Abstract
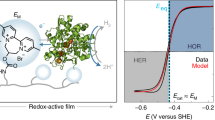
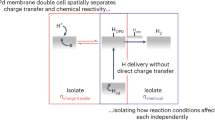
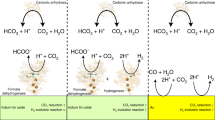
Many chemical and biological processes involve the transfer of both protons and electrons. The complex mechanistic details of these proton-coupled electron transfer (PCET) reactions require independent control of both electron and proton transfer. In this report, we make use of lipid-modified electrodes to modulate proton transport to a Cu-based catalyst that facilitates the O2 reduction reaction (ORR), a PCET process important in fuel cells and O2 reduction enzymes. By quantitatively controlling the kinetics of proton transport to the catalyst, we demonstrate that undesired side products such as H2O2 and O2− arise from a mismatch between proton and electron transfer rates. Whereas fast proton kinetics induce H2O2 formation and sluggish proton flux produces O2−, proton transfer rates commensurate with O–O bond breaking rates ensure that only the desired H2O product forms. This fundamental insight aids in the development of a comprehensive framework for understanding the ORR and PCET processes in general.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Weinberg, D. R. et al. Proton-coupled electron transfer. Chem. Rev. 112, 4016–4093 (2012).
Chang, C. J., Chang, M. C. Y., Damrauer, N. H. & Nocera, D. G. Proton-coupled electron transfer: a unifying mechanism for biological charge transport, amino acid radical initiation and propagation, and bond making/breaking reactions of water and oxygen. Biochim. Biophys. Acta 1655, 13–28 (2004).
Hammes-Schiffer, S. & Soudackov, A. V. Proton-coupled electron transfer in solution, proteins, and electrochemistry. J. Phys. Chem. B 112, 14108–14123 (2008).
Costentin, C., Robert, M. & Savéant, J.-M. Update 1 of: Electrochemical approach to the mechanistic study of proton-coupled electron transfer. Chem. Rev. 110, PR1–PR40 (2010).
Hammes-Schiffer, S. Theory of proton-coupled electron transfer in energy conversion processes. Acc. Chem. Res. 42, 1881–1889 (2009).
Horvath, S., Fernandez, L. E., Soudackov, A. V. & Hammes-Schiffer, S. Insights into proton-coupled electron transfer mechanisms of electrocatalytic H2 oxidation and production. Proc. Natl Acad. Sci. USA 109, 15663–15668 (2012).
Jaouen, F. et al. Recent advances in non-precious metal catalysis for oxygen-reduction reaction in polymer electrolyte fuel cells. Energy Environ. Sci. 4, 114–130 (2011).
Marcus, R. A. Electron transfer reactions in chemistry. Theory and experiment. Rev. Mod. Phys. 65, 599–610 (1993).
Migliore, A., Polizzi, N. F., Therien, M. J. & Beratan, D. N. Biochemistry and theory of proton-coupled electron transfer. Chem. Rev. 114, 3381–3465 (2014).
Henstridge, M. C., Laborda, E., Rees, N. V. & Compton, R. G. Marcus–Hush–Chidsey theory of electron transfer applied to voltammetry: a review. Electrochim. Acta 84, 12–20 (2012).
Soudackov, A. & Hammes-Schiffer, S. Derivation of rate expressions for nonadiabatic proton-coupled electron transfer reactions in solution. J. Phys. Chem. 113, 2385–2396 (2000).
Mayer, J. M. & Rhile, I. J. Thermodynamics and kinetics of proton-coupled electron transfer: stepwise vs. concerted pathways. Biochim. Biophys. Acta 1655, 51–58 (2004).
Mayer, J. M. Proton-coupled electron transfer: a reaction chemist’s view. Annu. Rev. Phys. Chem. 55, 363–390 (2004).
Xie, Y. & Anson, F. C. Analysis of the cyclic voltammetric responses exhibited by electrodes modified with monolayers of catalysts in the absence and presence of substrates. J. Electroanal. Chem. 384, 145–153 (1995).
Li, Y., Tse, E. C. M., Barile, C. J., Gewirth, A. A. & Zimmerman, S. C. Photoresponsive molecular switch for regulating transmembrane proton-transfer kinetics. J. Am. Chem. Soc. 137, 14059–14062 (2015).
Bard, A. J. & Faulkner, L. R. Electrochemical Methods: Fundamentals and Applications (Wiley, 2000).
Chidsey, C. E. D. Free energy and temperature dependence of electron transfer at the metal-electrolyte interface. Science 251, 919–922 (1991).
