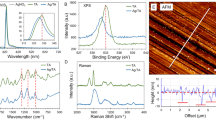
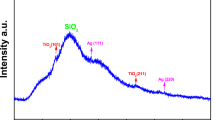
Abstract
Developing bactericidal coatings using simple green chemical methods could be a promising route to potential environmentally friendly applications. Here, we describe an environmentally friendly chemistry approach to synthesize metal-nanoparticle (MNP)-embedded paint, in a single step, from common household paint. The naturally occurring oxidative drying process in oils, involving free-radical exchange, was used as the fundamental mechanism for reducing metal salts and dispersing MNPs in the oil media, without the use of any external reducing or stabilizing agents. These well-dispersed MNP-in-oil dispersions can be used directly, akin to commercially available paints, on nearly all kinds of surface such as wood, glass, steel and different polymers. The surfaces coated with silver-nanoparticle paint showed excellent antimicrobial properties by killing both Gram-positive human pathogens (Staphylococcus aureus) and Gram-negative bacteria (Escherichia coli). The process we have developed here is quite general and can be applied in the synthesis of a variety of MNP-in-oil systems.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Daniel, S. (ed.) Bailey’s Industrial Oil and Fat Products (Wiley, New York, 1964).
Metzger, J. O. & Bornscheuer, U. Lipids as renewable resources: current state of chemical and biotechnological conversion and diversification. Appl. Microbiol. Biotechnol. 71, 13–22 (2006).
Bieleman, J. H. Additives for Coatings (Wiley-VCH, Weinheim, 2000).
Black, J. F. Metal-catalyzed autoxidation. The unrecognized consequences of metal-hydroperoxide complex formation. J. Am. Chem. Soc. 100, 527–535 (1978).
Reich, L. & Stivala, S. Autoxidation of Hydrocarbons and Polyolefins (Marcel Dekker, New York, 1969).
Bohannon, J. ‘Smart coatings’ research shows the virtues of superficiality. Science 309, 376–377 (2005).
Crisp, M. T. & Kotov, N. A. Preparation of nanoparticle coatings on surfaces of complex geometry. Nano Lett. 3, 173–177 (2003).
Klaus, T., Joerger, R., Olsson, E. & Granqvist, C.-G. Silver-based crystalline nanoparticles, microbially fabricated. Proc. Natl Acad. Sci. USA 96, 13611–13614 (1999).
Zhang, J. et al. Sonochemical formation of single-crystalline gold nanobelts. Angew. Chem. Int. Edn 45, 1116–1119 (2006).
Okitsu, K. et al. Synthesis of palladium nanoparticles with interstitial carbon by sonochemical reduction of tetrachloropalladate(II) in aqueous solution. J. Phys. Chem. B 101, 5470–5472 (1997).
Aymonier, C. et al. Hybrids of silver nanoparticles with amphiphilic hyperbranched macromolecules exhibiting antimicrobial properties. Chem. Commun. 3018–3019 (2002).
Lu, Y., Liu, G. L. & Lee, L. P. High-density silver nanoparticle film with temperature-controllable interparticle spacing for a tunable surface enhanced raman scattering substrate. Nano Lett. 5, 5–9 (2005).
Heilmann, A. Polymer Films with Embedded Metal Nanoparticles (Springer, New York, 2002).
Dowling, D. P. et al. Anti-bacterial silver coatings exhibiting enhanced activity through the addition of platinum. Surf. Coat. Technol. 163, 637–640 (2003).
Jiang, H., Manolache, S., Wong, A. C. L. & Denes, F. S. Plasma-enhanced deposition of silver nanoparticles onto polymer and metal surfaces for the generation of antimicrobial characteristics. J. Appl. Polym. Sci. 93, 1411–1422 (2004).
Dai, J. & Bruening, M. L. Catalytic nanoparticles formed by reduction of metal ions in multilayered polyelectrolyte films. Nano Lett. 2, 497–501 (2002).
Mallia, V. A., Vemula, P. K., John, G., Kumar, A. & Ajayan, P. M. In situ synthesis and assembly of gold nanoparticles embedded glass forming liquid crystals. Angew. Chem. Int. Edn 46, 3269–3274 (2007).
