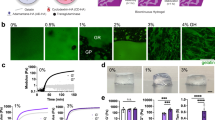
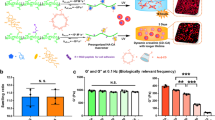
Abstract
Existing techniques to encapsulate cells into microscale hydrogels generally yield high polymer-to-cell ratios and lack control over the hydrogel’s mechanical properties1. Here, we report a microfluidic-based method for encapsulating single cells in an approximately six-micrometre layer of alginate that increases the proportion of cell-containing microgels by a factor of ten, with encapsulation efficiencies over 90%. We show that in vitro cell viability was maintained over a three-day period, that the microgels are mechanically tractable, and that, for microscale cell assemblages of encapsulated marrow stromal cells cultured in microwells, osteogenic differentiation of encapsulated cells depends on gel stiffness and cell density. We also show that intravenous injection of singly encapsulated marrow stromal cells into mice delays clearance kinetics and sustains donor-derived soluble factors in vivo. The encapsulation of single cells in tunable hydrogels should find use in a variety of tissue engineering and regenerative medicine applications.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Selimovic, S., Oh, J., Bae, H., Dokmeci, M. & Khademhosseini, A. Microscale strategies for generating cell-encapsulating hydrogels. Polymers 4, 1554 (2012).
Martinez, C. J. et al. A microfluidic approach to encapsulate living cells in uniform alginate hydrogel microparticles. Macromol. Biosci. 12, 946–951 (2012).
Tan, W. H. & Takeuchi, S. Monodisperse alginate hydrogel microbeads for cell encapsulation. Adv. Mater. 19, 2696–2701 (2007).
Chung, B. G., Lee, K., Khademhosseini, A. & Lee, S. Microfluidic fabrication of microengineered hydrogels and their application in tissue engineering. Lab Chip 12, 45–59 (2012).
Velasco, D., Tumarkin, E. & Kumacheva, E. Microfluidic encapsulation of cells in polymer microgels. Small 8, 1633–1642 (2012).
Wu, L., Chen, P., Dong, Y., Feng, X. & Liu, B. F. Encapsulation of single cells on a microfluidic device integrating droplet generation with fluorescence-activated droplet sorting. Biomed. Microdevices 15, 553–560 (2013).
Edd, J. F. et al. Controlled encapsulation of single-cells into monodisperse picolitre drops. Lab Chip 8, 1262–1264 (2008).
Kemna, E. W. M. et al. High-yield cell ordering and deterministic cell-in-droplet encapsulation using Dean flow in a curved microchannel. Lab Chip 12, 2881–2997 (2012).
Veerabadran, N. G., Goli, P. L., Stewart-Clark, S. S., Lvov, Y. M. & Mills, D. K. Nanoencapsulation of stem cells within polyelectrolyte multilayer shells. Macromol. Biosci. 7, 877–882 (2007).
Wilson, J. T. et al. Cell surface engineering with polyelectrolyte multilayer thin films. J. Am. Chem. Soc. 133, 7054–7064 (2011).
Tatsumi, K. et al. The non-invasive cell surface modification of hepatocytes with PEG-lipid derivatives. Biomaterials 33, 821–828 (2012).
Teramura, Y., Oommen, O. P., Olerud, J., Hilborn, J. & Nilsson, B. Microencapsulation of cells, including islets, within stable ultra-thin membranes of maleimide-conjugated PEG-lipid with multifunctional crosslinkers. Biomaterials 34, 2683–2693 (2013).
Huebsch, N. et al. Harnessing traction-mediated manipulation of the cell/matrix interface to control stem-cell fate. Nat. Mater. 9, 518–526 (2010).
Lee, Y. L. & Mooney, D. J. Alginate: properties and biomedical applications. Prog. Polym. Sci. 37, 106–126 (2012).
Köster, S. et al. Drop-based microfluidic devices for encapsulation of single cells. Lab Chip 8, 1110–1115 (2008).
Holtze, C. et al. Biocompatible surfactants for water-in-fluorocarbon emulsions. Lab Chip 8, 1632–1639 (2008).
Nakano, T., Kodama, H. & Honjo, T. Generation of lymphohematopoietic cells from embryonic stem cells in culture. Science 265, 1098–1101 (1994).
