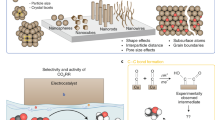
Abstract
Advances in electrocatalysis at solid–liquid interfaces are vital for driving the technological innovations that are needed to deliver reliable, affordable and environmentally friendly energy. Here, we highlight the key achievements in the development of new materials for efficient hydrogen and oxygen production in electrolysers and, in reverse, their use in fuel cells. A key issue addressed here is the degree to which the fundamental understanding of the synergy between covalent and non-covalent interactions can form the basis for any predictive ability in tailor-making real-world catalysts. Common descriptors such as the substrate–hydroxide binding energy and the interactions in the double layer between hydroxide-oxides and H---OH are found to control individual parts of the hydrogen and oxygen electrochemistry that govern the efficiency of water-based energy conversion and storage systems. Links between aqueous- and organic-based environments are also established, encouraging the 'fuel cell' and 'battery' communities to move forward together.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Schlapbach, L. & Zuttel, A. Hydrogen-storage materials for mobile applications. Nature 414, 353–358 (2001).
Schlapbach, L. Global Research Center for Environment and Energy Based on Nanomaterials Science (National Institute for Materials Science, 2013).
Kinoshita, D. K. Electrochemical Oxygen Technology (Wiley, 1992).
Dresselhaus, M. S. & Thomas, I. L. Alternative energy technologies. Nature 414, 332–337 (2001).
Gratzel, M. Photoelectrochemical cells. Nature 414, 338–344 (2001).
Tarascon, J.-M. & Armand, M. Issues and challenges facing rechargeable lithium batteries. Nature 414, 359–367 (2001).
Steele, B. C. & Heinzel, A. Materials for fuel-cell technologies. Nature 414, 345–352 (2001).
Tarasevich, M. R., Sadkowski, A. & Yeager, E. in Comprehensive Treatise of Electrochemistry Vol. 7 (Conway, B. E., Bockris, J. O., Yeager, E., Khan, S. U. M. & White, R. E.) 301–398 (Springer, 1983).
Adzic, R. R. Reaction Kinetics and Mechanisms on Metal Single-Crystal Electrode Surfaces (Plenum, 1990).
Markovic, N. M. & Ross, P. N. New electrocatalysts for fuel cells from model surfaces to commercial catalysts. CATTECH 4, 110–126 (2000).
Markovic, N. M. & Ross Jr., P. N. Surface science studies of model fuel cell electrocatalysts. Surf. Sci. Rep. 45, 117–229 (2002).
Koper, M. T. M. Electrocatalysis: Theory and experiment at the interface. Faraday Discuss. 140, 11–24 (2008).
Greeley, J. & Markovic, N. M. The road from animal electricity to green energy: combining experiment and theory in electrocatalysis. Energy Environ. Sci. 5, 9246–9256 (2012).
Markovic, N. M. Electrocatalysis: Interfacing electrochemistry. Nat. Mater. 12, 101–102 (2013).
Müller-Dethlefs, K. & Hobza, P. Noncovalent interactions: A challenge for experiment and theory. Chem. Rev. 100, 143–167 (2000).
Strmcnik, D. et al. The role of non-covalent interactions in electrocatalytic fuel-cell reactions on platinum. Nat. Chem. 1, 466–472 (2009).
Verma, P., Maire, P. & Novák, P. A review of the features and analyses of the solid electrolyte interphase in Li-ion batteries. Electrochim. Acta 55, 6332–6341 (2010).
Aurbach, D., Zinigrad, E., Cohen, Y. & Teller, H. A short review of failure mechanisms of lithium metal and lithiated graphite anodes in liquid electrolyte solutions. Solid State Ion. 148, 405–416 (2002).
Kordesch, K. Hydrogen–oxygen fuel cells with carbon electrodes. Ind. Eng. Chem. 52, 296–298 (1960).
Bacon, F. T. The high pressure hydrogen–oxygen fuel cell. Ind. Eng. Chem. 52, 301–303 (1960).
Gasteiger, H. A., Kocha, S. S., Sompalli, B. & Wagner, F. T. Activity benchmarks and requirements for Pt, Pt-alloy, and non-Pt oxygen reduction catalysts for PEMFCs. Appl. Catal. B Environ. 56, 9–35 (2005).
