Abstract
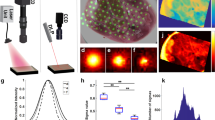
Transport parameters determine the access of drugs to tumors. However, technical difficulties preclude the measurement of these parameters deep inside living tissues. To this end, we adapted and further optimized two-photon fluorescence correlation microscopy (TPFCM) for in vivo measurement of transport parameters in tumors. TPFCM extends the detectable range of diffusion coefficients in tumors by one order of magnitude, and reveals both a fast and a slow component of diffusion. The ratio of these two components depends on molecular size and can be altered in vivo with hyaluronidase and collagenase. These studies indicate that TPFCM is a promising tool to dissect the barriers to drug delivery in tumors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Jain, R.K. The next frontier of molecular medicine: delivery of therapeutics. Nat. Med. 4, 655–657 (1998).
Chary, S.R. & Jain, R.K. Direct measurement of interstitial convection and diffusion of albumin in normal and neoplastic tissues by fluorescence photobleaching. Proc. Natl. Acad. Sci. USA 86, 5385–5389 (1989).
Flamion, B., Bungay, P.M., Gibson, C.C. & Spring, K.R. Flow rate measurements in isolated perfused kidney tubules by fluorescence photobleaching recovery. Biophys. J. 60, 1229–1242 (1991).
Pluen, A. et al. Role of tumor-host interactions in interstitial diffusion of macromolecules: cranial vs. subcutaneous tumors. Proc. Natl. Acad. Sci. USA 98, 4628–4633 (2001).
Netti, P.A., Berk, D.A., Swartz, M.A., Grodzinsky, A.J. & Jain, R.K. Role of extracellular matrix assembly in interstitial transport in solid tumors. Cancer Res. 60, 2497–2503 (2000).
Elson, E.L. & Magde, D. Fluorescence correlation spectroscopy. I. Conceptual basis and theory. Biopolymers 13, 1–27 (1974).
Magde, D., Elson, E.L. & Webb, W.W. Fluorescence correlation spectroscopy. II. An experimental realization. Biopolymers 13, 29–61 (1974).
Qian, H. & Elson, E.L. Analysis of confocal laser-microscope optics for 3-D fluorescence correlation spectroscopy. Appl. Opt. 30, 1185–1195 (1991).
Rigler, R., Mets, U., Windegren, J. & Kask, P. Fluorescence correlation spectroscopy with high count rate and low background: analysis of translational diffusion. Eur. Biophys. J. 22, 169–175 (1993).
Brock, R., Hink, M.A. & Jovin, T.M. Fluorescence correlation microscopy of cells in the presence of autofluorescence. Biophys. J. 75, 2547–2557 (1998).
Brock, R., Vamosi, G., Vereb, G. & Jovin, T.M. Rapid characterization of green fluorescent protein fusion proteins on the molecular and cellular level by fluorescence correlation microscopy. Proc. Natl. Acad. Sci. USA 96, 10123–10128 (1999).
Berland, K.M., So, P.T. & Gratton, E. Two-photon fluorescence correlation spectroscopy: method and application to the intracellular environment. Biophys. J. 68, 694–701 (1995).
Schwille, P., Haupts, U., Maiti, S. & Webb, W.W. Molecular dynamics in living cells observed by fluorescence correlation spectroscopy with one- and two-photon excitation. Biophys. J. 77, 2251–2265 (1999).
Berk, D.A., Yuan, F., Leunig, M. & Jain, R.K. Direct in vivo measurement of targeted binding in a human tumor xenograft. Proc. Natl. Acad. Sci. USA 94, 1785–1790 (1997).
Aukland, K. & Reed, R.K. Interstitial-lymphatic mechanisms in the control of extracellular fluid volume. Physiol. Rev. 73, 1–78 (1993).
Jain, R.K. Transport of molecules in the tumor interstitium: a review. Cancer Res. 47, 3039–3051 (1987).
Brown, E.B. et al. Dynamic imaging of collagen and its modulation in tumors in vivo using second-harmonic generation. Nature Med. 9, 796–800 (2003).
