Abstract
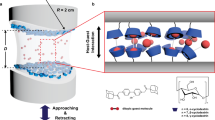
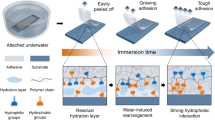
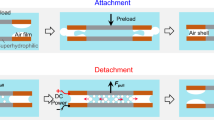
Polyelectrolyte complexation is critical to the formation and properties of many biological and polymeric materials, and is typically initiated by aqueous mixing1 followed by fluid–fluid phase separation, such as coacervation2,3,4,5. Yet little to nothing is known about how coacervates evolve into intricate solid microarchitectures. Inspired by the chemical features of the cement proteins of the sandcastle worm, here we report a versatile and strong wet-contact microporous adhesive resulting from polyelectrolyte complexation triggered by solvent exchange. After premixing a catechol-functionalized weak polyanion with a polycation in dimethyl sulphoxide (DMSO), the solution was applied underwater to various substrates whereupon electrostatic complexation, phase inversion, and rapid setting were simultaneously actuated by water–DMSO solvent exchange. Spatial and temporal coordination of complexation, inversion and setting fostered rapid (∼25 s) and robust underwater contact adhesion (Wad ≥ 2 J m−2) of complexed catecholic polyelectrolytes to all tested surfaces including plastics, glasses, metals and biological materials.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Faul, C. F. J. & Antonietti, M. Ionic self-assembly: facile synthesis of supramolecular materials. Adv. Mater. 15, 673–683 (2003).
Perry, S. L. et al. Chirality-selected phase behaviour in ionic polypeptide complexes. Nature Commun. 6, 6052 (2015).
Tang, T. Y. D. et al. Fatty acid membrane assembly on coacervate microdroplets as a step towards a hybrid protocell model. Nature Chem. 6, 527–533 (2014).
Sokolova, E. et al. Enhanced transcription rates in membrane-free protocells formed by coacervation of cell lysate. Proc. Natl Acad. Sci. USA 110, 11692–11697 (2013).
Xu, L. M. et al. Self-assembly of ultralong polyion nanoladders facilitated by ionic recognition and molecular stiffness. J. Am. Chem. Soc. 136, 1942–1947 (2014).
Lee, B. P., Messersmith, P. B., Israelachvili, J. N. & Waite, J. H. Mussel-inspired adhesives and coatings. Annu. Rev. Mater. Res. 41, 99–132 (2011).
Stewart, R. J., Ransom, T. C. & Hlady, V. Natural underwater adhesives. J. Polym. Sci. B 49, 757–771 (2011).
Sedo, J., Saiz-Poseu, J., Busque, F. & Ruiz-Molina, D. Catechol-based biomimetic functional materials. Adv. Mater. 25, 653–701 (2013).
Zhao, H., Sun, C. J., Stewart, R. J. & Waite, J. H. Cement proteins of the tube-building polychaete Phragmatopoma californica. J. Biol. Chem. 280, 42938–42944 (2005).
Lee, H., Lee, B. P. & Messersmith, P. B. A reversible wet/dry adhesive inspired by mussels and geckos. Nature 448, 338–342 (2007).
Shao, H. & Stewart, R. J. Biomimetic underwater adhesives with environmentally triggered setting mechanisms. Adv. Mater. 22, 729–733 (2010).
Stewart, R. J., Weaver, J. C., Morse, D. E. & Waite, J. H. The tube cement of Phragmatopoma californica: a solid foam. J. Exp. Biol. 207, 4727–4734 (2004).
Stevens, M. J., Steren, R. E., Hlady, V. & Stewart, R. J. Multiscale structure of the underwater adhesive of Phragmatopoma californica: a nanostructured latex with a steep microporosity gradient. Langmuir 23, 5045–5049 (2007).
Tamarin, A., Lewis, P. & Askey, J. Structure and formation of byssus attachment plaque in mytilus. J. Morphol. 149, 199–221 (1976).
Desmond, K., Zacchia, N. A., Waite, J. H. & Valentine, M. Dynamics of mussel plaque detachment. Soft Matter 11, 6832–6839 (2015).
Bungenberg de Jong, H. G. in Colloid Science Vol. II (ed. Kruyt, H. R.) 431–482 (Elsevier, 1949).
Hwang, D. S. et al. Viscosity and interfacial properties in a mussel-inspired adhesive coacervate. Soft Matter 6, 3232–3236 (2010).