Choi, K. et al. Hematopoietic and endothelial differentiation of human induced pluripotent stem cells. Stem Cells 27, 559–567 (2009).
Fernando, L. P. et al. Mechanism of cellular uptake of highly fluorescent conjugated polymer nanoparticles. Biomacromolecules 11, 2675–2682 (2010).
De la Vega, J. C., Elischer, P., Schneider, T. & Häfeli, U. O. Uniform polymer microspheres: monodispersity criteria, methods of formation and applications. Nanomedicine 8, 265–285 (2013).
Rowley, J. A., Madlambayan, G. & Mooney, D. J. Alginate hydrogels as synthetic extracellular matrix materials. Biomaterials 20, 45–53 (1999).
Chaudhuri, O. et al. Substrate stress relaxation regulates cell spreading. Nat. Commun. 6, 6365 (2015).
Desai, R. M. et al. Versatile click alginate hydrogels crosslinked via tetrazine-norbornene chemistry. Biomaterials 20, 30–37 (2015).
Steinhilber, D. et al. A microgel construction kit for bioorthogonal encapsulation and pH-controlled release of living cells. Angew. Chem. Int. Ed. 52, 13538–13543 (2013).
Chaudhuri, O. et al. Extracellular matrix stiffness and composition jointly regulate the induction of malignant phenotypes in mammary epithelium. Nat. Mater. 13, 970–978 (2014).
Karoubi, G., Ormiston, M. I., Stewart, D. J. & Courtman, D. W. Single-cell hydrogel encapsulation for enhanced survival of human marrow stromal cells. Biomaterials 30, 5445–5455 (2009).
Orive, G. et al. Cell encapsulation: promise and progress. Nat. Med. 9, 104–107 (2003).
Ma, M. et al. Core-shell hydrogel microscapsules for improved islets encapsulation. Adv. Healthc. Mater. 2, 667–672 (2013).
Pareta, R. A., McQuilling, J. P., Farney, A. C. & Opara, E. C. in Regenerative Medicine Applications in Organ Transplantation 1st edn (eds Orlando, G., Lerut, J., Soker, S. & Stratta, R. J.) 627–635 (Academic, 2014).
Fischer, U. M. et al. Pulmonary passage is a major obstacle for intravenous stem cell delivery: the pulmonary first-pass effect. Stem Cells Dev. 18, 683–691 (2009).
Wurdinger, T. A secreted luciferase for ex vivo monitoring of in vivo processes. Nat. Methods 5, 171–173 (2008).
Acknowledgements
This work was supported by the National Institutes of Health (NIH) Grants RO1EB014703 (D.J.M. and D.A.W.) and K99HL125884 (J.-W.S.), and the National Science Foundation (NSF) Graduate Research Fellowship Program (A.S.M.). S.U. was supported by the Deutsche Forschungsgemeinschaft (DFG).
Author information
Authors and Affiliations
Contributions
A.S.M., J.-W.S. and D.J.M. conceived and designed the experiments. S.U., H.W. and D.A.W. contributed to microfluidic design and fabrication. A.S.M. and J.-W.S. performed the experiments. A.S.M., J.-W.S. and D.J.M. analysed the data. A.S.M., J.-W.S. and D.J.M. wrote the manuscript. All authors discussed the results and commented on the manuscript. A.S.M. and J.-W.S. contributed equally to this work. The principal investigator is D.J.M.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 2659 kb)
Rights and permissions
About this article
Cite this article
Mao, A., Shin, JW., Utech, S. et al. Deterministic encapsulation of single cells in thin tunable microgels for niche modelling and therapeutic delivery. Nature Mater 16, 236–243 (2017). https://doi.org/10.1038/nmat4781
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmat4781
This article is cited by
-
Utilizing bioprinting to engineer spatially organized tissues from the bottom-up
Stem Cell Research & Therapy (2024)
-
High-throughput fabrication of antimicrobial phage microgels and example applications in food decontamination
Nature Protocols (2024)
-
Cell–extracellular matrix mechanotransduction in 3D
Nature Reviews Molecular Cell Biology (2023)
-
Middle-out methods for spatiotemporal tissue engineering of organoids
Nature Reviews Bioengineering (2023)
-
Bioprinting microporous functional living materials from protein-based core-shell microgels
Nature Communications (2023)