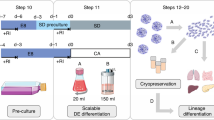
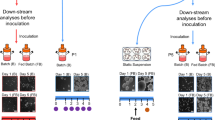
Abstract
We describe derivation of induced pluripotent stem cells (iPSCs) from terminally differentiated mouse cells in serum- and feeder-free stirred suspension cultures. Temporal analysis of global gene expression revealed high correlations between cells reprogrammed in suspension and cells reprogrammed in adhesion-dependent conditions. Suspension culture–reprogrammed iPSCs (SiPSCs) could be differentiated into all three germ layers in vitro and contributed to chimeric embryos in vivo. SiPSC generation allowed for efficient selection of reprogramming factor–expressing cells based on their differential survival and proliferation in suspension culture. Seamless integration of SiPSC reprogramming and directed differentiation enabled scalable production of beating cardiac cells in a continuous single cell– and small aggregate–based process. This method is an important step toward the development of robust PSC generation, expansion and differentiation technology.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Accession codes
References
Takahashi, K. et al. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 131, 861–872 (2007).
Takahashi, K. & Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126, 663–676 (2006).
Yu, J. et al. Induced pluripotent stem cell lines derived from human somatic cells. Science 318, 1917–1920 (2007).
Kirouac, D.C. & Zandstra, P.W. The systematic production of cells for cell therapies. Cell Stem Cell 3, 369–381 (2008).
Shapiro, A.M. et al. Islet transplantation in seven patients with type 1 diabetes mellitus using a glucocorticoid-free immunosuppressive regimen. N. Engl. J. Med. 343, 230–238 (2000).
Taylor, C.J. et al. Banking on human embryonic stem cells: estimating the number of donor cell lines needed for HLA matching. Lancet 366, 2019–2025 (2005).
Stadtfeld, M., Nagaya, M., Utikal, J., Weir, G. & Hochedlinger, K. Induced pluripotent stem cells generated without viral integration. Science 322, 945–949 (2008).
Yu, J. et al. Human induced pluripotent stem cells free of vector and transgene sequences. Science 324, 797–801 (2009).
Zhou, H. et al. Generation of induced pluripotent stem cells using recombinant proteins. Cell Stem Cell 4, 381–384 (2009).
Warren, L. et al. Highly efficient reprogramming to pluripotency and directed differentiation of human cells with synthetic modified mRNA. Cell Stem Cell 7, 618–630 (2010).
Woltjen, K. et al. piggyBac transposition reprograms fibroblasts to induced pluripotent stem cells. Nature 458, 766–770 (2009).
Maherali, N. & Hochedlinger, K. Guidelines and techniques for the generation of induced pluripotent stem cells. Cell Stem Cell 3, 595–605 (2008).
Steiner, D. et al. Derivation, propagation and controlled differentiation of human embryonic stem cells in suspension. Nat. Biotechnol. 28, 361–364 (2010).
Fok, E.Y. & Zandstra, P.W. Shear-controlled single-step mouse embryonic stem cell expansion and embryoid body-based differentiation. Stem Cells 23, 1333–1342 (2005).
Andang, M., Moliner, A., Doege, C.A., Ibanez, C.F. & Ernfors, P. Optimized mouse ES cell culture system by suspension growth in a fully defined medium. Nat. Protoc. 3, 1013–1017 (2008).
Zweigerdt, R., Olmer, R., Singh, H., Haverich, A. & Martin, U. Scalable expansion of human pluripotent stem cells in suspension culture. Nat. Protoc. 6, 689–700 (2011).
Samavarchi-Tehrani, P. et al. Functional genomics reveals a BMP-driven mesenchymal-to-epithelial transition in the initiation of somatic cell reprogramming. Cell Stem Cell 7, 64–77 (2010).
Song, H. et al. Interrogating functional integration between injected pluripotent stem cell-derived cells and surrogate cardiac tissue. Proc. Natl. Acad. Sci. USA 107, 3329–3334 (2010).
Kattman, S.J. et al. Stage-specific optimization of activin/nodal and BMP signaling promotes cardiac differentiation of mouse and human pluripotent stem cell lines. Cell Stem Cell 8, 228–240 (2011).
Dang, S.M., Gerecht-Nir, S., Chen, J., Itskovitz-Eldor, J. & Zandstra, P.W. Controlled, scalable embryonic stem cell differentiation culture. Stem Cells 22, 275–282 (2004).