Stewart, R. J., Wang, C. S. & Shao, H. Complex coacervates as a foundation for synthetic underwater adhesives. Adv. Colloid Interface Sci. 167, 85–93 (2011).
Rose, S. et al. Nanoparticle solutions as adhesives for gels and biological tissues. Nature 505, 382–385 (2014).
Ono, T., Sugimoto, T., Shinkai, S. & Sada, K. Lipophilic polyelectrolyte gels as super-absorbent polymers for nonpolar organic solvents. Nature Mater. 6, 429–433 (2007).
Yuan, J. Y., Mecerreyes, D. & Antonietti, M. Poly(ionic liquid)s: an update. Prog. Polym. Sci. 38, 1009–1036 (2013).
Poulsen, N. et al. Isolation and biochemical characterization of underwater adhesives from diatoms. Biofouling 30, 513–523 (2014).
Gibson, L. J. & Ashby, M. F. The mechanics of 3-dimensional cellular materials. Proc. R. Soc. Lond. A 382, 43–59 (1982).
Meyers, M. A., McKittrick, J. & Chen, P. Y. Structural biological materials: critical mechanics-materials connections. Science 339, 773–779 (2013).
Wu, D. C. et al. Design and preparation of porous polymers. Chem. Rev. 112, 3959–4015 (2012).
Johnson, K. L., Kendall, K. & Roberts, A. D. Surface energy and contact of elastic solids. Proc. R. Soc. Lond. A 324, 301–313 (1971).
Raviv, U. et al. Lubrication by charged polymers. Nature 425, 163–165 (2003).
Raviv, U. & Klein, J. Fluidity of bound hydration layers. Science 297, 1540–1543 (2002).
Ludema, K. C. & Tabor, D. The friction and visco-elastic properties of polymeric solids. Wear 9, 329–348 (1966).
Israelachvili, J. N. Intermolecular and Surface Forces 3rd edn, 494 (Academic, 2011).
Ahn, B. K., Lee, D. W., Israelachvili, J. N. & Waite, J. H. Surface-initiated self-healing of polymers in aqueous media. Nature Mater. 13, 867–872 (2014).
Acknowledgements
The authors gratefully acknowledge financial support from the National Science Foundation (NSF) through the MRSEC Program DMR-1121053 (MRL-UCSB), which also supported the MRL Central Facilities (a member of the NSF-funded Materials Research Facilities Network (www.mrfn.org)). J.H.W. and B.K.A. acknowledge support from the Office of Naval Research N000141310867. J.H.W. and J.N.I. also acknowledge support from the US National Institutes of Health (R01 DE018468). Authors thank R. Mirshafian for help with optical microscope and W. Wei for discussions.
Author information
Authors and Affiliations
Contributions
Q.Z. and J.H.W. conceived the concept of materials processing. Q.Z. was responsible for the experimental part with input from B.K.A. who advised on PAAcat synthesis, SFA, confocal microscopy and FTIR. S.S. synthesized PAAcat copolymers. D.W.L. planned and performed SFA experiments and analysed the data. D.W.L. and B.K.A. performed and analysed confocal microscope experiments. Q.Z., D.W.L., B.K.A. and J.H.W. analysed data and wrote the paper. All authors read and commented on the paper. J.H.W. supervised the overall project.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 4999 kb)
Supplementary Movie 1
Supplementary Movie 1 (WMV 8252 kb)
Supplementary Movie 2
Supplementary Movie 2 (WMV 1857 kb)
Rights and permissions
About this article
Cite this article
Zhao, Q., Lee, D., Ahn, B. et al. Underwater contact adhesion and microarchitecture in polyelectrolyte complexes actuated by solvent exchange. Nature Mater 15, 407–412 (2016). https://doi.org/10.1038/nmat4539
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmat4539
This article is cited by
-
Programmable adhesion and morphing of protein hydrogels for underwater robots
Nature Communications (2024)
-
Synthesis of robust underwater glues from common proteins via unfolding-aggregating strategy
Nature Communications (2023)
-
Ultrafast seawater desalination with covalent organic framework membranes
Nature Sustainability (2022)
-
Spontaneous water-on-water spreading of polyelectrolyte membranes inspired by skin formation
Nature Communications (2022)
-
Ultrastrong underwater adhesion on diverse substrates using non-canonical phenolic groups
Nature Communications (2022)