Brown, E.B., Wu, E.S., Zipfel, W. & Webb, W.W. Measurement of molecular diffusion in solution by multiphoton fluorescence photobleaching recovery. Biophys. J. 77, 2837–2849 (1999).
Ramanujan, S. et al. Diffusion and convection in collagen gels: implications for transport in the tumor interstitium. Biophys. J. 83, 1650–1660 (2002).
Shenoy, V. & Rosenblatt, J. Diffusion of macromolecules in collagen and hyaluronic acid, rigid-rod—flexible polymer, composite matrices. Macromolecules 28, 8751–8758 (1995).
Smith, K.J., Skelton, H.G., Turiansky, G. & Wagner, K.F. Hyaluronidase enhances the therapeutic effect of vinblastine in intralesional treatment of Kaposi's sarcoma. Military Medical Consortium for the Advancement of Retroviral Research (MMCARR). J. Am. Acad. Dermatol. 36, 239–242 (1997).
Beckenlehner, K. et al. Hyaluronidase enhances the activity of adriamycin in breast cancer models in vitro and in vivo. J. Cancer Res. Clin. Oncol. 118, 591–596 (1992).
Masuda, A., Ushida, K., Koshino, H., Yamashita, K. & Kluge, T. Novel distance dependence of diffusion constants in hyaluronan aqueous solution resulting from its characteristic nano-microstructure. J. Am. Chem. Soc. 123, 11468–11471 (2001).
De Smedt, S. et al. Structural information on hyaluronic acid solutions as studied by probe diffusion experiments. Macromolecules 27, 141–146 (1994).
Qiu, X.L. et al. Effect of hyaluronidase on albumin diffusion in lung interstitium. Lung 177, 273–288 (1999).
Pluen, A., Netti, P.A., Jain, R.K. & Berk, D.A. Diffusion of macromolecules in agarose gels: comparison of linear and globular configurations. Biophys. J. 77, 542–552 (1999).
Szoka, F. Jr. & Papahadjopoulos, D. Comparative properties and methods of preparation of lipid vesicles (liposomes). Annu. Rev. Biophys. Bioeng. 9, 467–508 (1980).
Leunig, M. et al. Angiogenesis, microvascular architecture, microhemodynamics, and interstitial fluid pressure during early growth of human adenocarcinoma LS174T in SCID mice. Cancer Res. 52, 6553–6560 (1992).
Brown, E.B. et al. In vivo measurement of gene expression, angiogenesis and physiological function in tumors using multiphoton laser scanning microscopy. Nat. Med. 7, 864–868 (2001).
Wohland, T., Rigler, R. & Vogel, H. The standard deviation in fluorescence correlation spectroscopy. Biophys. J. 80, 2987–2999 (2001).
Acknowledgements
We thank J. Kahn for dorsal skinfold chamber preparations; S. Roberge and R. Delgiacco for histological preparations; W. Zipfel for discussion and help with software analysis; and Z. Demou and M. Booth for comments on the manuscript. This work was supported by National Cancer Institute fellowships to G.A. (T32CA73479 and F32CA97818) and E.B.B. (F32CA88490), a National Science Foundation Graduate Research Fellowship to R.T.T., a Biotechnology Fellowship (T32GM08334) to T.D.M., and a Bioengineering Research Partnership Grant (R24 CA85140) and Program Project Grant (P01CA80124) to R.K.J.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Alexandrakis, G., Brown, E., Tong, R. et al. Two-photon fluorescence correlation microscopy reveals the two-phase nature of transport in tumors. Nat Med 10, 203–207 (2004). https://doi.org/10.1038/nm981
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm981
This article is cited by
-
Crystal ribcage: a platform for probing real-time lung function at cellular resolution
Nature Methods (2023)
-
Maximizing response to intratumoral immunotherapy in mice by tuning local retention
Nature Communications (2022)
-
Transport and interactions of nanoparticles in the kidneys
Nature Reviews Materials (2018)
-
Precise nanomedicine for intelligent therapy of cancer
Science China Chemistry (2018)
-
Consensus guidelines for the use and interpretation of angiogenesis assays
Angiogenesis (2018)