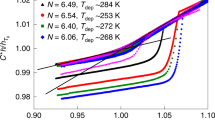
Abstract
Owing to the kinetic nature of the glass transition, the ability to significantly alter the properties of amorphous solids by the typical routes to the vitreous state is restricted. For instance, an order of magnitude change in the cooling rate merely modifies the value of the glass transition temperature (Tg) by a few degrees. Here we show that matrix-assisted pulsed laser evaporation (MAPLE) can be used to form ultrastable and nanostructured glassy polymer films which, relative to the standard poly(methyl methacrylate) glass formed on cooling at standard rates, are 40% less dense, have a 40 K higher Tg, and exhibit a two orders of magnitude enhancement in kinetic stability at high temperatures. The unique set of properties of MAPLE-deposited glasses may make them attractive in technologies where weight and stability are central design issues.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Angell, C. A. Formation of glasses from liquids and biopolymers. Science 267, 1924–1935 (1995).
Debenedetti, P. G. & Stillinger, F. H. Supercooled liquids and the glass transition. Nature 410, 259–267 (2001).
Ediger, M. D., Angell, C. A. & Nagel, S. R. Supercooled liquids and glasses. J. Phys. Chem. 100, 13200–13212 (1996).
McKenna, G. B. Glass Formation and Glassy Behavior (Pergamon, 1989).
Badrinarayanan, P., Zheng, W., Li, Q. & Simon, S. L. The glass transition temperature versus the fictive temperature. J. Non-Cryst. Solids 353, 2603–2612 (2007).
Swallen, S. F. et al. Organic glasses with expectional thermodynamic and kinetic stability. Science 315, 353–356 (2007).
Kearns, K. L., Still, T., Fytas, G. & Ediger, M. D. High-modulus organic glasses prepared by physical vapor deposition. Adv. Mater. 22, 39–42 (2010).
Kearns, K. L., Swallen, S. F., Ediger, M. D., Wu, T. & Yu, L. Influence of substrate temperature on the stability of glasses prepared by vapor deposition. J. Chem. Phys. 127, 154702 (2007).
Kearns, K. L., Swallen, S. F., Ediger, M. D., Wu, T. & Yu, L. Hiking down the energy landscape: Progress toward the Kauzmann temperature via vapor deposition. J. Phys. Chem. B 112, 4934–4942 (2008).
Swallen, S. F., Traynor, K., McMahon, R. J., Ediger, M. D. & Mates, T. E. Stable glass transformation to supercooled liquid via surface-initiated growth front. Phys. Rev. Lett. 102, 065503 (2009).
Leon-Gutierrez, E., Garcia, G., Lopeandia, A. F., Clavaguera-Mora, M. T. & Rodriquez-Viejo, J. Size effects and extraordinary stability of ultrathin vapor deposited glassy films of toluene. J. Phys. Chem. Lett. 1, 341–345 (2010).
Ishii, K., Nakayama, H., Hirabayashi, S. & Moriyama, R. Anomalously high-density glass of ehtylbenzene prepared by vapour deposition at temperatures close to the glass-transition temperature. Chem. Phys. Lett. 459, 109–112 (2008).
Pique, A. et al. Growth of organic thin films by the matrix assisted pulsed laser evaporation (MAPLE) technique. Thin Solid Films 355, 536–541 (1999).
Chrisey, D. B. et al. Laser deposition of polymer and biomaterial films. Chem. Rev. 103, 553–576 (2003).
Angell, C. A. & Wang, L-M. Hyperquenching and cold equilibration strategies for the study of liquid–liquid and protein folding transitions. Biophys. Chem. 105, 621–637 (2003).
Johari, G. P. Calorimetric features of high enthalpy amorphous solids and glass softening temperature of water. J. Phys. Chem. B 107, 9063–9070 (2003).
Stillinger, F. H. A topographic view of supercooled liquids and glass formation. Science 267, 1935–1939 (1995).
Cheng, W. et al. Elastic properties and glass transition of supported polymer thin films. Macromolecules 40, 7283–7290 (2007).
Dawson, K. J., Kearns, K. L., Yu, L., Steffen, W. & Ediger, M. D. Physical vapor deposition as a route to hidden amorphous states. Proc. Natl Acad. Sci. USA 106, 15165–15170 (2009).
Fakhraai, Z. & Forrest, J. A. Measuring the surface dynamics of glassy polymers. Science 319, 600–604 (2008).
Ellison, C. J. & Torkelson, J. M. The distribution of glass transition temperatures in nanoscopically confined glass formers. Nature Mater. 2, 695–700 (2003).
Priestley, R. D., Ellison, C. J., Broadbelt, L. J. & Torkelson, J. M. Structural relaxation of polymer glasses at surfaces, interfaces, and in between. Science 309, 456–459 (2005).
Peter, S., Meyer, H. & Baschnagel, J. Thickness-dependent reduction of the glass-transition temperature in thin polymer films with a free surface. J. Polym. Sci. B 44, 2951–2967 (2006).