Collman, J. P., Devaraj, N. K., Eberspacher, T. P. A. & Chidsey, C. E. D. Mixed azide-terminated monolayers: a platform for modifying electrode surfaces. Langmuir 22, 2457–2464 (2006).
Collman, J. P. et al. A cytochrome c oxidase model catalyzes oxygen to water reduction under rate-limiting electron flux. Science 315, 1565–1568 (2007).
Collman, J. P., Hosseini, A., Eberspacher, T. A. & Chidsey, C. E. D. Selective anodic desorption for assembly of different thiol monolayers on the individual electrodes of an array. Langmuir 25, 6517–6521 (2009).
Collman, J. P. et al. Role of a distal pocket in the catalytic O2 reduction by cytochrome c oxidase models immobilized on interdigitated array electrodes. Proc. Natl Acad. Sci. USA 106, 7320–7323 (2009).
Irebo, T., Reece, S. Y., Sjödin, M., Nocera, D. G. & Hammarström, L. Proton-coupled electron transfer of tyrosine oxidation: buffer dependence and parallel mechanisms. J. Am. Chem. Soc. 129, 15462–15464 (2007).
Fecenko, C. J., Meyer, T. J. & Thorp, H. H. Electrocatalytic oxidation of tyrosine by parallel rate-limiting proton transfer and multisite electron-proton transfer. J. Am. Chem. Soc. 128, 11020–11021 (2006).
Chen, Z., Vannucci, A. K., Concepcion, J. J., Jurss, J. W. & Meyer, T. J. Proton-coupled electron transfer at modified electrodes by multiple pathways. Proc. Natl Acad. Sci. USA 108, E1461–E1469 (2011).
Wenger, O. S. Proton-coupled electron transfer with photoexcited metal complexes. Acc. Chem. Res. 46, 1517–1526 (2013).
Rosenthal, J. & Nocera, D. G. Role of proton-coupled electron transfer in O–O bond activation. Acc. Chem. Res. 40, 543–553 (2007).
Sjödin, M. et al. Switching the redox mechanism: models for proton-coupled electron transfer from tyrosine and tryptophan. J. Am. Chem. Soc. 127, 3855–3863 (2005).
Hatcher, L. & Karlin, K. Oxidant types in copper–dioxygen chemistry: the ligand coordination defines the Cun-O2 structure and subsequent reactivity. J. Biol. Inorg. Chem. 9, 669–683 (2004).
Thorseth, M. A., Letko, C. S., Tse, E. C. M., Rauchfuss, T. B. & Gewirth, A. A. Ligand effects on the overpotential for dioxygen reduction by tris(2-pyridylmethyl)amine derivatives. Inorg. Chem. 52, 628–634 (2012).
McCrory, C. C. L., Ottenwaelder, X., Stack, T. D. P. & Chidsey, C. E. D. Kinetic and mechanistic studies of the electrocatalytic reduction of O2 to H2O with mononuclear Cu complexes of substituted 1,10-phenanthrolines. J. Phys. Chem. A 111, 12641–12650 (2007).
Thorseth, M. A., Tornow, C. E., Tse, E. C. M. & Gewirth, A. A. Cu complexes that catalyze the oxygen reduction reaction. Coord. Chem. Rev. 257, 130–139 (2013).
Barile, C. J. et al. Proton switch for modulating oxygen reduction by a copper electrocatalyst embedded in a hybrid bilayer membrane. Nature Mater. 13, 619–623 (2014).
Hosseini, A. et al. Hybrid bilayer membrane: a platform to study the role of proton flux on the efficiency of oxygen reduction by a molecular electrocatalyst. J. Am. Chem. Soc. 133, 11100–11102 (2011).
Hosseini, A. et al. Ferrocene embedded in an electrode-supported hybrid lipid bilayer membrane: a model system for electrocatalysis in a biomimetic environment. Langmuir 26, 17674–17678 (2010).
Tse, E. C. M. et al. Anion transport through lipids in a hybrid bilayer membrane. Anal. Chem. 87, 2403–2409 (2015).
Gewirth, A. A. & Thorum, M. S. Electroreduction of dioxygen for fuel-cell applications: materials and challenges. Inorg. Chem. 49, 3557–3566 (2010).
Carley, A. N. & Kleinfeld, A. M. Flip-flop is the rate-limiting step for transport of free fatty acids across lipid vesicle membranes. Biochemistry 48, 10437–10445 (2009).
