Mycobacterium tuberculosis. J. Clin. Invest. 117, 2279–2288 (2007).
Saikolappan, S. et al. The fbpA/sapM double knock out strain of Mycobacterium tuberculosis is highly attenuated and immunogenic in macrophages. PLoS ONE 7, e36198 (2012).
Hervas-Stubbs, S. et al. High frequency of CD4+ T cells specific for the TB10.4 protein correlates with protection against Mycobacterium tuberculosis infection. Infect. Immun. 74, 3396–3407 (2006).
Skjøt, R. L. et al. Epitope mapping of the immunodominant antigen TB10.4 and the two homologous proteins TB10.3 and TB12.9, which constitute a subfamily of the esat-6 gene family. Infect. Immun. 70, 5446–5453 (2002).
Sreejit, G. et al. The ESAT-6 protein of Mycobacterium tuberculosis interacts with beta-2-microglobulin (β2M) affecting antigen presentation function of macrophage. PLoS Pathog. 10, e1004446 (2014).
Rybniker, J. et al. Anticytolytic screen identifies inhibitors of mycobacterial virulence protein secretion. Cell Host Microbe 16, 538–548 (2014).
Gallegos, A. M., Pamer, E. G. & Glickman, M. S. Delayed protection by ESAT-6-specific effector CD4+ T cells after airborne M. tuberculosis infection. J. Exp. Med. 205, 2359–2368 (2008).
Wong, K. W. & Jacobs, W. R. Jr Critical role for NLRP3 in necrotic death triggered by Mycobacterium tuberculosis. Cell. Microbiol. 13, 1371–1384 (2011).
Banaiee, N., Kincaid, E. Z., Buchwald, U., Jacobs, W. R. Jr & Ernst, J. D. Potent inhibition of macrophage responses to IFN-γ by live virulent Mycobacterium tuberculosis is independent of mature mycobacterial lipoproteins but dependent on TLR2. J. Immunol. 176, 3019–3027 (2006).
Luber, C. A. et al. Quantitative proteomics reveals subset-specific viral recognition in dendritic cells. Immunity 32, 279–289 (2010).
Acknowledgements
We thank the New York University Cytometry Core at the Laura and Isaac Perlmutter Cancer Center (partially supported by a Cancer Center Support Grant, P30CA016087) for their assistance with flow cytometry, Beatrix Ueberheide and Jessica Chapman-Lim for assistance with proteomics (New York University Proteomics Resource Center), Michael Glickman (Memorial Sloan-Kettering Cancer Center) for the C7TCR Tg mice, BEI Resources, NIAID, NIH for Ag85B protein (Ag85B Recombinant Protein Reference Standard, NR-14870), and members of the Philips and Ernst laboratories, in particular Colette O'Shaughnessy and Ludovic Desvignes, for assistance and helpful discussions. This work was supported by National Institutes of Health grants R01s AI087682, AI026170, and AI098925, K08 AI119150-01 and UL1 TR000038.
Author information
Authors and Affiliations
Contributions
C.P.-C., and J.A.P. conceived and designed the study. C.P.-C. performed all the in vitro experiments with help from T.K., with the exception of the trafficking studies done by A.Z. C.P.-C. and S.S. designed and performed in vivo experiments. A.M. and H.S.P. created plasmids. J.M.T. and W.R.J. contributed the ΔesxH and Δpe5-ppe4 strains. C.P.-C. and P.S.G performed flow cytometry. C.P.-C. and J.A.P. interpreted the results and wrote the manuscript. J.D.E., W.R.J. and J.A.P. oversaw the project.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary Figures 1–4 (PDF 6033 kb)
Rights and permissions
About this article
Cite this article
Portal-Celhay, C., Tufariello, J., Srivastava, S. et al. Mycobacterium tuberculosis EsxH inhibits ESCRT-dependent CD4+ T-cell activation. Nat Microbiol 2, 16232 (2017). https://doi.org/10.1038/nmicrobiol.2016.232
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/nmicrobiol.2016.232
This article is cited by
-
From immunology to artificial intelligence: revolutionizing latent tuberculosis infection diagnosis with machine learning
Military Medical Research (2023)
-
A type VII-secreted lipase toxin with reverse domain arrangement
Nature Communications (2023)
-
Mycobacterium tuberculosis and its clever approaches to escape the deadly macrophage
World Journal of Microbiology and Biotechnology (2023)
-
Immune evasion and provocation by Mycobacterium tuberculosis
Nature Reviews Microbiology (2022)
-
Pore-forming Esx proteins mediate toxin secretion by Mycobacterium tuberculosis
Nature Communications (2021)