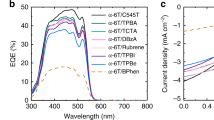
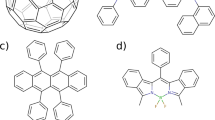
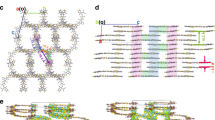
Abstract
Structural order in organic solar cells is paramount: it reduces energetic disorder, boosts charge and exciton mobilities, and assists exciton splitting. Owing to spatial localization of electronic states, microscopic descriptions of photovoltaic processes tend to overlook the influence of structural features at the mesoscale. Long-range electrostatic interactions nevertheless probe this ordering, making local properties depend on the mesoscopic order. Using a technique developed to address spatially aperiodic excitations in thin films and in bulk, we show how inclusion of mesoscale order resolves the controversy between experimental and theoretical results for the energy-level profile and alignment in a variety of photovoltaic systems, with direct experimental validation. Optimal use of long-range ordering also rationalizes the acceptor–donor–acceptor paradigm for molecular design of donor dyes. We predict open-circuit voltages of planar heterojunction solar cells in excellent agreement with experimental data, based only on crystal structures and interfacial orientation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Brédas, J-L., Norton, J. E., Cornil, J. & Coropceanu, V. Molecular understanding of organic solar cells: The challenges. Acc. Chem. Res. 42, 1691–1699 (2009).
Clarke, T. M. & Durrant, J. R. Charge photogeneration in organic solar cells. Chem. Rev. 110, 6736–6767 (2010).
Dou, L. et al. 25th anniversary article: A decade of organic/polymeric photovoltaic research. Adv. Mater. 25, 6642–6671 (2013).
Boudreault, P-L. T., Najari, A. & Leclerc, M. Processable low-bandgap polymers for photovoltaic applications. Chem. Mater. 23, 456–469 (2010).
Sommer, M., Hüttner, S. & Thelakkat, M. Donor–acceptor block copolymers for photovoltaic applications. J. Mater. Chem. 20, 10788–10797 (2010).
Mishra, A. & Bäuerle, P. Small molecule organic semiconductors on the move: Promises for future solar energy technology. Angew. Chem. Int. Ed. 51, 2020–2067 (2012).
Liu, Y. et al. Solution-processed small-molecule solar cells: Breaking the 10% power conversion efficiency. Sci. Rep. 3, 3356 (2013).
Meerheim, R., Körner, C. & Leo, K. Highly efficient organic multi-junction solar cells with a thiophene based donor material. Appl. Phys. Lett. 105, 063306 (2014).
Wilke, A. et al. Electric fields induced by energy level pinning at organic heterojunctions. Appl. Phys. Lett. 98, 123304 (2011).
Beljonne, D. et al. Electronic processes at organic–organic interfaces: Insight from modeling and implications for opto-electronic devices. Chem. Mater. 23, 591–609 (2011).
McMahon, D. P., Cheung, D. L. & Troisi, A. Why holes and electrons separate so well in polymer/fullerene photovoltaic cells. J. Phys. Chem. Lett. 2, 2737–2741 (2011).
Liu, A. et al. Control of electric field strength and orientation at the donor–acceptor interface in organic solar cells. Adv. Mater. 20, 1065–1070 (2008).
Gregg, B. A. Entropy of charge separation in organic photovoltaic cells: The benefit of higher dimensionality. J. Phys. Chem. Lett. 2, 3013–3015 (2011).
Jailaubekov, A. E. et al. Hot charge-transfer excitons set the time limit for charge separation at donor/acceptor interfaces in organic photovoltaics. Nature Mater. 12, 66–73 (2013).
Caruso, D. & Troisi, A. Long-range exciton dissociation in organic solar cells. Proc. Natl Acad. Sci. USA 109, 13498–13502 (2012).
Yost, S. R., Wang, L-P. & Van Voorhis, T. Molecular insight into the energy levels at the organic donor/acceptor interface: A quantum mechanics/molecular mechanics study. J. Phys. Chem. C 115, 14431–14436 (2011).