Koper, M. T. M. Fuel Cell Catalysis: A Surface Science Approach (Wiley, 2009).
Vielstich, W., Lamm, A., Gasteiger, H. A. & Yokokawa, H. Handbook of Fuel Cells (Wiley, 2009).
Gasteiger, H. A. & Markovic, N. M. Just a dream — or future reality? Science 324, 48–49 (2009).
Debe, M. K. Electrocatalyst approaches and challenges for automotive fuel cells. Nature 486, 43–51 (2012).
Walter, M. G. et al. Solar water splitting cells. Chem. Rev. 110, 6446–6473 (2010).
Nocera, D. G. The artificial leaf. Acc. Chem. Res. 45, 767–776 (2012).
Schalenbach, M., Carmo, M., Fritz, D. L., Mergel, J. & Stolten, D. Pressurized PEM water electrolysis: Efficiency and gas crossover. Int. J. Hydrogen Energy 38, 14921–14933 (2013).
Carmo, M., Fritz, D. L., Mergel, J. & Stolten, D. A comprehensive review on PEM water electrolysis. Int. J. Hydrogen Energy 38, 4901–4934 (2013).
Whittingham, M. S. Lithium batteries and cathode materials. Chem. Rev. Rev 104, 4271–4301 (2004).
Goodenough, J. B. Rechargeable batteries: Challenges old and new. J. Solid State Electrochem. 16, 2019–2029 (2012).
Goodenough, J. B. Energy storage in a sustainable modern society. Energy Environ. Sci. 7, 14–18 (2014).
Rosenman, A. et al. Review on Li–sulfur battery systems: An integral perspective. Adv. Energy Mater. 5, 1500212 (2015).
Wild, M. et al. Lithium sulfur batteries, a mechanistic review. Energy Environ. Sci. 8, 3477–3494 (2015).
Fotouhi, A., Auger, D. J., Propp, K., Longo, S. & Wild, M. A review on electric vehicle battery modelling: From lithium-ion toward lithium–sulphur. Renew. Sustain. Energy Rev. 56, 1008–1021 (2016).
Abraham, K. M. & Jiang, Z. A polymer electrolyte-based rechargeable lithium/oxygen battery. J. Electrochem. Soc. 143, 1–5 (1996).
Bruce, P. G., Freunberger, S. A., Hardwick, L. J. & Tarascon, J.-M. Li–O2 and Li–S batteries with high energy storage. Nat. Mater. 11, 172–172 (2011).
Luntz, A. C. & McCloskey, B. D. Nonaqueous Li–air batteries: a status report. Chem. Rev. 114, 11721–11750 (2014).
Younesi, R., Veith, G. M., Johansson, P., Edstrom, K. & Vegge, T. Lithium salts for advanced lithium batteries: Li–metal, Li–O2, and Li–S. Energy Environ. Sci. 8, 1905–1922 (2015).
Lu, J. et al. A lithium–oxygen battery based on lithium superoxide. Nature 529, 377–382 (2016).
Pickard, W. F., Shen, A. Q. & Hansing, N. J. Parking the power: Strategies and physical limitations for bulk energy storage in supply-demand matching on a grid whose input power is provided by intermittent sources. Renew. Sustain. Energy Rev. 13, 1934–1945 (2009).
Abdin, Z., Webb, C. J. & Gray, E. M. Solar hydrogen hybrid energy systems for off-grid electricity supply: A critical review. Renew. Sustain. Energy Rev. 52, 1791–1808 (2015).
Moorhouse, J. Modern Chlor-Alkali Technology. (Wiley, 2001).
O'Brien, T., Bommaraju, T. V. & Hine, F. Handbook of Chlor-Alkali Technology Vol. 4. (Kluwer Academic/Plenum, 2005).
Kordesch, K. et al. Alkaline fuel cells applications. J. Power Sources 86, 162–165 (2000).
Borup, R. et al. Scientific aspects of polymer electrolyte fuel cell durability and degradation. Chem. Rev. 107, 3904–3951 (2007).
Wang, Y., Chen, K. S., Mishler, J., Cho, S. C. & Adroher, X. C. A review of polymer electrolyte membrane fuel cells: Technology, applications, and needs on fundamental research. Appl. Energy 88, 981–1007 (2011).