Torres, J. A., Nealey, P. F. & de Pablo, J. J. Molecular simulation of ultrathin polymeric films near the glass transition. Phys. Rev. Lett. 85, 3221–3224 (2000).
Shi, Z., Debenedetti, P. G. & Stillinger, F. H. Properties of model atomic free-standing thin films. J. Chem. Phys. 134, 114524 (2011).
Singh, S. & de Pablo, J. J. A molecular view of vapor deposited glasses. J. Chem. Phys. 134, 194903 (2011).
Sellinger, A., Leveugle, E., Fitz-Gerald, J. M. & Zhigilei, L. V. Generation of surface features in films deposited by matrix assisted pulsed laser evaporation: The effects of the stress confinement and droplet landing velocity. Appl. Phys. A 92, 821–829 (2008).
Sima, F. et al. Levan nanostructured thin films by MAPLE assembling. Biomacromolecules 12, 2251–2256 (2011).
Leveugle, E. & Zhigilei, L. V. Molecular dynamics simulation study of the ejection and transport of polymer molecules in matrix-assisted pulsed laser evaporation. J. Appl. Phys. 102, 074914 (2007).
Zhigilei, L. V., Volkov, A. N., Leveugle, E. & Tabetah, M. The effect of the target structure and composition on the ejection and transport of polymer molecules and carbon nanotubes in matrix-assisted pulsed laser evaporation. Appl. Phys. A 105, 529–546 (2011).
De Gennes, P. G. Kinetics of collapse for a flexible coil. J. Phys. Lett. 46, 639–642 (1985).
Aseyev, V., Tenhu, H. & Winnik, F. M. Temperature dependence of the colloidal stability of neutral amphiphilic polymers in water. Adv. Polym. Sci. 196, 1–85 (2006).
McKenna, G. B. Glassy states: Concentration glasses and temperature glasses compared. J. Non-Cryst. Solids 353, 3820–3828 (2007).
Zheng, Y., Priestley, R. D. & McKenna, G. B. Physical aging of an epoxy subsequent to relative humidity jumps through the glass concentration. J. Polym. Sci. B 42, 2107–2121 (2004).
Mi, Y., Xue, G. & Lu, X. A new perspective of the glass transition of polymer single-chain nanoglobules. Macromolecules 36, 7560–7566 (2003).
Mi, Y., Xue, G. & Wang, X. Glass transition of nano-sized single chain globules. Polymer 43, 6701–6705 (2002).
Greiner, R. & Schwarzl, F. R. Thermal contraction and volume relaxation of amorphous polymers. Rheol. Acta 23, 378–395 (1984).
Hodge, I. A. Physical aging in polymer glasses. Science 267, 1945–1947 (1995).
Hutchinson, J. M. Physical aging of polymers. Prog. Polym. Sci. 20, 703–760 (1995).
Acknowledgements
We acknowledge support of the National Science Foundation (NSF) Materials Research Science and Engineering Center program through the Princeton Center for Complex Materials (DMR-0819860) and usage of the PRISM Imaging and Analysis Center at Princeton University. R.D.P. acknowledges partial support from the NSF through a CAREER Award (DMR-1053144). We thank R. A. Register and P. G. Debenedetti for their comments during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
Y.G. designed and set up the MAPLE system, performed experiments on MAPLE, DSC, GPC, NMR, WAXS, XRR, refractive index, temperature-dependent AFM, discussed and analysed the results and wrote the manuscript. A.M. designed the MAPLE system, and carried out laser calibration and trouble-shooting. D.S. performed BLS and SEM measurements. J.W.C. performed NMR and WAXS measurements and discussed the results. C.Z. performed MAPLE experiments. M.W. performed FTIR and AFM measurements. N.Y. assisted in WAXS and XRR characterization and provided equipment for temperature control on AFM. G.F. coordinated the BLS study, discussed results and commented on the manuscript. C.B.A. coordinated the set up of the MAPLE system, discussed the results and commented on the manuscript. R.D.P. conceived the idea, coordinated the project, discussed the results and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 678 kb)
Rights and permissions
About this article
Cite this article
Guo, Y., Morozov, A., Schneider, D. et al. Ultrastable nanostructured polymer glasses. Nature Mater 11, 337–343 (2012). https://doi.org/10.1038/nmat3234
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmat3234
This article is cited by
-
Hybrid magnetron sputtering of ceramic superlattices for application in a next generation of combustion engines
Scientific Reports (2022)
-
Ultrastable glasses: new perspectives for an old problem
La Rivista del Nuovo Cimento (2022)
-
The looks of a million-year-old polymer glass
Nature Materials (2020)
-
Ultrastable monodisperse polymer glass formed by physical vapour deposition
Nature Materials (2020)
-
Ultrastable metallic glasses formed on cold substrates
Nature Communications (2018)