Vemula, P. K., Aslam, U., Mallia, V. A. & John, G. In situ synthesis of gold nanoparticles using molecular gels and liquid crystals from vitamin-C amphiphiles. Chem. Mater. 19, 138–140 (2007).
Vemula, P. K. & John, G. Smart amphiphiles: hydro/organogelators for in situ reduction of gold. Chem. Commun. 2218–2220 (2006).
Sambhy, V., MacBride, M. M., Peterson, B. R. & Sen, A. Silver bromide nanoparticle/polymer composites: Dual action tunable antimicrobial materials. J. Am. Chem. Soc. 128, 9798–9808 (2006).
Lansdown, A. B. Silver. I: Its antibacterial properties and mechanism of action. J. Wound Care. 11, 125–130 (2002).
Kenawy, E.-R., Worley, S. D. & Broughton, R. The chemistry and applications of antimicrobial polymers: A state-of-the-art review. Biomacromolecules 8, 1359–1384 (2007).
Williams, R. L., Doherty, P. J., Vince, D. G., Grashoff, G. J. & Williams, D. F. The biocompatibility of silver. Crit. Rev. Biocompat. 5, 221–243 (1989).
Berger, T. J., Spadaro, J. A., Chapin, S. E. & Becker, R. O. Electrically generated silver ions: Quantitative effects on bacterial and mammalian cells. Antimicrob. Agents Chemother. 9, 357–358 (1976).
Alt, V. et al. An in vitro assessment of the antibacterial properties and cytotoxicity of nanoparticulate silver bone cement. Biomaterials 25, 4383 (2004).
Podsiadlo, P. et al. Layer-by-layer assembly of nacre-like nanostructured composites with antimicrobial properties. Langmuir 21, 11915–11921 (2005).
Morones, J. R. et al. The bactericidal effect of silver nanoparticles. Nanotechnology 16, 2346–2353 (2005).
Gogoi, S. K. et al. Green fluorescent protein-expressing Escherichia coli as a model system for investigating the antimicrobial activities of silver nanoparticles. Langmuir 22, 9322–9328 (2006).
Haldar, J., An, D., de Cienfuegos, L. A., Chen, J. & Klibanov, A. M. Polymeric coatings that inactivate both influenza virus and pathogenic bacteria. Proc. Natl Acad. Sci. USA 103, 17667–17671 (2006).
Lewis, K. & Klibanov, A. M. Surpassing nature: Rational design of sterile-surface materials. Trends Biotechnol. 23, 343–348 (2005).
Naik, R. R., Stringer, S. J., Agarwal, G., Jones, S. E. & Stone, M. O. Biomimetic synthesis and patterning of silver nanoparticles. Nature Mater. 1, 169–172 (2002).
Raveendran, P., Fu, J. & Wallen, S. L. Completely “green” synthesis and stabilization of metal nanoparticles. J. Am. Chem. Soc. 125, 13940–13941 (2003).
Yin, H. & Porter, N. A. New insights regarding the autoxidation of polyunsaturated fatty acids. Antioxid. Redox Signal. 7, 170–184 (2005).
Anastas, P. T. & Williamson, T. C. Green Chemistry: Frontiers in Benign Chemical Syntheses and Processes (Oxford Univ. Press, Oxford, 1998).
Tyman, J. H. P. Non-isoprenoid long chain phenols. Chem. Soc. Rev. 8, 499–537 (1979).
John, G. & Pillai, C. K. S. Self-crosslinkable monomer from cardanol: Crosslinked beads of poly(cardanyl acrylate) by suspension polymerization. Makromol. Chem. Rapid Commun. 13, 255–259 (1992).
John, G. & Pillai, C. K. S. Synthesis and characterization of a self-crosslinkable polymer from cardanol: Autooxidation of poly(cardanyl acrylate) to crosslinked film. J. Polym. Sci. A 31, 1069–1073 (1993).