Hickner, M. A., Herring, A. M. & Coughlin, E. B. Anion exchange membranes: Current status and moving forward. J. Polym. Sci. B 51, 1727–1735 (2013).
Varcoe, J. R. et al. Anion-exchange membranes in electrochemical energy systems. Energy Environ. Sci. 7, 3135–3191 (2014).
Zhang, H. W., Chen, D. Z., Xianze, Y. & Yin, S. B. Anion-exchange membranes for fuel cells: Synthesis strategies, properties and perspectives. Fuel Cells 15, 761–780 (2015).
He, Q. & Cairns, E. J. Review: Recent progress in electrocatalysts for oxygen reduction suitable for alkaline anion exchange membrane fuel cells. J. Electrochem. Soc. 162, F1504–F1539 (2015).
Wagner, F. T., Lakshmanan, B. & Mathias, M. F. Electrochemistry and the future of the automobile. J. Phys. Chem. Lett. 1, 2204–2219 (2010).
Gröger, O., Gasteiger, H. A. & Suchsland, J.-P. Review: Electromobility: Batteries or fuel cells? J. Electrochem. Soc. 162, A2605–A2622 (2015).
Parsons, R. The rate of electrolytic hydrogen evolution and the heat of adsorption of hydrogen. Trans. Farad. Soc. 54, 1053–1063 (1958).
Gerischer, H. Mechanism of electrolytic discharge of hydrogen and adsorption energy of atomic hydrogen. Bull. Soc. Chim. Belg. 67, 506 (1958).
Bockris, J. & Otagawa, T. Mechanism of oxygen evolution on perovskites. J. Phys. Chem. 87, 2960–2971 (1983).
Trasatti, S. Electrocatalysis in the anodic evolution of oxygen and chlorine. Electrochim. Acta 29, 1503–1512 (1984).
Rossmeisl, J., Logadottir, A. & Nørskov, J. K. Electrolysis of water on (oxidized) metal surfaces. Chem. Phys. 319, 178–184 (2005).
Norskov, J. K., Bligaard, T., Rossmeisl, J. & Christensen, C. H. Towards the computational design of solid catalysts. Nat. Chem. 1, 37–46 (2009).
Santos, E., Quaino, P. & Schmickler, W. Theory of electrocatalysis: Hydrogen evolution and more. Phys. Chem. Chem. Phys. 14, 11224–33 (2012).
Trasatti, S. Work function, electronegativity, and electrochemical behaviour of metals: III. Electrolytic hydrogen evolution in acid solutions. J. Electroanal. Chem. Interfacial Electrochem. 39, 163–184 (1972).
Conway, B. E. & Tilak, B. V. Interfacial processes involving electrocatalytic evolution and oxidation of H2, and the role of chemisorbed H. Electrochim. Acta 47, 3571–3594 (2002).
Birry, L. & Lasia, A. Studies of the hydrogen evolution reaction on Raney nickel–molybdenum electrodes. J. Appl. Electrochem. 34, 735–749 (2004).
Danilovic, N. et al. Enhancing the alkaline hydrogen evolution reaction activity through the bifunctionality of Ni(OH)2/metal catalysts. Angew. Chem. Int. Ed. 51, 12495–12498 (2012).
Strmcnik, D. et al. Improving the hydrogen oxidation reaction rate by promotion of hydroxyl adsorption. Nat. Chem. 5, 300–306 (2013).
McCrory, C. C. L. et al. Benchmarking HER and OER electrocatalysts for solar water splitting devices. J. Am. Chem. Soc. 137, 4347–4357 (2015).
Greeley, J. et al. Computational high-throughput screening of electrocatalytic materials for hydrogen evolution. Nat. Mater. 5, 909–913 (2006).
Cabán-Acevedo, M. et al. Efficient hydrogen evolution catalysis using ternary pyrite-type cobalt phosphosulphide. Nat. Mater. 14, 1245–51 (2015).
Jaramillo, T. F. et al. Identification of active edge sites for electrochemical H2 evolution from MoS2 nanocatalysts. Science 317, 100–102 (2007).
Kibsgaard, J., Chen, Z., Reinecke, B. N. & Jaramillo, T. F. Engineering the surface structure of MoS2 to preferentially expose active edge sites for electrocatalysis. Nat. Mater. 11, 963–969 (2012).
