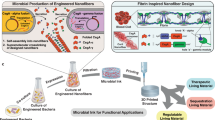
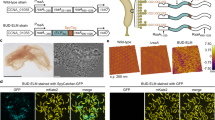
Abstract
Many natural biological systems—such as biofilms, shells and skeletal tissues—are able to assemble multifunctional and environmentally responsive multiscale assemblies of living and non-living components. Here, by using inducible genetic circuits and cellular communication circuits to regulate Escherichia coli curli amyloid production, we show that E. coli cells can organize self-assembling amyloid fibrils across multiple length scales, producing amyloid-based materials that are either externally controllable or undergo autonomous patterning. We also interfaced curli fibrils with inorganic materials, such as gold nanoparticles (AuNPs) and quantum dots (QDs), and used these capabilities to create an environmentally responsive biofilm-based electrical switch, produce gold nanowires and nanorods, co-localize AuNPs with CdTe/CdS QDs to modulate QD fluorescence lifetimes, and nucleate the formation of fluorescent ZnS QDs. This work lays a foundation for synthesizing, patterning, and controlling functional composite materials with engineered cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Fratzl, P. & Weinkamer, R. Nature’s hierarchical materials. Prog. Mater. Sci. 52, 1263–1334 (2007).
Kollmannsberger, P., Bidan, C. M., Dunlop, J. W. C. & Fratzl, P. The physics of tissue patterning and extracellular matrix organisation: How cells join forces. Soft Matter 7, 9549–9560 (2011).
Stevens, M. M. & George, J. H. Exploring and engineering the cell surface interface. Science 310, 1135–1138 (2005).
O’Toole, G., Kaplan, H. B. & Kolter, R. Biofilm formation as microbial development. Annu. Rev. Microbiol. 54, 49–79 (2000).
Epstein, A. K., Pokroy, B., Seminara, A. & Aizenberg, J. Bacterial biofilm shows persistent resistance to liquid wetting and gas penetration. Proc. Natl Acad. Sci. USA 108, 995–1000 (2011).
Belcher, A. M. et al. Control of crystal phase switching and orientation by soluble mollusc-shell proteins. Nature 381, 56–58 (1996).
Su, X. W., Zhang, D. M. & Heuer, A. H. Tissue regeneration in the shell of the Giant Queen Conch, Strombus gigas. Chem. Mater. 16, 581–593 (2004).
Aizenberg, J. et al. Skeleton of Euplectella sp: structural hierarchy from the nanoscale to the macroscale. Science 309, 275–278 (2005).
Weiner, S. & Wagner, H. D. The material bone: Structure mechanical function relations. Annu. Rev. Mater. Sci. 28, 271–298 (1998).
Brenner, K. & Arnold, F. H. Self-organization, layered structure, and aggregation enhance persistence of a synthetic biofilm consortium. PloS ONE 6, e16791 (2011).
Brenner, K., Karig, D. K., Weiss, R. & Arnold, F. H. Engineered bidirectional communication mediates a consensus in a microbial biofilm consortium. Proc. Natl Acad. Sci. USA 104, 17300–17304 (2007).
Hong, S. H. et al. Synthetic quorum-sensing circuit to control consortial biofilm formation and dispersal in a microfluidic device. Nature Commun. 3, 613 (2012).
Ma, Q., Yang, Z., Pu, M., Peti, W. & Wood, T. K. Engineering a novel c-di-GMP-binding protein for biofilm dispersal. Environ. Microbiol. 13, 631–642 (2011).
Lee, J., Jayaraman, A. & Wood, T. K. Indole is an inter-species biofilm signal mediated by SdiA. BMC Microbiol. 7, 42 (2007).
Payne, S. et al. Temporal control of self-organized pattern formation without morphogen gradients in bacteria. Mol. Syst. Biol. 9, 697 (2013).
Payne, S. & You, L. Engineered cell–cell communication and its applications. Adv. Biochem. Eng./Biotechnol. http://dx.doi.org/10.1007/10_2013_249 (2013).
Barnhart, M. M. & Chapman, M. R. Curli biogenesis and function. Annu. Rev. Microbiol. 60, 131–147 (2006).
Callura, J. M., Cantor, C. R. & Collins, J. J. Genetic switchboard for synthetic biology applications. Proc. Natl Acad. Sci. USA 109, 5850–5855 ( 2012).
Prigent-Combaret, C. et al. Developmental pathway for biofilm formation in curli-producing Escherichia coli strains: Role of flagella, curli and colanic acid. Environ. Microbiol. 2, 450–464 (2000).
Vidal, O. et al. Isolation of an Escherichia coli K-12 mutant strain able to form biofilms on inert surfaces: Involvement of a new ompR allele that increases curli expression. J. Bacteriology 180, 2442–2449 (1998).
