Abstract
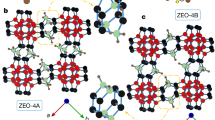
Layered silicates with three-dimensional microporosity within the layers have the potential to enable new applications in catalysis, adsorption and ion-exchange. Until now no such materials have been reported. However, here we present the synthesis and structure of AMH-3, a silicate with three-dimensionally microporous layers, obtained in high purity and crystallinity. AMH-3 is composed of silicate layers containing eight-membered rings in all three principal crystal directions, and spaced by strontium cations, sodium cations and water molecules. Because of its three-dimensional pore structure, acid and thermal stability, this layered material could find applications in polymer–silicate composites for membrane applications, for synthesis of combined microporous–mesoporous materials, and for the formation of new zeolites and microporous films. Its existence also opens new possibilities for the synthesis of other layered silicates with multidimensional microporous framework layers.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Carrado, K.A. Synthetic organo- and polymer-clays: preparation, characterization, and materials applications. Appl. Clay Sci. 17, 1–23 (2000).
Corma, A. From microporous to mesoporous molecular sieve materials and their use in catalysis. Chem. Rev. 97, 2373–2419 (1997).
Kloprogge, J.T. Synthesis of smectites and porous pillared clay catalysts: A review. J. Porous Mater. 5, 5–41 (1998).
Leonowicz, M.E., Lawton, J.A., Lawton, S.L. & Rubin, M.K. MCM-22 - a molecular-sieve with 2 independent multidimensional channel systems. Science 264, 1910–1913 (1994).
Corma, A., Diaz, U., Domine, M.E. & Fornes, V. New aluminosilicate and titanosilicate delaminated materials active for acid catalysis, and oxidation reactions using H2O2 . J. Am. Chem. Soc. 122, 2804–2809 (2000).
Corma, A., Fornes, V., Martinez-Triguero, J. & Pergher, S.B. Delaminated zeolites: Combining the benefits of zeolites and mesoporous materials for catalytic uses. J. Catal. 186, 57–63 (1999).
Wolf, I., Gies, H. & Fyfe, C.A. Ordering of silicate layers by hydrogen-bonding networks: Solid state NMR investigation of the perfect three-dimensional registration in the layer silicate RUB-18. J. Phys. Chem. B 103, 5933–5938 (1999).
Corma, A., Fornes, V., Pergher, S.B., Maesen, T.L.M. & Buglass, J.G. Delaminated zeolite precursors as selective acidic catalysts. Nature 396, 353–356 (1998).
Krishnamoorti, R., Vaia, R.A. & Giannelis, E.P. Structure and dynamics of polymer-layered silicate nanocomposites. Chem. Mater. 8, 1728–1734 (1996).
Umemura, Y., Yamagishi, A., Schoonheydt, R., Persoons, A. & De Schryver, F. Fabrication of hybrid films of alkylammonium cations (CnH2n+1 NH3+; n=4–18) and a smectite clay by the Langmuir-Blodgett method. Langmuir 17, 449–455 (2001).
Wang, Z. & Pinnavaia, T.J. Hybrid organic-inorganic nanocomposites: Exfoliation of magadiite nanolayers in an elastomeric epoxy polymer. Chem. Mater. 10, 1820–1826 (1998).
Yano, K., Usuki, A., Okada, A., Kurauchi, T. & Kamigaito, O. Synthesis and properties of polyimide clay hybrid. J. Polym. Sci. A 31, 2493–2498 (1993).
Cussler, E.L., Hughes, S.E., Ward, W.J. & Aris, R. Barrier membranes. J. Membrane Sci. 38, 161–174 (1988).
Kan, Q.B., Fornes, V., Rey, F. & Corma, A. Transformation of layered aluminosilicates and gallosilicates with kanemite structure into mesoporous materials. J. Mater. Chem. 10, 993–1000 (2000).
Kuroda, K. Orientation of guest molecules and formation of mesoporous silica induced by layered silicate-organic interactions. Mol. Cryst. Liq. Cryst. 341, 1087–1092 (2000).
van Koningsveld, H., Vanbekkum, H. & Jansen, J.C. On the location and disorder of the tetrapropylammonium (TPA) ion in zeolite ZSM-5 with improved framework accuracy. Acta Crystallogr. B 43, 127–132 (1987).
van koningsveld, H. High-Temperature (350-K) orthorhombic framework structure of zeolite H-ZSM-5. Acta Crystallogr. B 46, 731–735 (1990).