Kibsgaard, J., Jaramillo, T. F. & Besenbacher, F. Building an appropriate active-site motif into a hydrogen-evolution catalyst with thiomolybdate [Mo3S13]2− clusters. Nat. Chem. 6, 248–253 (2014).
Faber, M. S. et al. High-performance electrocatalysis using metallic cobalt pyrite (CoS2) micro- and nanostructures. J. Am. Chem. Soc. 136, 10053–10061 (2014).
Faber, M. S. & Jin, S. Earth-abundant inorganic electrocatalysts and their nanostructures for energy conversion applications. Energy Environ. Sci. 7, 3519–3542 (2014).
Li, H. et al. Activating and optimizing MoS2 basal planes for hydrogen evolution through the formation of strained sulphur vacancies. Nat. Mater. 15, 48–53 (2015).
Staszak-Jirkovský, J. et al. Design of active and stable Co–Mo–Sx chalcogels as pH-universal catalysts for the hydrogen evolution reaction. Nat. Mater. 15, 197–204 (2015).
Subbaraman, R. et al. Enhancing hydrogen evolution activity in water splitting by tailoring Li+–Ni(OH)2–Pt interfaces. Science 334, 1256–60 (2011).
Subbaraman, R. et al. Trends in activity for the water electrolyser reactions on 3d M(Ni,Co,Fe,Mn) hydr(oxy)oxide catalysts. Nat. Mater. 11, 550–7 (2012).
Petrii, O. A. & Tsirlina, G. A. Electrocatalytic activity prediction for hydrogen electrode reaction: intuition, art, science. Electrochim. Acta 39, 1739–1747 (1994).
Quaino, P., Juarez, F., Santos, E. & Schmickler, W. Volcano plots in hydrogen electrocatalysis — uses and abuses. Beilstein J. Nanotechnol. 5, 846–854 (2014).
Markovic, N. M., Sarraf, S. T., Gasteigert, H. A. & Ross, P. N. Hydrogen electrochemsitry on platinum low-index single crystal surfaces in alkaline solution. J. Chem. Soc. Faraday Trans. 92, 3719–3725 (1996).
Marković, N. M., Grgur, B. N. & Ross, P. N. Temperature-dependent hydrogen electrochemistry on platinum low-index single-crystal surfaces in acid solutions. J. Phys. Chem. B 101, 5405–5413 (1997).
Sheng, W., Gasteiger, H. A. & Shao-Horn, Y. Hydrogen oxidation and evolution reaction kinetics on platinum: Acid vs alkaline electrolytes. J. Electrochem. Soc. 157, B1529–B1536 (2010).
Strmcnik, D., Lopes, P. P., Genorio, B., Stamenkovic, V. R. & Markovic, N. M. Design principles for hydrogen evolution reaction catalyst materials. Nano Energy http://dx.doi.org/10.1016/j.nanoen.2016.04.017 (2016).
Subbaraman, R. et al. Enhancing hydrogen evolution activity in water splitting by tailoring Li+–Ni(OH)2–Pt interfaces. Science 334, 1256–1260 (2011).
Henderson, M. A. The interaction of water with solid surfaces: Fundamental aspects revisited. Surf. Sci. Rep. 46, 1–308 (2002).
Bligaard, T. et al. The Brønsted–Evans–Polanyi relation and the volcano curve in heterogeneous catalysis. J. Catal. 224, 206–217 (2004).
Surendranath, Y. & Nocera, D. G. in Progress in Inorganic Chemistry Vol. 57, 505–560 (2011).
Danilovic, N. et al. Using surface segregation to design stable Ru–Ir oxides for the oxygen evolution reaction in acidic environments. Angew. Chem. Int. Ed. 53, 14016–14021 (2014).
Fabbri, E. et al. Developments and perspectives of oxide-based catalysts for the oxygen evolution reaction. Catal. Sci. Technol. 4, 3800–3821 (2014).
Jiao, Y., Zheng, Y., Jaroniec, M. & Qiao, S. Z. Design of electrocatalysts for oxygen- and hydrogen-involving energy conversion reactions. Chem. Soc. Rev. 44, 2060–2086 (2015).
Matsumoto, Y. & Sato, E. Electrocatalytic properties of transition metal oxides for oxygen evolution reaction. Mater. Chem. Phys. 14, 397–426 (1986).