Amit, M. et al. Dynamic suspension culture for scalable expansion of undifferentiated human pluripotent stem cells. Nat. Protoc. 6, 572–579 (2011).
Brambrink, T. et al. Sequential expression of pluripotency markers during direct reprogramming of mouse somatic cells. Cell Stem Cell 2, 151–159 (2008).
Stadtfeld, M., Maherali, N., Breault, D.T. & Hochedlinger, K. Defining molecular cornerstones during fibroblast to iPS cell reprogramming in mouse. Cell Stem Cell 2, 230–240 (2008).
Sommer, C.A. et al. Excision of reprogramming transgenes improves the differentiation potential of iPS cells generated with a single excisable vector. Stem Cells 28, 64–74 (2010).
Maherali, N. et al. A high-efficiency system for the generation and study of human induced pluripotent stem cells. Cell Stem Cell 3, 340–345 (2008).
Belteki, G. et al. Conditional and inducible transgene expression in mice through the combinatorial use of Cre-mediated recombination and tetracycline induction. Nucleic Acids Res. 33, e51 (2005).
Ungrin, M.D., Joshi, C., Nica, A., Bauwens, C. & Zandstra, P.W. Reproducible, ultra high-throughput formation of multicellular organization from single cell suspension-derived human embryonic stem cell aggregates. PLoS One 3, e1565 (2008).
Mitta, B. et al. Advanced modular self-inactivating lentiviral expression vectors for multigene interventions in mammalian cells and in vivo transduction. Nucleic Acids Res. 30, e113 (2002).
Huang, W., Sherman, B.T. & Lempicki, R.A. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat. Protoc. 4, 44–57 (2009).
Watanabe, K. et al. Directed differentiation of telencephalic precursors from embryonic stem cells. Nat. Neurosci. 8, 288–296 (2005).
Acknowledgements
We thank members of the Toronto Centre for Phenogenomics for chimera generation and K. Woltjen (Kyoto University) for providing 6C secondary fibroblasts as well as providing PB-TET-cMyc and PB-TET-MKOS plasmids. PMF351 was provided by M. Fussenegger (Swiss Federal Institute of Technology). This work was funded by Natural Sciences and Engineering Research Council of Canada, the Canadian Institute of Health Research (MOP-57885) and an Ontario Research Fund Global Leadership Round in Genomics and Life Sciences award to A.N.; D.A.F. was supported by a McLean award to P.W.Z.; P.W.Z. was supported as the Canada Research Chair in Stem Cell Bioengineering.
Author information
Authors and Affiliations
Contributions
D.A.F. designed and performed most experiments. P.D.T. performed chimera experiments, part of quantitative PCR experiments and contributed to drafting the manuscript. H.S. performed cTnT differentiation experiments and did confocal imaging. R.P.B. performed western blot analysis and did part of the secondary MEF, spleen and adult fibroblast reprogramming experiments. N.S. did part of the secondary MEF and spleen reprogramming experiments. S.S. performed secondary DN1 blood reprogramming experiments. G.C. performed statistical analysis. A.N. helped to design the project. D.A.F. and P.W.Z. designed the project and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8, Supplementary Tables 1–3 (PDF 7538 kb)
Supplementary Video 1
Spontaneously beating aggregates generated from primary fibroblast cells reprogrammed and differentiated towards the cardiac lineage in an integrated suspension-culture process. (MOV 391 kb)
Rights and permissions
About this article
Cite this article
Fluri, D., Tonge, P., Song, H. et al. Derivation, expansion and differentiation of induced pluripotent stem cells in continuous suspension cultures. Nat Methods 9, 509–516 (2012). https://doi.org/10.1038/nmeth.1939
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmeth.1939
This article is cited by
-
Organoids
Nature Reviews Methods Primers (2022)
-
“Cutting the Mustard” with Induced Pluripotent Stem Cells: An Overview and Applications in Healthcare Paradigm
Stem Cell Reviews and Reports (2022)
-
Critical Analysis of cGMP Large-Scale Expansion Process in Bioreactors of Human Induced Pluripotent Stem Cells in the Framework of Quality by Design
BioDrugs (2021)
-
Engineering human ventricular heart muscles based on a highly efficient system for purification of human pluripotent stem cell-derived ventricular cardiomyocytes
Stem Cell Research & Therapy (2017)
-
Induced pluripotent stem cells: at the heart of cardiovascular precision medicine
Nature Reviews Cardiology (2016)