Rühle, V. et al. Microscopic simulations of charge transport in disordered organic semiconductors. J. Chem. Theory Comput. 7, 3335–3345 (2011).
Fu, Y-T., Risko, C. & Brédas, J-L. Intermixing at the pentacene-fullerene bilayer interface: A molecular dynamics study. Adv. Mater. 25, 878–882 (2013).
Muccioli, L. et al. Supramolecular organization of functional organic materials in the bulk and at organic/organic interfaces: A modeling and computer simulation approach. Top. Curr. Chem. 352, 39–101 (2014).
Mothy, S. et al. Tuning the interfacial electronic structure at organic heterojunctions by chemical design. J. Phys. Chem. Lett. 3, 2374–2378 (2012).
Verlaak, S. et al. Electronic structure and geminate pair energetics at organic–organic interfaces: The case of pentacene/C60 heterojunctions. Adv. Funct. Mater. 19, 3809–3814 (2009).
Linares, M. et al. On the interface dipole at the pentacene-fullerene heterojunction: A theoretical study. J. Phys. Chem. C 114, 3215–3224 (2010).
May, F., Baumeier, B., Lennartz, C. & Andrienko, D. Can lattice models predict the density of states of amorphous organic semiconductors? Phys. Rev. Lett. 109, 136401 (2012).
Idé, J. et al. Interfacial dipole and band bending in model pentacene/C60 heterojunctions. Int. J. Quantum Chem. 113, 580–584 (2013).
Yost, S. R. & Van Voorhis, T. Electrostatic effects at organic semiconductor interfaces: A mechanism for “cold” exciton breakup. J. Phys. Chem. C 117, 5617–5625 (2013).
Idé, J. et al. Charge dissociation at interfaces between discotic liquid crystals: The surprising role of column mismatch. J. Am. Chem. Soc. 136, 2911–2920 (2014).
Stone, A. J. The Theory of Intermolecular Forces (Clarendon Press, 1997).
De Leeuw, S. W., Perram, J. W. & Smith, E. R. Simulation of electrostatic systems in periodic boundary conditions. i. lattice sums and dielectric constants. Proc. R. Soc. Lond. A 373, 27–56 (1980).
Chen, W., Qi, D-C., Huang, H., Gao, X. & Wee, A. T. S. Organic–organic heterojunction interfaces: Effect of molecular orientation. Adv. Funct. Mater. 21, 410–424 (2011).
Heimel, G., Salzmann, I., Duhm, S. & Koch, N. Design of organic semiconductors from molecular electrostatics. Chem. Mater. 23, 359–377 (2011).
Duhm, S. et al. Orientation-dependent ionization energies and interface dipoles in ordered molecular assemblies. Nature Mater. 7, 326–332 (2008).
Garcia-Belmonte, G. & Bisquert, J. Open-circuit voltage limit caused by recombination through tail states in bulk heterojunction polymer-fullerene solar cells. Appl. Phys. Lett. 96, 113301 (2010).
Schrader, M. et al. Comparative study of microscopic charge dynamics in crystalline acceptor-substituted oligothiophenes. J. Am. Chem. Soc. 134, 6052–6056 (2012).
Wilke, A. et al. Correlation between interface energetics and open circuit voltage in organic photovoltaic cells. Appl. Phys. Lett. 101, 233301 (2012).
Widmer, J., Tietze, M., Leo, K. & Riede, M. Open-circuit voltage and effective gap of organic solar cells. Adv. Funct. Mater. 23, 5814–5821 (2013).
Koster, L. J. A., Mihailetchi, V. D., Ramaker, R. & Blom, P. W. M. Light intensity dependence of open-circuit voltage of polymer:Fullerene solar cells. Appl. Phys. Lett. 86, 123509 (2005).
Potscavage, W. J., Sharma, A. & Kippelen, B. Critical interfaces in organic solar cells and their influence on the open-circuit voltage. Acc. Chem. Res. 42, 1758–1767 (2009).