Rose, M. C. & Steuhr, J. Kinetics of proton transfer reactions in aqueous solution. III. Rates of internally hydrogen-bonded systems. J. Am. Chem. Soc. 90, 7205–7209 (1968).
Wu, G., More, K. L., Johnston, C. M. & Zelenay, P. High-performance electrocatalysts for oxygen reduction derived from polyaniline, iron, and cobalt. Science 332, 443–447 (2011).
Li, M. F., Liao, L. W., Yuan, D. F., Mei, D. & Chen, Y.-X. pH effect on oxygen reduction reaction at Pt(111) electrode. Electrochim. Acta 110, 780–789 (2013).
Ghoneim, M. M., Clouser, S. & Yeager, E. Oxygen reduction kinetics in deuterated phosphoric acid. J. Electrochem. Soc. 132, 1160–1162 (1985).
Higgins, D. C. & Chen, Z. Recent progress in non-precious metal catalysts for PEM fuel cell applications. Can. J. Chem. Eng. 91, 1881–1895 (2013).
Nogala, W. et al. Scanning electrochemical microscopy activity mapping of electrodes modified with laccase encapsulated in sol–gel processed matrix. Bioelectrochemistry 79, 101–107 (2010).
Proshlyakov, D. A. et al. Oxygen activation and reduction in respiration: involvement of redox-active tyrosine 244. Science 290, 1588–1591 (2000).
Brändén, G., Gennis, R. B. & Brzezinski, P. Transmembrane proton translocation by cytochrome c oxidase. Biochim. Biophys. Acta 1757, 1052–1063 (2006).
Bento, I. et al. Mechanisms underlying dioxygen reduction in laccases. Structural and modelling studies focusing on proton transfer. BMC Struct. Biol. 10, 28 (2010).
Albrecht, O., Gruler, H. & Sackmann, E. Polymorphism of phospholipid monolayers. J. Phys. France 39, 301–313 (1978).
Gong, K., Du, F., Xia, Z., Durstock, M. & Dai, L. Nitrogen-doped carbon nanotube arrays with high electrocatalytic activity for oxygen reduction. Science 323, 760–764 (2009).
Campillo-Brocal, J. C., Lucas-Elio, P. & Sanchez-Amat, A. Identification in Marinomonas mediterranea of a novel quinoprotein with glycine oxidase activity. MicrobiologyOpen 2, 684–694 (2013).
Acknowledgements
E.C.M.T. acknowledges a Croucher Foundation Scholarship. C.J.B. acknowledges a National Science Foundation Graduate Research Fellowship (NSF DGE-1144245) and a Springborn Fellowship. We thank the US Department of Energy (DE-FG02-95ER46260) for support of this research. This work was carried out in part in the Frederick Seitz Materials Research Laboratory Central Facilities, which are partially supported by the US Department of Energy (DE-FG02-07ER46453 and DE-FG02-07ER46471). We acknowledge J. Varnell for providing PANI-Fe-C, E. Barile for help with artwork, and R. Gennis, T. Rauchfuss, K. Suslick and R. Nuzzo for insightful discussions.
Author information
Authors and Affiliations
Contributions
E.C.M.T., C.J.B., S.C.Z., A.H. and A.A.G. designed the experiments. E.C.M.T., C.J.B. and N.A.K. performed the experiments. Y.L. synthesized BTT. E.C.M.T., C.J.B., Y.L., J.P.G., S.C.Z., A.H. and A.A.G. wrote the paper. E.C.M.T., C.J.B., N.A.K., J.P.G., A.H. and A.A.G. analysed the data. All authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 4599 kb)
Rights and permissions
About this article
Cite this article
Tse, E., Barile, C., Kirchschlager, N. et al. Proton transfer dynamics control the mechanism of O2 reduction by a non-precious metal electrocatalyst. Nature Mater 15, 754–759 (2016). https://doi.org/10.1038/nmat4636
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmat4636
This article is cited by
-
Overcoming the Electrode Challenges of High-Temperature Proton Exchange Membrane Fuel Cells
Electrochemical Energy Reviews (2023)
-
Hybrid bilayer membranes as platforms for biomimicry and catalysis
Nature Reviews Chemistry (2022)
-
Promoting biomass electrooxidation via modulating proton and oxygen anion deintercalation in hydroxide
Nature Communications (2022)
-
Silica-facilitated proton transfer for high-temperature proton-exchange membrane fuel cells
Science China Chemistry (2021)
-
Lattice-strained metal–organic-framework arrays for bifunctional oxygen electrocatalysis
Nature Energy (2019)