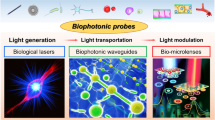
Abstract
Optofluidic biolasers are emerging as a highly sensitive way to measure changes in biological molecules. Biolasers, which incorporate biological material into the gain medium and contain an optical cavity in a fluidic environment, can use the amplification that occurs during laser generation to quantify tiny changes in biological processes in the gain medium. We describe the principle of the optofluidic biolaser, review recent progress and provide our outlooks on potential applications and directions for developing this technology.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Psaltis, D., Quake, S.R. & Yang, C. Developing optofluidic technology through the fusion of microfluidics and optics. Nature 442, 381–386 (2006).
Monat, C., Domachuk, P. & Eggleton, B.J. Integrated optofluidics: a new river of light. Nat. Photonics 1, 106–114 (2007).
Fan, X. & White, I.M. Optofluidic microsystems for chemical and biological analysis. Nat. Photonics 5, 591–597 (2011).
Schmidt, H. & Hawkins, A.R. The photonic integration of non-solid media using optofluidics. Nat. Photonics 5, 598–604 (2011).
Hawkins, A.R. & Schmidt, H. Handbook of Optofluidics (CRC Press, Boca Raton, Florida, USA, 2010).
Fainman, Y., Lee, L.P., Psaltis, D. & Yang, C. Optofluidics: Fundamentals, Devices, and Applications (McGraw-Hill, New York, 2010).
Gather, M.C. & Yun, S.H. Single-cell biological lasers. Nat. Photonics 5, 406–410 (2011). First live-cell laser.
Polson, R.C. & Vardeny, Z.V. Random lasing in human tissues. Appl. Phys. Lett. 85, 1289 (2004). Random lasers are important in dealing with disordered gain media such as tissues.
Song, Q. et al. Random lasing in bone tissue. Opt. Lett. 35, 1425–1427 (2010).
Helbo, B., Kristensen, A. & Menon, A. A micro-cavity fluidic dye laser. J. Micromech. Microeng. 13, 307–311 (2003).
Cheng, Y., Sugioka, K. & Midorikawa, K. Microfluidic laser embedded in glass by three-dimensional femtosecond laser microprocessing. Opt. Lett. 29, 2007–2009 (2004).
Balslev, S. & Kristensen, A. Microfluidic single-mode laser using high-order Bragg grating and antiguiding segments. Opt. Express 13, 344–351 (2005).
Li, Z., Zhang, Z., Emery, T., Scherer, A. & Psaltis, D. Single mode optofluidic distributed feedback dye laser. Opt. Express 14, 696–701 (2006).
Shopova, S.I., Zhu, H., Fan, X. & Zhang, P. Optofluidic ring resonator based dye laser. Appl. Phys. Lett. 90, 221101 (2007).
Shopova, S.I. et al. Opto-fluidic ring resonator lasers based on highly efficient resonant energy transfer. Opt. Express 15, 12735–12742 (2007).
Li, Z. & Psaltis, D. Optofluidic dye lasers. Microfluid. Nanofluidics 4, 145–158 (2008).
Galas, J.C., Peroz, C., Kou, Q. & Chen, Y. Microfluidic dye laser intracavity absorption. Appl. Phys. Lett. 89, 224101 (2006).
Sun, Y., Shopova, S.I., Wu, C.-S., Arnold, S. & Fan, X. Bioinspired optofluidic FRET lasers via DNA scaffolds. Proc. Natl. Acad. Sci. USA 107, 16039–16042 (2010).Optofluidic biolaser through DNA-controlled FRET processes.
Gather, M.C. & Yun, S.H. Lasing from Escherichia coli bacteria genetically programmed to express green fluorescent protein. Opt. Lett. 36, 3299–3301 (2011).
