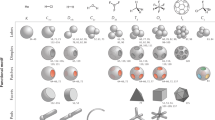
Abstract
A revolution in novel nanoparticles and colloidal building blocks has been enabled by recent breakthroughs in particle synthesis. These new particles are poised to become the ‘atoms’ and ‘molecules’ of tomorrow’s materials if they can be successfully assembled into useful structures. Here, we discuss the recent progress made in the synthesis of nanocrystals and colloidal particles and draw analogies between these new particulate building blocks and better-studied molecules and supramolecular objects. We argue for a conceptual framework for these new building blocks based on anisotropy attributes and discuss the prognosis for future progress in exploiting anisotropy for materials design and assembly.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Matijevic, E. Monodispersed metal (hydrous) oxides—a fascinating field of colloid science. Acc. Chem. Res. 14, 22–29 (1981).
Buining, P. A., Pathmamanoharan, C., Jansen, J. B. H. & Lekkerkerker, H. N. W. Preparation of colloidal boehmite needles by hydrothermal treatment of aluminum alkoxide precursors. J. Am. Chem. Soc. 74, 1303–1307 (1991).
Ozaki, M., Kratohvil, S. & Matijevic, E. Formation of mondispersed spindle-type hematite particles. J. Colloid Interface Sci. 102, 146–151 (1984).
van der Kooij, F. M., Kassapidou, K. & Lekkerkerker, H. N. W. Liquid crystal phase transitions in suspensions of polydisperse plate-like particles. Nature 406, 868–871 (2000).
LizMarzan, L. M., Giersig, M. & Mulvaney, P. Synthesis of nanosized gold-silica core-shell particles. Langmuir 12, 4329–4335 (1996).
Caruso, F., Caruso, R. A. & Möhwald, H. Nanoengineering of inorganic and hybrid hollow spheres by colloidal templating. Science 282, 1111–1114 (1998).
Keville, K. M., Franses, E. I. & Caruthers, J. M. Preparation and characterization of monodisperse polymer microspheroids. J. Colloid Interface Sci. 144, 103–126 (1991).
Adams, M., Dogic, Z., Keller, S. L. & Fraden, S. Entropically driven microphase transitions in mixtures of colloidal rods and spheres. Nature 393, 349–352 (1998).
Jana, N. R., Gearheart, L. & Murphy, C. J. Wet chemical synthesis of high aspect ratio cylindrical gold nanorods. J. Phys. Chem. B 105, 4065–4067 (2001).
Sun, Y. G. & Xia, Y. N. Shape-controlled synthesis of gold and silver nanoparticles. Science 298, 2176–2179 (2002).
Manna, L., Scher, E. C. & Alivisatos, A. P. Synthesis of soluble and processable rod-, arrow-, teardrop-, and tetrapod-shaped CdSe nanocrystals. J. Am. Chem. Soc. 122, 12700–12706 (2000).
Jin, R. C. et al. Photoinduced conversion of silver nanospheres to nanoprisms. Science 294, 1901–1903 (2001).
Cayre, O. J., Paunov, V. N. & Velev, O. D. Fabrication of asymmetrically coated colloid particles by microcontact printing techniques. J. Mater. Chem. 13, 2445–2450 (2003).
Roh, K. H., Martin, D. C. & Lahann, J. Biphasic Janus particles with nanoscale anisotropy. Nature Mater. 4, 759–763 (2005).
Dendukuri, D., Pregibon, D. C., Collins, J., Hatton, T. A. & Doyle, P. S. Continuous-flow lithography for high-throughput microparticle synthesis. Nature Mater. 5, 365–369 (2006).
Braeckmans, K., De Smedt, S. C., Leblans, M., Pauwels, R. & Demeester, J. Encoding microcarriers: Present and future technologies. Nature Rev. Drug Discovery 1, 447–456 (2002).
Manoharan, V. N., Elsesser, M. T. & Pine, D. J. Dense packing and symmetry in small clusters of microspheres. Science 301, 483–487 (2003).
Yin, Y. D., Lu, Y., Gates, B. & Xia, Y. N. Template-assisted self-assembly: A practical route to complex aggregates of monodispersed colloids with well-defined sizes, shapes, and structures. J. Am. Chem. Soc. 123, 8718–8729 (2001).
