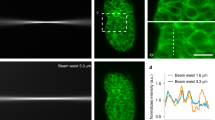
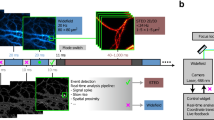
Abstract
The impact of light-sheet fluorescence microscopy (LSFM) is visible in fields as diverse as developmental and cell biology, anatomical science, biophysics and neuroscience. Although adoption among biologists has been steady, LSFM has not displaced more traditional imaging methods despite its often-superior performance. One reason for this is that the field has largely conformed to a do-it-yourself ethic, although the challenges of big image data cannot be overstated. With the most powerful implementations of LSFM available to only a few groups worldwide, the scope of this technique is unnecessarily limited. Here we elucidate the key developments and define a simple set of underlying principles governing LSFM. In doing so, we aim to clarify the decisions to be made for those who wish to develop and use bespoke light-sheet systems and to assist in identifying the best approaches to apply this powerful technique to myriad biological questions.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout










Similar content being viewed by others
References
Chhetri, R.K. et al. Whole-animal functional and developmental imaging with isotropic spatial resolution. Nat. Methods 12, 1171–1178 (2015).
Arrenberg, A.B., Stainier, D.Y.R., Baier, H. & Huisken, J. Optogenetic control of cardiac function. Science 330, 971–974 (2010).
Mickoleit, M. et al. High-resolution reconstruction of the beating zebrafish heart. Nat. Methods 11, 919–922 (2014).
Rauzi, M. et al. Embryo-scale tissue mechanics during Drosophila gastrulation movements. Nat. Commun. 6, 8677 (2015).
Schmid, B. et al. High-speed panoramic light-sheet microscopy reveals global endodermal cell dynamics. Nat. Commun. 4, 2207 (2013).
Welf, E.S. et al. Quantitative multiscale cell imaging in controlled 3D microenvironments. Dev. Cell 36, 462–475 (2016).
Strnad, P. et al. Inverted light-sheet microscope for imaging mouse pre-implantation development. Nat. Methods 13, 139–142 (2016).
Wu, Y. et al. Inverted selective-plane illumination microscopy (iSPIM) enables coupled cell identity lineaging and neurodevelopmental imaging in Caenorhabditis elegans. Proc. Natl. Acad. Sci. USA 108, 17708–17713 (2011).
Wu, Y. et al. Spatially isotropic four-dimensional imaging with dual-view-plane illumination microscopy. Nat. Biotechnol. 31, 1032–1038 (2013).
Kaufmann, A., Mickoleit, M., Weber, M. & Huisken, J. Multilayer mounting enables long-term imaging of zebrafish development in a light-sheet microscope. Development 139, 3242–3247 (2012).
Vladimirov, N. et al. Light-sheet functional imaging in fictively behaving zebrafish. Nat. Methods 11, 883–884 (2014).
Wolf, S. et al. Whole-brain functional imaging with two-photon light-sheet microscopy. Nat. Methods 12, 379–380 (2015).
Pitrone, P.G. et al. OpenSPIM: an open-access light-sheet microscopy platform. Nat. Methods 10, 598–599 (2013).
Gualda, E.J. et al. OpenSpinMicroscopy: an open-source integrated microscopy platform. Nat. Methods 10, 599–600 (2013).
Huisken, J., Swoger, J., Del Bene, F., Wittbrodt, J. & Stelzer, E.H.K. Optical sectioning deep inside live embryos by selective-plane illumination microscopy. Science 305, 1007–1009 (2004).
Huisken, J. & Stainier, D.Y.R. Even fluorescence excitation by multidirectional selective-plane illumination microscopy (mSPIM). Opt. Lett. 32, 2608–2610 (2007).
Keller, P.J., Schmidt, A.D., Wittbrodt, J. & Stelzer, E.H.K. Reconstruction of zebrafish early embryonic development by scanned light-sheet microscopy. Science 322, 1065–1069 (2008).
Chen, B.-C. et al. Lattice light-sheet microscopy: imaging molecules to embryos at high spatiotemporal resolution. Science 346, 1257998 (2014).
Cella Zanacchi, F. et al. Live-cell 3D super-resolution imaging in thick biological samples. Nat. Methods 8, 1047–1049 (2011).