Sun, Y. & Fan, X. Distinguishing DNA by analog-to-digital-like conversion by using optofluidic lasers. Angew. Chem. Int. Ed. Engl. 51, 1236–1239 (2012).First paper to use the optofluidic biolaser for analysis of biomolecular interactions; the theoretical analysis therein laid the foundation for optofluidic biolaser sensors.
Lee, W. & Fan, X. Intracavity DNA melting analysis with optofluidic lasers. Anal. Chem. 84, 9558–9563 (2012).
Zhang, X., Lee, W. & Fan, X. Bio-switchable optofluidic lasers based on DNA Holliday junctions. Lab Chip 12, 3673–3675 (2012).Sensitive detection of biomolecule conformational change using the optofluidic biolaser.
Chen, Q. et al. Highly sensitive fluorescent protein FRET detection using optofluidic lasers. Lab Chip 13, 2679–2681 (2013).
Chen, Y. et al. Optofluidic microcavities: dye-lasers and biosensors. Biomicrofluidics 4, 043002 (2010).
Nizamoglu, S., Gather, M.C. & Yun, S.H. All-biomaterial laser using vitamin and biopolymers. Adv. Mater. doi:10.1002/adma.201300818 (31 July 2013).
Joannopoulos, J.D., Johnson, S.G., Winn, J.N. & Meade, R.D. Photonic Crystals: Molding the Flow of Light 2nd edn. (Princeton Univ. Press, 2008).
Cao, H. et al. Random laser action in semiconductor powder. Phys. Rev. Lett. 82, 2278–2281 (1999).
Cerdán, L. et al. FRET-assisted laser emission in colloidal suspensions of dye-doped latex nanoparticles. Nat. Photonics 6, 621–626 (2012).
Huang, M.H. et al. Room-temperature ultraviolet nanowire nanolasers. Science 292, 1897–1899 (2001).
Noginov, M.A. et al. Demonstration of a spaser-based nanolaser. Nature 460, 1110–1112 (2009).
Ma, R.-M., Oulton, R.F., Sorger, V.J., Bartal, G. & Zhang, X. Room-temperature sub-diffraction-limited plasmon laser by total internal reflection. Nat. Mater. 10, 110–113 (2011).
Cho, C.-H., Aspetti, C.O., Park, J. & Agarwal, R. Silicon coupled with plasmon nanocavities generates bright visible hot luminescence. Nat. Photonics 7, 285–289 (2013).
Oulton, R.F. et al. Plasmon lasers at deep subwavelength scale. Nature 461, 629–632 (2009).
Horiuchi, T., Niwa, O. & Hatakenaka, N. Evidence for laser action driven by electrochemiluminescence. Nature 394, 659–661 (1998).
Chen, Q. et al. Self-assembled DNA tetrahedral optofluidic lasers with precise and tunable gain control. Lab Chip 13, 3351–3354 (2013).
Hell, S.W. Far-field optical nanoscopy. Science 316, 1153–1158 (2007).
Wang, M.C., Min, W., Freudiger, C.W., Ruvkun, G. & Xie, X.S. RNAi screening for fat regulatory genes with SRS microscopy. Nat. Methods 8, 135–138 (2011).
Min, W. et al. Imaging chromophores with undetectable fluorescence by stimulated emission microscopy. Nature 461, 1105–1109 (2009).
Wang, P. et al. Far-field imaging of non-fluorescent species with sub-diffraction resolution. Nat. Photonics 7, 449–453 (2013).
Nadkarni, S.K. et al. Characterization of atherosclerotic plaques by laser speckle imaging. Circulation 112, 885–892 (2005).
Mason, T.G. & Weitz, D.A. Optical measurements of frequency-dependent linear viscoelastic moduli of complex fluids. Phys. Rev. Lett. 74, 1250–1253 (1995).
Siegman, A.E. Lasers (Univ. Science Books, 1986).
International Commission on Non-Ionizing Radiation Protection. Revision of guidelines on limits of exposure to laser radiation of wavelengths between 400 nm and 1.4 μm. Health Phys. 79, 431–440 (2000).
