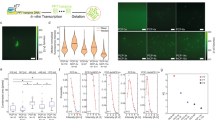
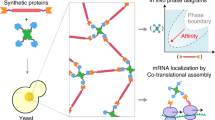
Abstract
Some protein components of intracellular non-membrane-bound entities, such as RNA granules, are known to form hydrogels in vitro. The physico-chemical properties and functional role of these intracellular hydrogels are difficult to study, primarily due to technical challenges in probing these materials in situ. Here, we present iPOLYMER, a strategy for a rapid induction of protein-based hydrogels inside living cells that explores the chemically inducible dimerization paradigm. Biochemical and biophysical characterizations aided by computational modelling show that the polymer network formed in the cytosol resembles a physiological hydrogel-like entity that acts as a size-dependent molecular sieve. We functionalize these polymers with RNA-binding motifs that sequester polyadenine-containing nucleotides to synthetically mimic RNA granules. These results show that iPOLYMER can be used to synthetically reconstitute the nucleation of biologically functional entities, including RNA granules in intact cells.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Hoffman, A. S. Hydrogels for biomedical applications. Adv. Drug Deliv. Rev. 54, 3–12 (2002).
Frantz, C., Stewart, K. M. & Weaver, V. M. The extracellular matrix at a glance. J. Cell Sci. 123, 4195–4200 (2010).
Lieleg, O. & Ribbeck, K. Biological hydrogels as selective diffusion barriers. Trends Cell Biol. 21, 543–551 (2011).
Lai, S. K., Wang, Y.-Y., Wirtz, D. & Hanes, J. Micro- and macrorheology of mucus. Adv. Drug Deliv. Rev. 61, 86–100 (2009).
Weber, S. C. & Brangwynne, C. P. Getting RNA and protein in phase. Cell 149, 1188–1191 (2012).
Kato, M. et al. Cell-free formation of RNA granules: low complexity sequence domains form dynamic fibers within hydrogels. Cell 149, 753–767 (2012).
Hyman, A. A. & Simons, K. Cell biology. Beyond oil and water–phase transitions in cells. Science 337, 1047–1049 (2012).
Frey, S., Richter, R. P. & Görlich, D. FG-rich repeats of nuclear pore proteins form a three-dimensional meshwork with hydrogel-like properties. Science 314, 815–817 (2006).
Brangwynne, C. P. et al. Germline P granules are liquid droplets that localize by controlled dissolution/condensation. Science 324, 1729–1732 (2009).
Aggarwal, S. et al. Myelin membrane assembly is driven by a phase transition of myelin basic proteins into a cohesive protein meshwork. PLoS Biol. 11, e1001577 (2013).
Sackmann, E. How actin/myosin crosstalks guide the adhesion, locomotion and polarization of cells. Biochim. Biophys. Acta 1853, 3132–3142 (2015).
Deek, J., Chung, P. J., Kayser, J., Bausch, A. R. & Safinya, C. R. Neurofilament sidearms modulate parallel and crossed-filament orientations inducing nematic to isotropic and re-entrant birefringent hydrogels. Nat. Commun. 4, 2224 (2013).
Brangwynne, C. P., Mitchison, T. J. & Hyman, A. A. Active liquid-like behavior of nucleoli determines their size and shape in Xenopus laevis oocytes. Proc. Natl Acad. Sci. USA 108, 4334–4339 (2011).
Han, T. W. et al. Cell-free formation of RNA granules: bound RNAs identify features and components of cellular assemblies. Cell 149, 768–779 (2012).
Patel, A. et al. A liquid-to-solid phase transition of the ALS protein FUS accelerated by disease mutation. Cell 162, 1066–1077 (2015).
Molliex, A. et al. Phase separation by low complexity domains promotes stress granule assembly and drives pathological fibrillization. Cell 163, 123–133 (2015).
Lin, Y., Protter, D. S. W., Rosen, M. K. & Parker, R. Formation and maturation of phase-separated liquid droplets by RNA-binding proteins. Mol. Cell 60, 208–219 (2015).
Xiang, S. et al. The LC domain of hnRNPA2 adopts similar conformations in hydrogel polymers, liquid-like droplets, and nuclei. Cell 163, 829–839 (2015).
Murakami, T. et al. ALS/FTD mutation-induced phase transition of FUS liquid droplets and reversible hydrogels into irreversible hydrogels impairs RNP granule function. Neuron 88, 678–690 (2015).
Zhang, H. et al. RNA controls PolyQ protein phase transitions. Mol. Cell 60, 220–230 (2015).
Kwon, I. et al. Poly-dipeptides encoded by the C9orf72 repeats bind nucleoli, impede RNA biogenesis, and kill cells. Science 345, 1139–1145 (2014).