Brese, N.E. & Okeeffe, M. Bond-valence parameters for solids. Acta Crystallogr. B 47, 192–197 (1991).
Engelhardt, G. & Michel, D. High-Resolution Solid-State NMR of Silicates and Zeolites (Wiley, New York, 1987).
Merlino, S. Lovdarite, K4Na12(Be8Si28O72).18H2O, a Zeolite-like mineral - structural features and OD character. Eur. J. Mineral. 2, 809–817 (1990).
Altomare, A. et al. EXTRA: A program for extracting structure factor amplitudes from powder diffraction data. J. Appl. Crystallogr. 28, 842–846 (1995).
Altomare, A. et al. EXPO: a program for full powder pattern decomposition and crystal structure solution. J. Appl. Crystallogr. 32, 339–340 (1999).
Larson, A.C. & von Dreele, R.B. (Los Alamos National Laboratory, Los Alamos NM, 1986).
Stegun, I.A. (ed.) Handbook of Mathematical Functions (US Government Printing Office, Washington DC, 1972).
Rappe, A.K., Casewit, C.J., Colwell, K.S., Goddard, W.A. & Skiff, W.M. Uff, a full periodic-table force-field for molecular mechanics and molecular-dynamics simulations. J. Am. Chem. Soc. 114, 10024–10035 (1992).
Rappe, A.K. & Goddard, W.A. Charge equilibration for molecular-dynamics simulations. J. Phys. Chem. 95, 3358–3363 (1991).
Schaak, R.E. & Mallouk, T.E. Perovskites by design: A toolbox of solid-state reactions. Chem. Mater. 14, 1455–1471 (2002).
Yao, Y.W., Natarajan, S., Chen, J.S. & Pang, W.Q. Synthesis and structural characterization of a new layered aluminophosphate intercalated with triply-protonated triethylenetetramine C6H21N4 Al3P4O16 . J. Solid State Chem. 146, 458–463 (1999).
Gao, Q.M. et al. Nonaqueous synthesis and characterization of a new 2- dimensional layered aluminophosphate Al3P4O16 3 CH3CH2NH3 . J. Solid State Chem. 129, 37–44 (1997).
Vidal, L., Marichal, C., Gramlich, V., Patarin, J. & Gabelica, Z. Mu-7, a new layered aluminophosphate CH3NH3 (3) Al3P4O16 with a 4 x 8 network: Characterization, structure, and possible crystallization mechanism. Chem. Mater. 11, 2728–2736 (1999).
Wei, B., Yu, J.H., Shi, Z., Qiu, S.L. & Li, J.Y. A new layered aluminophosphate Al2P4O16 C6H22N4 C2H10N2 with 4.12-net porous sheets. J. Chem. Soc. Dalton Trans. 1979–1980 (2000).
Yuan, H.M. et al. Dual function of racemic isopropanolamine as solvent and as template for the synthesis of a new layered aluminophosphate: NH3CH2CH(OH)CH3 Al3P4O16 . J. Solid State Chem. 151, 145–149 (2000).
Jiang, T., Lough, A.J., Ozin, G.A., Young, D. & Bedard, R.L. Synthesis and structure of the novel nanoporous tin(IV) sulfide material TPA-SnS-3. Chem. Mater. 7, 245–248 (1995).
Bowes, C.L. et al. Microporous layered tin sulfide, SnS-1: molecular sieve or intercalant? J. Mater. Chem. 8, 711–720 (1998).
Burnett, M.N. & Johnson, C.K. ORTEP-III: Oak ridge thermal ellipsoid plot program for crystal structure illustrations. Oak Ridge National Laboratory Report ORNL-6895 (1996).
Acknowledgements
We acknowledge support from NASA-Microgravity (98-HEDS-05-218), NSF (CTS 0091406) and Engelhard Co.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Jeong, HK., Nair, S., Vogt, T. et al. A highly crystalline layered silicate with three-dimensionally microporous layers. Nature Mater 2, 53–58 (2003). https://doi.org/10.1038/nmat795
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmat795
This article is cited by
-
Nickel(II) ion-intercalated MXene membranes for enhanced H2/CO2 separation
Frontiers of Chemical Science and Engineering (2021)
-
MXene molecular sieving membranes for highly efficient gas separation
Nature Communications (2018)
-
Mesoporous catalytic membranes: Synthetic control of pore size and wall composition
Catalysis Letters (2005)