Hung, C. et al. Escherichia coli biofilms have an organized and complex extracellular matrix structure. mBio 4, e00645–e00613 (2013).
Wang, X., Hammer, N. D. & Chapman, M. R. The molecular basis of functional bacterial amyloid polymerization and nucleation. J. Biol. Chem. 283, 21530–21539 (2008).
Basu, S., Gerchman, Y., Collins, C. H., Arnold, F. H. & Weiss, R. A synthetic multicellular system for programmed pattern formation. Nature 434, 1130–1134 (2005).
Bacchus, W et al. Synthetic two-way communication between mammalian cells. Nature Biotechnol. 30, 991–996 (2012).
Tabor, J. J. et al. A synthetic genetic edge detection program. Cell 137, 1272–1281 (2009).
Liu, C. et al. Sequential establishment of stripe patterns in an expanding cell population. Science 334, 238–241 (2011).
Jang, B., Park, J. Y., Tung, C. H., Kim, I. H. & Choi, Y. Gold nanorod-photosensitizer complex for near-infrared fluorescence imaging and photodynamic/photothermal therapy in vivo. ACS Nano 5, 1086–1094 (2011).
Dreaden, E. C. et al. Small molecule-gold nanorod conjugates selectively target and induce macrophage cytotoxicity towards breast cancer cells. Small 8, 2819–2822 (2012).
Libutti, S. K. et al. Phase I and pharmacokinetic studies of CYT-6091, a novel PEGylated colloidal gold-rhTNF nanomedicine. Clinical Cancer Research: An Official J. Am. Assoc. Can. Res. 16, 6139–6149 (2010).
Zakeri, B. et al. Peptide tag forming a rapid covalent bond to a protein, through engineering a bacterial adhesin. Proc. Natl Acad. Sci. USA 109, E690–E697 (2012).
Polman, A. & Atwater, H. A. Photonic design principles for ultrahigh-efficiency photovoltaics. Nature Mater. 11, 174–177 (2012).
Reineck, P. et al. A solid-state plasmonic solar cell via metal nanoparticle self-assembly. Adv. Mater. 24, 4750–4755 (2012).
Curto, A. G. et al. Unidirectional emission of a quantum dot coupled to a nanoantenna. Science 329, 930–933 (2010).
Yuan, Z. L. et al. Electrically driven single-photon source. Science 295, 102–105 (2002).
Mao, C. et al. Viral assembly of oriented quantum dot nanowires. Proc. Natl Acad. Sci. USA 100, 6946–6951 (2003).
Zhang, S. G. Fabrication of novel biomaterials through molecular self-assembly. Nature Biotechnol. 21, 1171–1178 (2003).
King, N. P. et al. Computational design of self-assembling protein nanomaterials with atomic level accuracy. Science 336, 1171–1174 (2012)10.1126/science.1219364.
Mart, R. J., Osborne, R. D., Stevens, M. M. & Ulijn, R. V. Peptide-based stimuli-responsive biomaterials. Soft Matter 2, 822–835 (2006).
Webber, M. J. et al. Supramolecular nanostructures that mimic VEGF as a strategy for ischemic tissue repair. Proc. Natl Acad. Sci. USA 108, 13438–13443 (2011).
So, C. R., Tamerler, C. & Sarikaya, M. Adsorption, diffusion, and self-assembly of an engineered gold-binding peptide on Au(111) investigated by atomic force microscopy. Angew. Chem. Int. Ed. 48, 5174–5177 (2009).
Channon, K. J., Devlin, G. L. & MacPhee, C. E. Efficient energy transfer within self-assembling peptide fibers: A route to light-harvesting nanomaterials. J. Am. Chem. Soc. 131, 12520–12521 (2009).
Scheibel, T. et al. Conducting nanowires built by controlled self-assembly of amyloid fibers and selective metal deposition. Proc. Natl Acad. Sci. USA 100, 4527–4532 (2003).
Smith, J. F., Knowles, T. P., Dobson, C. M., Macphee, C. E. & Welland, M. E. Characterization of the nanoscale properties of individual amyloid fibrils. Proc. Natl Acad. Sci. USA 103, 15806–15811 (2006).
Felgner, P. L. et al. Lipofection: A highly efficient, lipid-mediated DNA-transfection procedure. Proc. Natl Acad. Sci. USA 84, 7413–7417 (1987).
Winfree, E., Liu,, F., Wenzler, L. A. & Seeman, N. C. Design and self-assembly of two-dimensional DNA crystals. Nature 394, 539–544 (1998).