Li, Y. R., King, O. D., Shorter, J. & Gitler, A. D. Stress granules as crucibles of ALS pathogenesis. J. Cell Biol. 201, 361–372 (2013).
Wippich, F. et al. Dual specificity kinase DYRK3 couples stress granule condensation/dissolution to mTORC1 signaling. Cell 152, 791–805 (2013).
Khademhosseini, A. & Langer, R. Microengineered hydrogels for tissue engineering. Biomaterials 28, 5087–5092 (2007).
Hoare, T. R. & Kohane, D. S. Hydrogels in drug delivery: progress and challenges. Polymer 49, 1993–2007 (2008).
Iwasaki, T. & Wang, Y.-L. Cytoplasmic force gradient in migrating adhesive cells. Biophys. J. 94, L35–L37 (2008).
Yang, Z. M., Xu, K. M., Guo, Z. F., Guo, Z. H. & Xu, B. Intracellular enzymatic formation of nanofibers results in hydrogelation and regulated cell death. Adv. Mater. 19, 3152–3156 (2007).
Hülsmann, B. B., Labokha, A. A. & Görlich, D. The permeability of reconstituted nuclear pores provides direct evidence for the selective phase model. Cell 150, 738–751 (2012).
DeRose, R., Miyamoto, T. & Inoue, T. Manipulating signaling at will: chemically-inducible dimerization (CID) techniques resolve problems in cell biology. Pflüg. Arch. Eur. J. Physiol. 465, 409–417 (2013).
Tanaka, F. Polymer Physics: Applications to Molecular Association and Thermoreversible Gelation (Cambridge Univ. Press, 2011).
Li, P. et al. Phase transitions in the assembly of multivalent signalling proteins. Nature 483, 336–340 (2012).
Lin, Y.-C. et al. Rapidly reversible manipulation of molecular activity with dual chemical dimerizers. Angew. Chem. Int. Ed Engl. 52, 6450–6454 (2013).
Lin, Y.-C. et al. Chemically inducible diffusion trap at cilia reveals molecular sieve-like barrier. Nat. Chem. Biol. 9, 437–443 (2013).
McLauchlan, H. J., James, J., Lucocq, J. M. & Ponnambalam, S. Characterization and regulation of constitutive transport intermediates involved in trafficking from the trans-Golgi network. Cell Biol. Int. 25, 705–713 (2001).
Kedersha, N., Ivanov, P. & Anderson, P. Stress granules and cell signaling: more than just a passing phase? Trends Biochem. Sci. 38, 494–506 (2013).
Souquere, S. et al. Unravelling the ultrastructure of stress granules and associated P-bodies in human cells. J. Cell Sci. 122, 3619–3626 (2009).
Gilks, N. et al. Stress granule assembly is mediated by prion-like aggregation of TIA-1. Mol. Biol. Cell 15, 5383–5398 (2004).
Kedersha, N. L., Gupta, M., Li, W., Miller, I. & Anderson, P. RNA-binding proteins TIA-1 and TIAR link the phosphorylation of eIF-2 α to the assembly of mammalian stress granules. J. Cell Biol. 147, 1431–1442 (1999).
Kedersha, N. & Anderson, P. Mammalian stress granules and processing bodies. Methods Enzymol. 431, 61–81 (2007).
Kimball, S. R., Horetsky, R. L., Ron, D., Jefferson, L. S. & Harding, H. P. Mammalian stress granules represent sites of accumulation of stalled translation initiation complexes. Am. J. Physiol. Cell Physiol. 284, C273–C284 (2003).
Guntas, G. et al. Engineering an improved light-induced dimer (iLID) for controlling the localization and activity of signaling proteins. Proc. Natl Acad. Sci. USA 112, 112–117 (2015).
Taslimi, A. et al. An optimized optogenetic clustering tool for probing protein interaction and function. Nat. Commun. 5, 4925 (2014).
Lee, S. et al. Reversible protein inactivation by optogenetic trapping in cells. Nat. Methods 11, 633–636 (2014).
Miyazaki, Y. et al. A method to rapidly create protein aggregates in living cells. Nat. Commun. 7, 11689 (2016).
Inobe, T. & Nukina, N. Rapamycin-induced oligomer formation system of FRB-FKBP fusion proteins. J. Biosci. Bioeng. 122, 40–46 (2016).
Shin, Y. et al. Spatiotemporal control of intracellular phase transitions using light-activated optoDroplets. Cell 168, 159–171 (2017).
Jucker, M. & Walker, L. C. Self-propagation of pathogenic protein aggregates in neurodegenerative diseases. Nature 501, 45–51 (2013).