Hänsch, T.W. Edible lasers and other delights of the 1970s. Opt. Photonics News 16, 14–16 (2005).
Song, W., Vasdekis, A.E., Li, Z. & Psaltis, D. Optofluidic evanescent dye laser based on a distributed feedback circular grating. Appl. Phys. Lett. 94, 161110 (2009).
Qian, S.-X., Snow, J.B., Tzeng, H.-M. & Chang, R.K. Lasing droplets: highlighting the liquid-air interface by laser emission. Science 231, 486–488 (1986).
Moon, H.-J., Chough, Y.-T. & An, K. Cylindrical microcavity laser based on the evanescent-wave-coupled gain. Phys. Rev. Lett. 85, 3161–3164 (2000).
Azzouz, H. et al. Levitated droplet dye laser. Opt. Express 14, 4374–4379 (2006).
Kiraz, A. et al. Lasing from single, stationary, dye-doped glycerol/water microdroplets located on a superhydrophobic surface. Opt. Commun. 276, 145–148 (2007).
Jiang, X., Song, Q., Xu, L., Fu, J. & Tong, L. Microfiber knot dye laser based on the evanescent-wave-coupled gain. Appl. Phys. Lett. 90, 233501 (2007).
Tanyeri, M., Perron, R. & Kennedy, I.M. Lasing droplets in a microfabricated channel. Opt. Lett. 32, 2529–2531 (2007).
Tang, S.K.Y. et al. A multi-color fast-switching microfluidic droplet dye laser. Lab Chip 9, 2767–2771 (2009).
Schäfer, J. et al. Quantum dot microdrop laser. Nano Lett. 8, 1709–1712 (2008).
Lee, W., Luo, Y., Zhu, Q. & Fan, X. Versatile optofluidic ring resonator lasers based on microdroplets. Opt. Express 19, 19668–19674 (2011).
Yang, Y. et al. A tunable 3D optofluidic waveguide dye laser via two centrifugal Dean flow streams. Lab Chip 11, 3182–3187 (2011).
Aubry, G. et al. A multicolor microfluidic droplet dye laser with single mode emission. Appl. Phys. Lett. 98, 111111 (2011).
Christiansen, M.B., Kristensen, A., Xiao, S. & Mortensen, N.A. Photonic integration in k-space: Enhancing the performance of photonic crystal dye lasers. Appl. Phys. Lett. 93, 231101 (2008).
Wu, X., Chen, Q., Sun, Y. & Fan, X. Bio-inspired optofluidic lasers with luciferin. Appl. Phys. Lett. 102, 203706 (2013).
Lacey, S. et al. Versatile opto-fluidic ring resonator lasers with ultra-low threshold. Opt. Express 15, 15523–15530 (2007).
Acknowledgements
The authors acknowledge support from the US National Science Foundation (grants CBET-1037097 and ECCS-1045621 and CBET-1158638 to X.F. and ECCS-1101947 and CBET-1264356 to S.-H.Y.) and US National Institutes of Health (P41EB015903 to S.-H.Y.). We thank D. Psaltis, Z. Li and M. Gather for providing original figures (Figs. 2b and 4b) and I. White for proofreading the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Fan, X., Yun, SH. The potential of optofluidic biolasers. Nat Methods 11, 141–147 (2014). https://doi.org/10.1038/nmeth.2805
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmeth.2805
This article is cited by
-
Single-particle photoacoustic vibrational spectroscopy using optical microresonators
Nature Photonics (2023)
-
Highly nonlinear optic nucleic acid thin-solid film to generate short pulse laser
Scientific Reports (2023)
-
Self-organized lasers from reconfigurable colloidal assemblies
Nature Physics (2022)
-
Sensitivity and spectral control of network lasers
Nature Communications (2022)
-
Deep tissue localization and sensing using optical microcavity probes
Nature Communications (2022)