Huang, B., Jones, S.A., Brandenburg, B. & Zhuang, X. Whole-cell 3D STORM reveals interactions between cellular structures with nanometer-scale resolution. Nat. Methods 5, 1047–1052 (2008).
Gebhardt, J.C.M. et al. Single-molecule imaging of transcription factor binding to DNA in live mammalian cells. Nat. Methods 10, 421–426 (2013).
Galland, R. et al. 3D high- and super-resolution imaging using single-objective SPIM. Nat. Methods 12, 641–644 (2015).
Li, T. et al. Axial-plane optical microscopy. Sci. Rep. 4, 7253 (2014).
Theer, P., Dragneva, D. & Knop, M. πSPIM: high-NA high-resolution isotropic light-sheet imaging in cell culture dishes. Sci. Rep. 6, 32880 (2016).
Tokunaga, M., Imamoto, N. & Sakata-Sogawa, K. Highly inclined thin illumination enables clear single-molecule imaging in cells. Nat. Methods 5, 159–161 (2008).
Gao, L. Optimization of the excitation light sheet in selective-plane illumination microscopy. Biomed. Opt. Express 6, 881–890 (2015).
Planchon, T.A. et al. Rapid three-dimensional isotropic imaging of living cells using Bessel beam plane illumination. Nat. Methods 8, 417–423 (2011).
Gao, L. et al. Non-invasive imaging beyond the diffraction limit of 3D dynamics in thickly fluorescent specimens. Cell 151, 1370–1385 (2012).
Manton, J.D. & Rees, E.J. triSPIM: light-sheet microscopy with isotropic super-resolution. Opt. Lett. 41, 4170–4173 (2016).
Zhao, T. et al. Multicolor 4D fluorescence microscopy using ultrathin Bessel light sheets. Sci. Rep. 6, 26159 (2016).
Vettenburg, T. et al. Light-sheet microscopy using an Airy beam. Nat. Methods 11, 541–544 (2014).
Zong, W. et al. Large-field high-resolution two-photon digital-scanned light-sheet microscopy. Cell Res. 25, 254–257 (2015).
Dean, K.M. & Fiolka, R. Uniform and scalable light-sheets generated by extended focusing. Opt. Express 22, 26141–26152 (2014).
Dean, K.M., Roudot, P., Welf, E.S., Danuser, G. & Fiolka, R. Deconvolution-free subcellular imaging with axially swept light-sheet microscopy. Biophys. J. 108, 2807–2815 (2015).
Buytaert, J.A. & Dirckx, J.J. Design and quantitative resolution measurements of an optical virtual sectioning three-dimensional imaging technique for biomedical specimens, featuring two-micrometer slicing resolution. J. Biomed. Opt. 12, 14039 (2011).
Gao, L. Extend the field of view of selective-plane illumination microscopy by tiling the excitation light sheet. Opt. Express 23, 6102–6111 (2015).
Fu, Q., Martin, B.L., Matus, D.Q. & Gao, L. Imaging multicellular specimens with real-time optimized tiling light-sheet selective-plane illumination microscopy. Nat. Commun. 7, 11088 (2016).
Friedrich, M., Gan, Q., Ermolayev, V. & Harms, G.S. STED-SPIM: stimulated emission depletion improves sheet-illumination microscopy resolution. Biophys. J. 100, L43–L45 (2011).
Hoyer, P. et al. Breaking the diffraction limit of light-sheet fluorescence microscopy by RESOLFT. Proc. Natl. Acad. Sci. USA 113, 3442–3446 (2016).
Palero, J., Santos, S.I.C.O., Artigas, D. & Loza-Alvarez, P. A simple scanless two-photon fluorescence microscope using selective plane illumination. Opt. Express 18, 8491–8498 (2010).
Truong, T.V., Supatto, W., Koos, D.S., Choi, J.M. & Fraser, S.E. Deep and fast live imaging with two-photon scanned light-sheet microscopy. Nat. Methods 8, 757–760 (2011).
Tomer, R., Khairy, K., Amat, F. & Keller, P.J. Quantitative high-speed imaging of entire developing embryos with simultaneous multiview light-sheet microscopy. Nat. Methods 9, 755–763 (2012).
Lemon, W.C. et al. Whole-central nervous system functional imaging in larval Drosophila. Nat. Commun. 6, 7924 (2015).