Singh, N. K., Tiwari, S. K., Anitha, K. L. & Singh, R. N. Electrocatalytic properties of spinel-type Mn(x)Fe(3–x)O4 synthesized below 100 °C for oxygen evolution in KOH solutions. J. Chem. Soc. Farad. Trans. 92, 2397–2400 (1996).
Nikolov, I. et al. Electrocatalytic activity of spinel related cobalties MxCo3−xO4 (M = Li, Ni, Cu) in the oxygen evolution reaction. J. Electroanal. Chem. 429, 157–168 (1997).
Suntivich, J. et al. Design principles for oxygen-reduction activity on perovskite oxide catalysts for fuel cells and metal–air batteries. Nat. Chem. 3, 546–550 (2011).
Hong, W. T. et al. Toward the rational design of non-precious transition metal oxides for oxygen electrocatalysis. Energy Environ. Sci. 8, 1404–1427 (2015).
Singh, R. N., Pandey, J. P., Singh, N. K., Lal, B. & Chartier, P. Sol-gel derived spinel MxCo3−xO4 (M = Ni, Cu; 0< x < 1) films and oxygen evolution. Electrochim. Acta 45, 1911–1919 (2000).
Forgie, R., Bugosh, G., Neyerlin, K. C., Liu, Z. & Strasser, P. Bimetallic Ru electrocatalysts for the OER and electrolytic water splitting in acidic media. Electrochem. Solid-State Lett. 13, B36–B39 (2010).
Suntivich, J., May, K. J., Gasteiger, H. a, Goodenough, J. B. & Shao-horn, Y. A perovskite oxide optimized for molecular orbital principles. Science 334, 2010–2012 (2011).
Chang, S. H. et al. Functional links between stability and reactivity of strontium ruthenate single crystals during oxygen evolution. Nat. Commun. 5, 4191 (2014).
Danilovic, N. et al. Activity–stability trends for the oxygen evolution reaction on monometallic oxides in acidic environments. J. Phys. Chem. Lett. 5, 2474–2478 (2014).
Cherevko, S. et al. Dissolution of noble metals during oxygen evolution in acidic media. ChemCatChem 6, 2219–2223 (2014).
Chang, S. H. et al. Activity–stability relationship in the surface electrochemistry of the oxygen evolution reaction. Faraday Discuss. 176, 125–133 (2015).
Binninger, T. et al. Thermodynamic explanation of the universal correlation between oxygen evolution activity and corrosion of oxide catalysts. Sci. Rep. 5, 12167 (2015).
Lopes, P. P. et al. Relationships between atomic level surface structure and stability/activity of platinum surface atoms in aqueous environments. ACS Catal. 6, 2536–2544 (2016).
Nørskov, J. K. et al. Origin of the overpotential for oxygen reduction at a fuel-cell cathode. J. Phys. Chem. B 108, 17886–17892 (2004).
Stamenkovic, V. et al. Changing the activity of electrocatalysts for oxygen reduction by tuning the surface electronic structure. Angew. Chemie Int. Ed. 45, 2897–2901 (2006).
Adzic, R. R. et al. Platinum monolayer fuel cell electrocatalysts. Top. Catal. 46, 249–262 (2007).
Greeley, J. et al. Alloys of platinum and early transition metals as oxygen reduction electrocatalysts. Nat. Chem. 1, 552–556 (2009).
Calle-Vallejo, F. et al. Finding optimal surface sites on heterogeneous catalysts by counting nearest neighbors. Science 350, 185–189 (2015).
Bashyam, R. & Zelenay, P. A class of non-precious metal composite catalysts for fuel cells. Nature 443, 63–66 (2006).
Lefèvre, M., Proietti, E., Jaouen, F. & Dodelet, J.-P. Iron-based catalysts with improved oxygen reduction activity in polymer electrolyte fuel cells. Science 324, 71–74 (2009).
Jaouen, F. et al. Recent advances in non-precious metal catalysis for oxygen-reduction reaction in polymer electrolyte fuel cells. Energy Environ. Sci. 4, 114 (2011).
Shao, M., Chang, Q., Dodelet, J. & Chenitz, R. Recent advances in electrocatalysts for oxygen reduction reaction. Chem. Rev. 116, 3594–3657 (2016).
Auinger, M. et al. Near-surface ion distribution and buffer effects during electrochemical reactions. Phys. Chem. Chem. Phys. 13, 16384–16394 (2011).