Rolland, J. P. et al. Direct fabrication and harvesting of monodisperse, shape-specific nanobiomaterials. J. Am. Chem. Soc. 127, 10096–10100 (2005).
Hernandez, C. J. & Mason, T. G. Colloidal alphabet soup: Monodisperse dispersions of shape-designed lithoparticles. J. Phys. Chem. C 111, 4477–4480 (2007).
Shankar, S. S. et al. Biological synthesis of triangular gold nanoprisms. Nature Mater. 3, 482–488 (2004).
Bansal, V., Sanyal, A., Rautaray, D., Ahmad, A. & Sastry, M. Bioleaching of sand by the fungus Fusarium oxysporum as a means of producing extracellular silica nanoparticles. Adv. Mater. 17, 889–892 (2005).
Brown, S., Sarikaya, M. & Johnson, E. A genetic analysis of crystal growth. J. Mol. Biol. 299, 725–735 (2000).
Gou, L. F. & Murphy, C. J. Solution-phase synthesis of Cu2O nanocubes. Nano Lett. 3, 231–234 (2003).
Ahmadi, T. S., Wang, Z. L., Green, T. C., Henglein, A. & El-Sayed, M. A. Shape-controlled synthesis of colloidal platinum nanoparticles. Science 272, 1924–1926 (1996).
Malikova, N., Pastoriza-Santos, I., Schierhorn, M., Kotov, N. A. & Liz-Marzan, L. M. Layer-by-layer assembled mixed spherical and planar gold nanoparticles: Control of interparticle interactions. Langmuir 18, 3694–3697 (2002).
Greyson, E. C., Barton, J. E. & Odom, T. W. Tetrahedral zinc blende tin sulfide nano- and microcrystals. Small 2, 368–371 (2006).
Hong, L., Cacciuto, A., Luijten, E. & Granick, S. Clusters of charged Janus spheres. Nano Lett. 6, 2510–2514 (2006).
Love, J. C., Gates, B. D., Wolfe, D. B., Paul, K. E. & Whitesides, G. M. Fabrication and wetting properties of metallic half-shells with submicron diameters. Nano Lett. 2, 891–894 (2002).
Jackson, A. M., Myerson, J. W. & Stellacci, F. Spontaneous assembly of subnanometre-ordered domains in the ligand shell of monolayer-protected nanoparticles. Nature Mater. 3, 330–336 (2004).
Chen, S. H., Wang, Z. L., Ballato, J., Foulger, S. H. & Carroll, D. L. Monopod, bipod, tripod, and tetrapod gold nanocrystals. J. Am. Chem. Soc. 125, 16186–16187 (2003).
Lee, S.-M., Jun, Y., Cho, S.-N. & Cheon, J. Single-crystalline star-shaped nanocrystals and their evolution: Programming the geometry of nano-building blocks. J. Am. Chem. Soc. 124, 11244–11245 (2002).
Tang, Z. Y., Wang, Y., Shanbhag, S., Giersig, M. & Kotov, N. A. Spontaneous transformation of CdTe nanoparticles into angled Te nanocrystals: From particles and rods to checkmarks, X-marks, and other unusual shapes. J. Am. Chem. Soc. 128, 6730–6736 (2006).
Zhang, G., Wang, D. Y. & Möhwald, H. Decoration of microspheres with gold nanodots-giving colloidal spheres valences. Angew. Chem. Int. Edn 44, 7767–7770 (2005).
DeVries, G. A. et al. Divalent metal nanoparticles. Science 315, 358–361 (2007).
Burda, C., Chen, X., Narayanan, R. & El-Sayed, M. A. Chemistry and properties of nanocrystals of different shapes. Chem. Rev. 105, 1025–1102 (2005).
Xia, Y. & Halas, N. J. Synthesis and plasmonic properties of nanostructures. Mater. Res. Soc. Bull. 30, 338–343 (2005).
Glotzer, S. C. Some assembly required. Science 306, 419–420 (2004).