Jin, R. et al. Photoinduced conversion of silver nanospheres to nanoprisms. Science 294, 1901–1903 (2001).
van Gorkum, R. & Bouwman, E. The oxidative drying of alkyd paint catalysed by metal complexes. Coordination Chem. Rev. 249, 1709–1728 (2005).
Tang, L., Zhang, Y., Qian, Z. & Shen, X. The mechanism of Fe2+-initiated lipid peroxidation in liposomes: The dual function of ferrous ions, the roles of the pre-existing lipid peroxides and the lipid peroxyl radical. Biochem. J. 352, 27–36 (2000).
Ahmed-Choudhury, J., Orsler, D. J. & Coleman, R. Hepatobiliary effects of tertiary-butylhydroperoxide (tBOOH) in isolated rat hepatocyte couplets. Toxicol. Appl. Pharmacol. 152, 270–275 (1998).
Yin, H., Morrow, J. D. & Porter, N. A. Identification of a novel class of endoperoxides from arachidonate autoxidation. J. Biol. Chem. 279, 3766–3776 (2004).
Esterbauer, H., Schauer, R. J. & Zollner, H. Chemistry and biochemistry of 4-hydroxynonenal, malonaldehyde and related aldehydes. Free Radic. Biol. Med. 11, 81–128 (1991).
Nath, S., Ghosh, S. K., Panigrahi, S. & Pal, T. Aldehyde assisted wet chemical route to synthesize gold nanoparticles. Ind. J. Chem. A 43, 1147–1151 (2004).
Zhang, Z., Berg, A., Levanon, H., Fessenden, R. W. & Meisel, D. On the interactions of free radicals with gold nanoparticles. J. Am. Chem. Soc. 125, 7959–7963 (2003).
Russel, A. D., Path, F. R. C. & Hugo, W. B. Antimicrobial activity and action of silver. Prog. Med. Chem. 31, 351–370 (1994).
Zachariadis, P. C. et al. Synthesis, characterization and in vitro study of the cytostatic and antiviral activity of new polymeric silver(I) complexes with ribbon structures derived from the conjugated heterocyclic thioamide 2-mercapto-3,4,5,6-tetrahydropyrimidine. Eur. J. Inorg. Chem. 2004, 1420–1426 (2004).
Sondi, I. & Salopek-Sondi, B. Silver nanoparticles as antimicrobial agent: a case study on E. coli as a model for Gram-negative bacteria. J. Colloid Interface Sci. 275, 177–182 (2004).
Sudhir, K. Preparation, characterization, and surface modification of silver particles. Langmuir 14, 1021–1025 (1998).
Acknowledgements
J. Haldar from MIT is acknowledged for discussions. A. Janakiraman, Department of Biology, CCNY is acknowledged for assisting with the microbial experiments. G.J. acknowledges the Science Interdepartmental Electron Microscope and Imaging Center at CCNY. P.M.A. acknowledges funding from NSEC at RPI.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Information
Supplementary figures S1-S6 and supplementary table S1 (PDF 472 kb)
Rights and permissions
About this article
Cite this article
Kumar, A., Vemula, P., Ajayan, P. et al. Silver-nanoparticle-embedded antimicrobial paints based on vegetable oil. Nature Mater 7, 236–241 (2008). https://doi.org/10.1038/nmat2099
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmat2099
This article is cited by
-
Revolutionizing Textile Applications: Unveiling the Potential of NiFe2O4 Nanoparticles for Enhanced Performance
Fibers and Polymers (2024)
-
Green Synthesis of Silver Nanoparticles by Fusarium oxysporum and its Function Against Aspergillus and Fusarium Fungi
Indian Journal of Microbiology (2024)
-
Supertool for Superbugs—Smart “Nano-ointment of Graphene Usnic Acid Nanoparticles” Against Antimicrobial Resistance (AMRs)
BioNanoScience (2023)
-
Silver nanoparticles produced via a green synthesis using the rhamnolipid as a reducing agent and stabilizer
Applied Nanoscience (2023)
-
Breaking the nanoparticle’s dispersible limit via rotatable surface ligands
Nature Communications (2022)