Durst, J. et al. New insights into the electrochemical hydrogen oxidation and evolution reaction mechanism. Energy Environ. Sci. 7, 2255–2260 (2014).
Rossmeisl, J., Chan, K., Skúlason, E., Björketun, M. E. & Tripkovic, V. On the pH dependence of electrochemical proton transfer barriers. Catal. Today 262, 36–40 (2016).
Watanabe, M. & Motoo, S. Electrocatalysis by ad-atoms. Part III. Enhancement of the oxidation of carbon monoxide on platinum by ruthenium ad-atoms. J. Electroanal. Chem. Interfacial Electrochem. 61, 147–153 (1975).
Gasteiger, H. A., Markovic, N., Ross, P. N. & Cairns, E. J. Methanol electrooxidation on well-characterized Pt–Ru alloys. J. Phys. Chem. 97, 12020–12029 (1993).
Gasteiger, H. A., Markovic, N. M. & Ross, P. N. H2 and CO electrooxidation on well-characterized Pt, Ru, and Pt–Ru. 1. Rotating disk electrode studies of the pure gases including temperature effects. J. Phys. Chem. 99, 8290–8301 (1995).
Marković, N. M., Schmidt, T. J., Stamenković, V. & Ross, P. N. Oxygen reduction reaction on Pt and Pt bimetallic surfaces: A selective review. Fuel Cells 1, 105–116 (2001).
Paulus, U. A. et al. Oxygen reduction on high surface area Pt-based alloy catalysts in comparison to well defined smooth bulk alloy electrodes. Electrochim. Acta 47, 3787–3798 (2002).
Paulus, U. A. et al. Oxygen reduction on carbon-supported Pt–Ni and Pt–Co alloy catalysts. J. Phys. Chem. B 106, 4181–4191 (2002).
Stamenkovic, V. R. et al. Improved oxygen reduction activity on Pt3Ni(111) via increased surface site availability. Science 315, 493–7 (2007).
Srivastava, R., Mani, P., Hahn, N. & Strasser, P. Efficient oxygen reduction fuel cell electrocatalysis on voltammetrically dealloyed Pt–Cu–Co nanoparticles. Angew. Chem. Int. Ed. 46, 8988–8991 (2007).
Zhang, J., Vukmirovic, M. B., Xu, Y., Mavrikakis, M. & Adzic, R. R. Controlling the catalytic activity of platinum-monolayer electrocatalysts for oxygen reduction with different substrates. Angew. Chem. Int. Ed. 44, 2132–2135 (2005).
Yang, L. et al. Tuning the catalytic activity of Ru@Pt core−shell nanoparticles for the oxygen reduction reaction by varying the shell thickness. J. Phys. Chem. C 117, 1748–1753 (2013).
Li, D. et al. Functional links between Pt single crystal morphology and nanoparticles with di ff erent size and shape: The oxygen reduction reaction case. Energy Environ. Sci. 7, 4061–4069 (2014).
Huang, X. et al. High-performance transition metal-doped Pt3Ni octahedra for oxygen reduction reaction. Science 348, 1230–1234 (2014).
Stamenkovic, V. R. et al. Trends in electrocatalysis on extended and nanoscale Pt-bimetallic alloy surfaces. Nat. Mater. 6, 241–247 (2007).
van der Vliet, D. F. et al. Unique electrochemical adsorption properties of Pt-skin surfaces. Angew. Chemie Int. Ed. 51, 3139–3142 (2012).
Arenz, M., Schmidt, T. J., Wandelt, K., Ross, P. N. & Markovic, N. M. The oxygen reduction reaction on thin palladium films supported on a Pt(111) electrode. J. Phys. Chem. B 107, 9813–9819 (2003).
van der Vliet, D. F. et al. Mesostructured thin films as electrocatalysts with tunable composition and surface morphology. Nat. Mater. 11, 1051–1058 (2012).
Wang, C. et al. Monodisperse Pt(3)Co nanoparticles as electrocatalyst: The effects of particle size and pretreatment on electrocatalytic reduction of oxygen. Phys. Chem. Chem. Phys. 12, 6933–6939 (2010).
Wang, C. et al. Multimetallic Au/FePt3 nanoparticles as highly durable electrocatalyst. Nano Lett. 11, 919–926 (2011).