Supatto, W., Truong, T.V., Débarre, D. & Beaurepaire, E. Advances in multiphoton microscopy for imaging embryos. Curr. Opin. Genet. Dev. 21, 538–548 (2011).
Ji, N., Magee, J.C. & Betzig, E. High-speed, low-photodamage nonlinear imaging using passive pulse splitters. Nat. Methods 5, 197–202 (2008).
Mahou, P., Vermot, J., Beaurepaire, E. & Supatto, W. Multicolor two-photon light-sheet microscopy. Nat. Methods 11, 600–601 (2014).
Fahrbach, F., Simon, P. & Rohrbach, A. Microscopy with self-reconstructing beams. Nat. Photonics 4, 780–785 (2010).
Fahrbach, F.O., Gurchenkov, V., Alessandri, K., Nassoy, P. & Rohrbach, A. Self-reconstructing sectioned Bessel beams offer submicron optical sectioning for large fields of view in light-sheet microscopy. Opt. Express 21, 11425–11440 (2013).
Fahrbach, F.O., Gurchenkov, V., Alessandri, K., Nassoy, P. & Rohrbach, A. Light-sheet microscopy in thick media using scanned Bessel beams and two-photon fluorescence excitation. Opt. Express 21, 13824–13839 (2013).
Keller, P.J. et al. Fast, high-contrast imaging of animal development with scanned light-sheet-based structured-illumination microscopy. Nat. Methods 7, 637–642 (2010).
Fahrbach, F.O. & Rohrbach, A. Propagation stability of self-reconstructing Bessel beams enables contrast-enhanced imaging in thick media. Nat. Commun. 3, 632 (2012).
Silvestri, L., Bria, A., Sacconi, L., Iannello, G. & Pavone, F.S. Confocal light-sheet microscopy: micron-scale neuroanatomy of the entire mouse brain. Opt. Express 20, 20582–20598 (2012).
Baumgart, E. & Kubitscheck, U. Scanned light-sheet microscopy with confocal slit detection. Opt. Express 20, 21805–21814 (2012).
Yang, Z. et al. Dual-slit confocal light-sheet microscopy for in vivo whole-brain imaging of zebrafish. Biomed. Opt. Express 6, 1797–1811 (2015).
de Medeiros, G. et al. Confocal multiview light-sheet microscopy. Nat. Commun. 6, 8881 (2015).
Verveer, P.J. et al. High-resolution three-dimensional imaging of large specimens with light-sheet-based microscopy. Nat. Methods 4, 311–313 (2007).
Preibisch, S., Saalfeld, S., Schindelin, J. & Tomancak, P. Software for bead-based registration of selective-plane illumination microscopy data. Nat. Methods 7, 418–419 (2010).
Schmid, B. & Huisken, J. Real-time multiview deconvolution. Bioinformatics 31, 3398–3400 (2015).
Preibisch, S. et al. Efficient Bayesian-based multiview deconvolution. Nat. Methods 11, 645–648 (2014).
Swoger, J., Verveer, P., Greger, K., Huisken, J. & Stelzer, E.H.K. Multiview image fusion improves resolution in three-dimensional microscopy. Opt. Express 15, 8029–8042 (2007).
Krzic, U., Gunther, S., Saunders, T.E., Streichan, S.J. & Hufnagel, L. Multiview light-sheet microscope for rapid in toto imaging. Nat. Methods 9, 730–733 (2012).
Reynaud, E.G., Peychl, J., Huisken, J. & Tomancak, P. Guide to light-sheet microscopy for adventurous biologists. Nat. Methods 12, 30–34 (2015).
Dodt, H.-U. et al. Ultramicroscopy: three-dimensional visualization of neuronal networks in the whole mouse brain. Nat. Methods 4, 331–336 (2007).
Saghafi, S., Becker, K., Hahn, C. & Dodt, H.U. 3D-ultramicroscopy utilizing aspheric optics. J. Biophotonics 7, 117–125 (2014).
Golub, I., Chebbi, B. & Golub, J. Toward the optical 'magic carpet': reducing the divergence of a light sheet below the diffraction limit. Opt. Lett. 40, 5121–5124 (2015).