Vandewal, K., Tvingstedt, K., Gadisa, A., Inganäs, O. & Manca, J. V. On the origin of the open-circuit voltage of polymer–fullerene solar cells. Nature Mater. 8, 904–909 (2009).
Maurano, A. et al. Recombination dynamics as a key determinant of open circuit voltage in organic bulk heterojunction solar cells: A comparison of four different donor polymers. Adv. Mater. 22, 4987–4992 (2010).
Wynands, D. et al. Spectroscopic ellipsometry characterization of vacuum-deposited organic films for the application in organic solar cells. Org. Electron. 13, 885–893 (2012).
Pfeiffer, M. et al. Organic photoactive device. US patent 8426727 (2013).
Stone, A. J. Distributed multipole analysis: Stability for large basis sets. J. Chem. Theory Comput. 1, 1128–1132 (2005).
Thole, B. Molecular polarizabilities calculated with a modified dipole interaction. Chem. Phys. 59, 341–350 (1981).
Van Duijnen, P. T. & Swart, M. Molecular and atomic polarizabilities: Thole’s model revisited. J. Phys. Chem. A 102, 2399–2407 (1998).
Fitzner, R. et al. Correlation of π-conjugated oligomer structure with film morphology and organic solar cell performance. J. Am. Chem. Soc. 134, 11064–11067 (2012).
Bürckstümmer, H. et al. Efficient solution-processed bulk heterojunction solar cells by antiparallel supramolecular arrangement of dipolar donor–acceptor dyes. Angew. Chem. Int. Ed. 50, 11628–11632 (2011).
Zhang, F. et al. Energy level alignment and morphology of interfaces between molecular and polymeric organic semiconductors. Org. Electron. 8, 606–614 (2007).
Heimel, G. & Koch, N. in Interface Controlled Organic Thin Films Vol. 129 (eds Al-Shamery, K., Horowitz, G., Sitter, H. & Rubahn, H-G.) 141–145 (Springer, 2009).
Tietze, M. L. et al. Correlation of open-circuit voltage and energy levels in zinc-phthalocyanine: C60 bulk heterojunction solar cells with varied mixing ratio. Phys. Rev. B 88, 085119 (2013).
Kinoshita, Y., Hasobe, T. & Murata, H. Controlling open-circuit voltage of organic photovoltaic cells by inserting thin layer of Zn–phthalocyanine at pentacene/C60 interface. Jpn. J. Appl. Phys. 47, 1234–1237 (2008).
Acknowledgements
This work has been supported by the BMBF programme MEDOS (FKZ 03EK3503B). We are grateful to T. Bereau for critical reading of the manuscript and P. Bäuerle and C. Körner for fruitful collaborations.
Author information
Authors and Affiliations
Contributions
C.P., B.B. and D.A. conceived and performed simulations. M.T., C.E. and K.L. contributed experimental results on DCVnTs. S.O., D.H., F.W. and K.M. contributed experimental results on merocyanines. C.P. and D.A. wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 277 kb)
Rights and permissions
About this article
Cite this article
Poelking, C., Tietze, M., Elschner, C. et al. Impact of mesoscale order on open-circuit voltage in organic solar cells. Nature Mater 14, 434–439 (2015). https://doi.org/10.1038/nmat4167
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmat4167
This article is cited by
-
Molecular orientation-dependent energetic shifts in solution-processed non-fullerene acceptors and their impact on organic photovoltaic performance
Nature Communications (2023)
-
Models and mechanisms of ternary organic solar cells
Nature Reviews Materials (2023)
-
Reduced bimolecular charge recombination in efficient organic solar cells comprising non-fullerene acceptors
Scientific Reports (2023)
-
Controlling doping efficiency in organic semiconductors by tuning short-range overscreening
Nature Communications (2023)
-
Open-circuit voltage of organic solar cells: interfacial roughness makes the difference
Communications Physics (2022)