Kang, Y. et al. Multimetallic core/interlayer/shell nanostructures as advanced electrocatalysts. Nano Lett. 14, 6361–6367 (2014).
Wang, C. et al. Correlation between surface chemistry and electrocatalytic properties of monodisperse PtxNi1-x nanoparticles. Adv. Funct. Mater. 21, 147–152 (2011).
Chen, C. et al. Highly crystalline multimetallic nanoframes with three-dimensional electrocatalytic surfaces. Science 343, 1339–1343 (2014).
Yoshida, T. & Kojima, K. Toyota MIRAI fuel cell vehicle and progress toward a future hydrogen society. Interface 24, 45–49 (2015).
Zurilla, R. W., Sen, R. K. & Yeager, E. The kinetics of the oxygen reduction reaction on gold in alkaline solution. J. Electrochem. Soc. 125, 1103–1109 (1978).
Adzić, R. R., Marković, N. M. & Vešović, V. B. Structural effects in electrocatalysis: Oxygen reduction on the Au(100) single crystal electrode. J. Electroanal. Chem. Interfacial Electrochem. 165, 105–120 (1984).
Adžić, R. R., Strbac, S. & Anastasijević, N. Electrocatalysis of oxygen on single crystal gold electrodes. Mater. Chem. Phys. 22, 349–375 (1989).
Sawyer, D. T. & Roberts, J. L. Electrochemistry of oxygen and superoxide ion in dimethylsulfoxide at platinum, gold and mercury electrodes. J. Electroanal. Chem. 12, 90–101 (1966).
Staszak-Jirkovský, J. et al. Water as a promoter and catalyst for dioxygen electrochemistry in aqueous and organic media. ACS Catal. 5, 6600–6607 (2015).
Genorio, B. et al. Superoxide (electro)chemistry on well-defined surfaces in organic environments. J. Phys. Chem. C http://dx.doi.org/10.1021/acs.jpcc.5b12230 (2016).
Shterenberg, I. et al. Evaluation of (CF3SO2)2N− (TFSI) based electrolyte solutions for Mg batteries. J. Electrochem. Soc. 162, 7118–7128 (2015).
Tepavcevic, S. et al. Nanostructured layered cathode for rechargeable Mg-ion batteries. ACS Nano 9, 8194–8205 (2015).
Strmcnik, D. et al. When small is big: The role of impurities in electrocatalysis. Top. Catal. 58, 1174–1180 (2015).
Lamy, C. et al. Recent advances in the development of direct alcohol fuel cells (DAFC). J. Power Sources 105, 283–296 (2002).
Schouten, K. J. P., Calle-Vallejo, F. & Koper, M. T. M. A step closer to the electrochemical production of liquid fuels. Angew. Chem. Int. Ed. Engl. 53, 10858–10860 (2014).
Rosca, V., Duca, M., DeGroot, M. T. & Koper, M. T. M. Nitrogen cycle electrocatalysis. Chem. Rev. 109, 2209–2244 (2009).
Acknowledgements
The research was conducted at Argonne National Laboratory, a US Department of Energy Office of Science laboratory, operated by UChicago Argonne, LLC, under contract no. DE-AC02-06CH11357. We acknowledge support from the Office of Science, Office of Basic Energy Sciences, Materials Sciences and Engineering Division, the Office of Energy Efficiency and Renewable Energy, Fuel Cell Technologies Program and from the Joint Center of Energy Storage Research (JCESR), an Energy Innovation Hub funded by the US Department of Energy.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Stamenkovic, V., Strmcnik, D., Lopes, P. et al. Energy and fuels from electrochemical interfaces. Nature Mater 16, 57–69 (2017). https://doi.org/10.1038/nmat4738
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmat4738
This article is cited by
-
A strongly coupled Ru–CrOx cluster–cluster heterostructure for efficient alkaline hydrogen electrocatalysis
Nature Catalysis (2024)
-
Understanding entropic barriers
Nature Energy (2024)
-
Active learning guides discovery of a champion four-metal perovskite oxide for oxygen evolution electrocatalysis
Nature Materials (2024)
-
Nitrogen–sulfur co-doped porous carbon derived from asphalt as efficient catalyst for oxygen reduction
Rare Metals (2024)
-
Navigating surface reconstruction of spinel oxides for electrochemical water oxidation
Nature Communications (2023)