Rothemund, P. W. Folding DNA to create nanoscale shapes and patterns. Nature 440, 297–302 (2006).
Tian, B. et al. Macroporous nanowire nanoelectronic scaffolds for synthetic tissues. Nature Mater. 11, 986–994 (2012).
Hwang, S. W. et al. A physically transient form of silicon electronics. Science 337, 1640–1644 (2012).
Amsden, J. J. et al. Rapid nanoimprinting of silk fibroin films for biophotonic applications. Adv. Mater. 22, 1746–1749 (2010).
Lutolf, M. P. & Hubbell, J. A. Synthetic biomaterials as instructive extracellular microenvironments for morphogenesis in tissue engineering. Nature Biotechnol. 23, 47–55 (2005).
Prewitz, M. C. et al. Tightly anchored tissue-mimetic matrices as instructive stem cell microenvironments. Nature Methods 10, 788–794 (2013).
Chiu, W. K. & Yu, K. M. Direct digital manufacturing of three-dimensional functionally graded material objects. Computer-Aided Design 40, 1080–1093 (2008).
Xia, Y., Rogers, J. A., Paul, K. E. & Whitesides, G. M. Unconventional methods for fabricating and patterning nanostructures. Chem. Rev. 99, 1823–1848 (1999).
Kolodkin-Gal, I. et al. D-amino acids trigger biofilm disassembly. Science 328, 627–629 (2010).
Gubeli, R. J., Burger, K. & Weber, W. Synthetic biology for mammalian cell technology and materials sciences. Biotechnol. Adv. 31, 68–78 (2013).
Acknowledgements
We thank J. J. Collins (Biomedical Engineering, Boston University) for donating riboregulator plasmids, R. Weiss (Electrical Engineering and Computer Science, MIT) for the gift of a LuxI plasmid, C. Dorel (Biosciences Department, INSA Lyon) for the gift of E. coli MG1655 ompR234, M. Chapman (Department of Molecular, Cellular, and Developmental Biology, University of Michigan Ann Arbor) for the gift of anti-CsgA antibodies, K. Ribbeck (Department of Biological Engineering, MIT) for use of confocal microscopy facilities, and L. Cameron (Confocal and Light Microscopy Core, Dana Farber Cancer Institute) for assistance with FLIM. We thank C. Zhong, K. Lowenhaupt and P. Siuti from the Lu lab, S. Keating from the lab of N. Oxman (Media Lab, MIT), K. Frederick from the lab of S. Lindquist, S. Lindquist (Whitehead Institute), and E. Dreaden from the lab of P. Hammond (Chemical Engineering, MIT) for helpful discussions. We thank C. Zhong from the Lu lab for the gift of purified CsgA protein. We also thank M. Mimee and O. Purcell from the Lu lab for a close reading of this manuscript. This work was supported by the Office of Naval Research and the Army Research Office. This work was also supported in part by the MRSEC Program of the National Science Foundation under award number DMR-0819762. A.Y.C. acknowledges graduate research support from the Hertz Foundation, the Department of Defense, and NIH Medical Scientist Training Program grant T32GM007753. A.N.B. acknowledges support from NIH-NIEHS Training Grant in Toxicology 5 T32 ES7020-37. T.K.L. acknowledges support from the Presidential Early Career Award for Scientists and Engineers and the NIH New Innovator Award (1DP2OD008435).
Author information
Authors and Affiliations
Contributions
T.K.L. and A.Y.C. conceived the experiments. A.Y.C., Z.D., A.N.B., U.O.S.S., M.Y.L. and R.J.C. performed the experiments, A.Y.C., Z.D., A.N.B. and T.K.L. analysed the data, discussed results, and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
T.K.L. and A.Y.C. have filed a provisional application based on this work with the US Patent and Trademark Office.
Supplementary information
Supplementary Information
Supplementary Information (PDF 3789 kb)
Rights and permissions
About this article
Cite this article
Chen, A., Deng, Z., Billings, A. et al. Synthesis and patterning of tunable multiscale materials with engineered cells. Nature Mater 13, 515–523 (2014). https://doi.org/10.1038/nmat3912
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmat3912
This article is cited by
-
Beneficial applications of biofilms
Nature Reviews Microbiology (2023)
-
Bacterial biofilm functionalization through Bap amyloid engineering
npj Biofilms and Microbiomes (2022)
-
Protein nanowires with tunable functionality and programmable self-assembly using sequence-controlled synthesis
Nature Communications (2022)
-
Engineering living and regenerative fungal–bacterial biocomposite structures
Nature Materials (2022)
-
Synthetic phosphoethanolamine-modified oligosaccharides reveal the importance of glycan length and substitution in biofilm-inspired assemblies
Nature Communications (2022)