Lesniak, W. G. et al. Biodistribution of fluorescently labeled PAMAM dendrimers in neonatal rabbits: effect of neuroinflammation. Mol. Pharm. 10, 4560–4571 (2013).
Banaszynski, L. A., Liu, C. W. & Wandless, T. J. Characterization of the FKBP.rapamycin.FRB ternary complex. J. Am. Chem. Soc. 127, 4715–4721 (2005).
Doi, M. Second quantization representation for classical many-particle system. J. Phys. Math. Gen. 9, 1465–1477 (1976).
Doi, M. Stochastic theory of diffusion-controlled reaction. J. Phys. Math. Gen. 9, 1479–1495 (1976).
Isaacson, S. A. A convergent reaction–diffusion master equation. J. Chem. Phys. 139, 54101 (2013).
Isaacson, S. A. & Peskin, C. S. Incorporating diffusion in complex geometries into stochastic chemical kinetics simulations. SIAM J. Sci. Comput. 28, 47–74 (2006).
Isaacson, S. A. Relationship between the reaction–diffusion master equation and particle tracking models. J. Phys. Math. Theor. 41, 65003 (2008).
Fange, D., Berg, O. G., Sjöberg, P. & Elf, J. Stochastic reaction–diffusion kinetics in the microscopic limit. Proc. Natl Acad. Sci. USA 107, 19820–19825 (2010).
Hellander, S., Hellander, A. & Petzold, L. Reaction–diffusion master equation in the microscopic limit. Phys. Rev. E Stat. Nonlin. Soft Matter Phys. 85, 42901 (2012).
Acknowledgements
We are grateful to N. Kedersha and P. Anderson who provided helpful discussions and reagents related to stress granules, to J. L. Pfaltz who collaborated with A.S.A. to develop a modified C++ code for identifying chordless cycles in graphs, and to R. Reed, A. Ewald, H. Sesaki, M. Iijima and S. Regot for sharing their resources for our experiments. We also extend our appreciation to J. P. Gong, I. Hamachi, R. Yoshida for valuable comments on our work. This work was mainly supported by the Johns Hopkins University Catalyst Fund to T.I., and in part by the National Institutes of Health (NIH) (GM092930, DK102910, CA103175 and DK089502 to T.I., and T32GM007445 to A.S.), and the National Science Foundation (NSF) (CCF-1217213 to J.G.).
Author information
Authors and Affiliations
Contributions
H.N., A.A.L. and T.I. conceived the project. H.N., A.A.L., A.S., Y.-C.L., M.T., R.D. and D.B., performed molecular biology as well as cell biology experiments. H.N., A.A.L., S.R. and A.S. purified proteins under the guidance of W.H. and S.B.G. The biochemical and biophysical experiments were mostly performed by H.N. and A.A.L., and partially by S.R. and Y.-C.L. H.N., A.A.L. and T.I. wrote the manuscript with the help of J.G. A.S.A. and J.G. developed the computational model, analysed the computational results, and wrote the computational parts of the paper. A.S.A. wrote appropriate code and conducted the computational experiments. S.W. performed correlated EM measurement and analysis. E.R. and B.H. performed development and demonstration of light-inducible iPOLYMER with H.N.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 5698 kb)
Supplementary Information
Supplementary movie 1 (AVI 5224 kb)
Supplementary Information
Supplementary movie 2 (AVI 4778 kb)
Supplementary Information
Supplementary movie 3 (AVI 4679 kb)
Supplementary Information
Supplementary movie 4 (AVI 4747 kb)
Supplementary Information
Supplementary movie 5 (AVI 4808 kb)
Supplementary Information
Supplementary movie 6 (AVI 616 kb)
Supplementary Information
Supplementary movie 7 (AVI 6101 kb)
Supplementary Information
Supplementary movie 8 (AVI 10182 kb)
Rights and permissions
About this article
Cite this article
Nakamura, H., Lee, A., Afshar, A. et al. Intracellular production of hydrogels and synthetic RNA granules by multivalent molecular interactions. Nature Mater 17, 79–89 (2018). https://doi.org/10.1038/nmat5006
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmat5006
This article is cited by
-
A platform to induce and mature biomolecular condensates using chemicals and light
Nature Chemical Biology (2024)
-
Rational design of phase separating peptides based on phase separating protein sequence of p53
Scientific Reports (2023)
-
RNAs undergo phase transitions with lower critical solution temperatures
Nature Chemistry (2023)
-
3D Models of Sarcomas: The Next-generation Tool for Personalized Medicine
Phenomics (2023)
-
Phase separation in immune signalling
Nature Reviews Immunology (2022)