Wilding, D. et al. Pupil filters for extending the field of view in light-sheet microscopy. Opt. Lett. 41, 1205–1208 (2016).
Tomer, R., Ye, L., Hsueh, B. & Deisseroth, K. Advanced CLARITY for rapid and high-resolution imaging of intact tissues. Nat. Protoc. 9, 1682–1697 (2014).
Fahrbach, F.O., Voigt, F.F., Schmid, B., Helmchen, F. & Huisken, J. Rapid 3D light-sheet microscopy with a tunable lens. Opt. Express 21, 21010–21026 (2013).
Ahrens, M.B., Orger, M.B., Robson, D.N., Li, J.M. & Keller, P.J. Whole-brain functional imaging at cellular resolution using light-sheet microscopy. Nat. Methods 10, 413–420 (2013).
Olarte, O.E., Andilla, J., Artigas, D. & Loza-Alvarez, P. Decoupled illumination detection in light-sheet microscopy for fast volumetric imaging. Optica 2, 702 (2015).
Quirin, S. et al. Calcium imaging of neural circuits with extended depth-of-field light-sheet microscopy. Opt. Lett. 41, 855–858 (2016).
Tomer, R. et al. SPED light-sheet microscopy: fast mapping of biological system structure and function. Cell 163, 1796–1806 (2015).
Dunsby, C. Optically sectioned imaging by oblique-plane microscopy. Opt. Express 16, 20306–20316 (2008).
Bouchard, M.B. et al. Swept confocally aligned planar excitation (SCAPE) microscopy for high-speed volumetric imaging of behaving organisms. Nat. Photonics 9, 113–119 (2015).
Jahr, W., Schmid, B., Schmied, C., Fahrbach, F.O. & Huisken, J. Hyperspectral light-sheet microscopy. Nat. Commun. 6, 7990 (2015).
Cutrale, F. et al. Hyperspectral phasor analysis enables multiplexed 5D in vivo imaging. Nat. Methods 14, 149–152 (2017).
Ji, N. Adaptive optical-fluorescence microscopy for biological imaging. Nat. Methods http://dx.doi.org/10.1038/nmeth.4218 (2017).
Masson, A. et al. High-resolution in-depth imaging of optically cleared thick samples using an adaptive SPIM. Sci. Rep. 5, 16898 (2015).
Wilding, D., Pozzi, P., Soloviev, O., Vdovin, G. & Verhaegen, M. Adaptive illumination based on direct wavefront sensing in a light-sheet fluorescence microscope. Opt. Express 24, 24896–24906 (2016).
Simmonds, R.D. & Booth, M.J. Modelling of multiconjugate adaptive optics for spatially variant aberrations in microscopy. J. Opt. 15, 094010 (2013).
Royer, L.A. et al. Adaptive light-sheet microscopy for long-term, high-resolution imaging in living organisms. Nat. Biotechnol. 34, 1267–1278 (2016).
Scherf, N. & Huisken, J. The smart and gentle microscope. Nat. Biotechnol. 33, 815–818 (2015).
Hoebe, R.A. et al. Controlled light-exposure microscopy reduces photobleaching and phototoxicity in fluorescence live-cell imaging. Nat. Biotechnol. 25, 249–253 (2007).
Conrad, C. et al. Micropilot: automation of fluorescence-microscopy-based imaging for systems biology. Nat. Methods 8, 246–249 (2011).
Chmielewski, A.K. et al. Fast imaging of live organisms with sculpted light sheets. Sci. Rep. 5, 9385 (2015).
Prevedel, R. et al. Simultaneous whole-animal 3D imaging of neuronal activity using light-field microscopy. Nat. Methods 11, 727–730 (2014).
McGorty, R. et al. Open-top selective-plane illumination microscope for conventionally mounted specimens. Opt. Express 23, 16142–16153 (2015).
Chung, K., Crane, M.M. & Lu, H. Automated on-chip rapid microscopy, phenotyping and sorting of C. elegans. Nat. Methods 5, 637–643 (2008).
Pardo-Martin, C. et al. High-throughput hyperdimensional vertebrate phenotyping. Nat. Commun. 4, 1467 (2013).
Pardo-Martin, C. et al. High-throughput in vivo vertebrate screening. Nat. Methods 7, 634–636 (2010).