Glotzer, S. C., Solomon, M. J. & Kotov, N. A. Self-assembly: From nanoscale to microscale colloids. AIChE J. 50, 2978–2985 (2004).
van Blaaderen, A. Colloids get complex. Nature 439, 545–546 (2006).
Yethiraj, A. & van Blaaderen, A. A colloidal model system with an interaction tunable from hard sphere to soft and dipolar. Nature 421, 513–517 (2003).
Leunissen, M. E. et al. Ionic colloidal crystals of oppositely charged particles. Nature 437, 235–240 (2005).
Bartlett, P. & Campbell, A. I. Three-dimensional binary superlattices of oppositely charged colloids. Phys. Rev. Lett. 95, 128302 (2005).
Maskaly, G. R., Garcia, E. R., Carter, W. C. & Chiang, Y.-M. Ionic colloidal crystals: Ordered, multicomponent structures via controlled heterocoagulation. Phys. Rev. E 73, 014402 (2006).
Shevchenko, E. V., Talapin, D. V., Kotov, N. A., O’Brien, S. & Murray, C. B. Structural diversity in binary nanoparticle superlattices. Nature 439, 55–59 (2006).
Hynninen, A. P., Christova, C. G., van Roij, R., van Blaaderen, A. & Dijkstra, M. Prediction and observation of crystal structures of oppositely charged colloids. Phys. Rev. Lett. 96, 138308 (2006).
Zhang, Z. L. & Glotzer, S. C. Self-assembly of patchy particles. Nano Lett. 4, 1407–1413 (2004).
Mokari, T., Rothenberg, E., Popov, I., Costi, R. & Banin, U. Selective growth of metal tips onto semiconductor quantum rods and tetrapods. Science 304, 1787–1790 (2004).
Zhang, Z. L., Horsch, M. A., Lamm, M. H. & Glotzer, S. C. Tethered nano building blocks: Toward a conceptual framework for nanoparticle self-assembly. Nano Lett. 3, 1341–1346 (2003).
Loweth, C. J., Caldwell, W. B., Peng, X. P., Alivisatos, A. P. & Schultz, P. G. DNA-based assembly of gold nanocrystals. Angew. Chem. Int. Edn 38, 1808–1812 (1999).
Nelson, D. R. Toward a tetravalent chemistry of colloids. Nano Lett. 2, 1125–1129 (2002).
De Michele, C., Gabrielli, S., Tartaglia, P. & Sciortino, F. Dynamics in the presence of attractive patchy interactions. J. Phys. Chem. B 110, 8064–8079 (2006).
Bianchi, E., Largo, J., Tartaglia, P., Zaccarelli, E. & Sciortino, F. Phase diagram of patchy colloids: Towards empty liquids. Phys. Rev. Lett. 97, 168301 (2006).
Kamien, R. D. The geometry of soft materials: A primer. Rev. Mod. Phys. 74, 953–971 (2002).
Ziherl, P. & Kamien, R. D. Maximizing entropy by minimzing area: Towards a new principle of self-organization. J. Phys. Chem. B 105, 10147–10158 (2001).
de Gennes, P. G. Soft matter. Rev. Mod. Phys. 64, 645–648 (1992).
Casagrande, C. & Veyssié, M. Janus beads—realization and 1st observation of interfacial properties. C.R. Acad. Sci. 306, 1423–1425 (1988).
Iacovella, C. R., Horsch, M. A., Zhang, Z. & Glotzer, S. C. Phase diagrams of self-assembled mono-tethered nanospheres from molecular simulation and comparison to surfactants. Langmuir 21, 9488–9494 (2005).
Van Workum, K. & Douglas, J. F. Symmetry, equivalence, and molecular self-assembly. Phys. Rev. E 73, 031502 (2006).
Mohraz, A. & Solomon, M. J. Direct visualization of colloidal rod assembly by confocal microscopy. Langmuir 21, 5298–5306 (2005).
Donev, A., Burton, J., Stillinger, F. H. & Torquato, S. Tetratic order in the phase behavior of a hard-rectangle system. Phys. Rev. B 73, 054109 (2006).