Gualda, E.J. et al. SPIM-fluid: open source light-sheet-based platform for high-throughput imaging. Biomed. Opt. Express 6, 4447–4456 (2015).
Heemskerk, I. & Streichan, S.J. Tissue cartography: compressing bio-image data by dimensional reduction. Nat. Methods 12, 1139–1142 (2015).
Guan, Z. et al. Compact plane illumination plugin device to enable light-sheet fluorescence imaging of multicellular organisms on an inverted wide-field microscope. Biomed. Opt. Express 7, 194–208 (2015).
Paiè, P., Bragheri, F., Bassi, A. & Osellame, R. Selective-plane illumination microscopy on a chip. Lab Chip 16, 1556–1560 (2016).
Engelbrecht, C.J., Voigt, F. & Helmchen, F. Miniaturized selective-plane illumination microscopy for high-contrast in vivo fluorescence imaging. Opt. Lett. 35, 1413–1415 (2010).
Plöschner, M. et al. Multimode fiber: light-sheet microscopy at the tip of a needle. Sci. Rep. 5, 18050 (2015).
Oshima, Y. et al. Light-sheet-excited spontaneous Raman imaging of a living fish by optical sectioning in a wide-field Raman microscope. Opt. Express 20, 16195–16204 (2012).
Rocha-Mendoza, I. et al. Rapid spontaneous Raman light-sheet microscopy using cw lasers and tunable filters. Biomed. Opt. Express 6, 3449–3461 (2015).
Yang, Z., Downie, H., Rozbicki, E., Dupuy, L.X. & MacDonald, M.P. Light sheet tomography (LST) for in situ imaging of plant roots. Opt. Express 21, 16239–16247 (2013).
Mayer, J. et al. OPTiSPIM: integrating optical projection tomography in light-sheet microscopy extends specimen characterization to nonfluorescent contrasts. Opt. Lett. 39, 1053–1056 (2014).
Bassi, A., Schmid, B. & Huisken, J. Optical tomography complements light-sheet microscopy for in toto imaging of zebrafish development. Development 142, 1016–1020 (2015).
Doerr, J. et al. Whole-brain 3D mapping of human neural transplant innervation. Nat. Commun. 8, 14162 (2017).
Engelbrecht, C.J. et al. Three-dimensional laser microsurgery in light-sheet-based microscopy (SPIM). Opt. Express 15, 6420–6430 (2007).
Yang, Z., Piksarv, P., Ferrier, D.E.K., Gunn-Moore, F.J. & Dholakia, K. Macro-optical trapping for sample confinement in light-sheet microscopy. Biomed. Opt. Express 6, 2778–2785 (2015).
Fahrbach, F.O. & Rohrbach, A. A line-scanned light-sheet microscope with phase-shaped self-reconstructing beams. Opt. Express 18, 24229–24244 (2010).
Acknowledgements
This work was supported by the Max Planck Society (R.M.P. and J.H.), the European Research Council (ERC Consolidator grant 647885; J.H.) and a fellowship from the Human Frontier Science Program (HFSP) (LT000321/2015-C; R.M.P.). We thank members of the Huisken lab for discussions and critical comments on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
J.H. is a co-inventor on patent US 20060033987 and an inventor on patent US 20110115895, which are related to light-sheet microscopy.
Supplementary information
Supplementary Text and Figures
Supplementary Figure 1, Supplementary Tables 1–5 and Supplementary Notes 1–8 (PDF 1760 kb)
Rights and permissions
About this article
Cite this article
Power, R., Huisken, J. A guide to light-sheet fluorescence microscopy for multiscale imaging. Nat Methods 14, 360–373 (2017). https://doi.org/10.1038/nmeth.4224
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmeth.4224
This article is cited by
-
An end-to-end workflow for nondestructive 3D pathology
Nature Protocols (2024)
-
Image restoration of degraded time-lapse microscopy data mediated by near-infrared imaging
Nature Methods (2024)
-
Current and future applications of light-sheet imaging for identifying molecular and developmental processes in autism spectrum disorders
Molecular Psychiatry (2024)
-
Smart lattice light-sheet microscopy for imaging rare and complex cellular events
Nature Methods (2024)
-
Collimating three-axicon zoom system for interferometric Bessel beam side lobe cancellation
Optical and Quantum Electronics (2024)