Blaak, R., Mulder, B. M. & Frenkel, D. Cubatic phase for tetrapods. J. Chem. Phys. 120, 5486–5492 (2004).
Schilling, T., Pronk, S., Mulder, B. & Frenkel, D. Monte Carlo study of hard pentagons. Phys. Rev. E 71, 036138 (2005).
John, B. S., Stroock, A. & Escobedo, F. A. Cubatic liquid-crystalline behavior in a system of hard cuboids. J. Chem. Phys. 120, 9383–9389 (2004).
Gracias, D. H., Tien, J., Breen, T. L., Hsu, C. & Whitesides, G. M. Forming electrical networks in three dimensions by self-assembly. Science 289, 1170–1172 (2000).
Zhang, Z. L., Keys, A. S., Chen, T. & Glotzer, S. C. Self-assembly of patchy particles into diamond structures through molecular mimicry. Langmuir 21, 11547–11551 (2005).
Zerrouki, D. et al. Preparation of doublet, triangular, and tetrahedral colloidal clusters by controlled emulsification. Langmuir 22, 57–62 (2006).
Rechtsman, M. C., Stillinger, F. H. & Torquato, S. Self-assembly of the simple cubic lattice with an isotropic potential. Phys. Rev. E 74, 021404 (2006).
Tkachenko, A. V. Morphological diversity of DNA-colloidal self-assembly. Phys. Rev. Lett. 89, 148303 (2002).
Petukhov, A. V. et al. Observation of a hexatic columnar liquid crystal of polydisperse colloidal disks. Phys. Rev. Lett. 95, 077801 (2005).
Manna, L., Milliron, D. J., Meisel, A., Scher, E. C. & Alivisatos, A. P. Controlled growth of tetrapod-branched inorganic nanocrystals. Nature Mater. 2, 382–385 (2003).
Johnson, P. M., van Kats, C. M. & van Blaaderen, A. Synthesis of colloidal silica dumbbells. Langmuir 21, 11510–11517 (2005).
Lu, Y. et al. Asymmetric dimers can be formed by dewetting half-shells of gold deposited on the surfaces of spherical oxide colloids. J. Am. Chem. Soc. 125, 12724–12725 (2003).
Cho, K.-S., Talapin, D. V., Gaschler, W. & Murray, C. Designing PbSe nanowires and nanorings through oriented attachment of nanoparticles. J. Am. Chem. Soc. 127, 7140–7147 (2005).
Peng, X. G. et al. Shape control of CdSe nanocrystals. Nature 404, 59–61 (2000).
Yu, Y.-Y., Chang, S.-S., Lee, C.-L. & Wang, C. C. R. Gold nanorods: Electrochemical synthesis and optical properties. J. Phys. Chem. B 101, 6661–6664 (1997).
Jackson, A. M., Hu, Y., Silva, P. J. & Stellacci, F. From homoligand- to mixed-ligand-monolayer-protected metal nanoparticles: A scanning tunneling microscopy investigation. J. Am. Chem. Soc. 128, 11135–11149 (2006).
Martin, B. R. et al. Orthogonal self-assembly on colloidal gold-platinum nanorods. Adv. Mater. 11, 1021–1025 (1999).
Acknowledgements
S.C.G. acknowledges support from DoE, NSF and NASA. M.J.S. acknowledges support from NASA and NSF. We are grateful to C. R. Iacovella in the Glotzer group for his graphic artistry in rendering the figures.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Glotzer, S., Solomon, M. Anisotropy of building blocks and their assembly into complex structures. Nature Mater 6, 557–562 (2007). https://doi.org/10.1038/nmat1949
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nmat1949
This article is cited by
-
The forefront of chemical engineering research
Nature Chemical Engineering (2024)
-
On the Global Minimum of the Classical Potential Energy for Clusters Bound by Many-Body Forces
Journal of Statistical Physics (2024)
-
Liquid spherical shells are a non-equilibrium steady state of active droplets
Nature Communications (2023)
-
Machine learning for nanoplasmonics
Nature Nanotechnology (2023)
-
Linear growth of colloidal dumbbells into three-lobed polymer nanoparticles mediated by a gradient in surface wettability
Colloid and Polymer Science (2023)