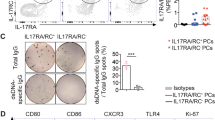
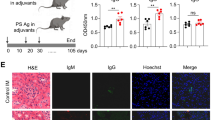
Abstract
Autoantibody-mediated diseases like myasthenia gravis, autoimmune hemolytic anemia and systemic lupus erythematosus represent a therapeutic challenge. In particular, long-lived plasma cells producing autoantibodies resist current therapeutic and experimental approaches. Recently, we showed that the sensitivity of myeloma cells toward proteasome inhibitors directly correlates with their immunoglobulin synthesis rates. Therefore, we hypothesized that normal plasma cells are also hypersensitive to proteasome inhibition owing to their extremely high amount of protein biosynthesis. Here we show that the proteasome inhibitor bortezomib, which is approved for the treatment of multiple myeloma, eliminates both short- and long-lived plasma cells by activation of the terminal unfolded protein response. Treatment with bortezomib depleted plasma cells producing antibodies to double-stranded DNA, eliminated autoantibody production, ameliorated glomerulonephritis and prolonged survival of two mouse strains with lupus-like disease, NZB/W F1 and MRL/lpr mice. Hence, the elimination of autoreactive plasma cells by proteasome inhibitors might represent a new treatment strategy for antibody-mediated diseases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Lipsky, P.E. Systemic lupus erythematosus: an autoimmune disease of B cell hyperactivity. Nat. Immunol. 2, 764–766 (2001).
Mills, J.A. Systemic lupus erythematosus. N. Engl. J. Med. 330, 1871–1879 (1994).
Manz, R.A. & Radbruch, A. Plasma cells for a lifetime? Eur. J. Immunol. 32, 923–927 (2002).
Miller, J.J., III & Cole, L.J. Resistance of long-lived lymphocytes and plasma cells in rat lymph nodes to treatment with prednisone, cyclophosphamide, 6 mercaptopurine and actinomycin D. J. Exp. Med. 126, 109–125 (1967).
Slifka, M.K. & Ahmed, R. Long-lived plasma cells: a mechanism for maintaining persistent antibody production. Curr. Opin. Immunol. 10, 252–258 (1998).
Ang, H.A., Apperley, J.F. & Ward, K.N. Persistence of antibody to human parvovirus B19 after allogeneic bone marrow transplantation: role of prior recipient immunity. Blood 89, 4646–4651 (1997).
van Tol, M.J. et al. The origin of IgG production and homogeneous IgG components after allogeneic bone marrow transplantation. Blood 87, 818–826 (1996).
Theofilopoulos, A.N. & Dixon, F.J. Murine models of systemic lupus erythematosus. Adv. Immunol. 37, 269–390 (1985).
Hoyer, B.F. et al. Short-lived plasmablasts and long-lived plasma cells contribute to chronic humoral autoimmunity in NZB/W mice. J. Exp. Med. 199, 1577–1584 (2004).
Manz, R.A., Hauser, A.E., Hiepe, F. & Radbruch, A. Maintenance of serum antibody levels. Annu. Rev. Immunol. 23, 367–386 (2005).
Adams, J. The proteasome: a suitable antineoplastic target. Nat. Rev. Cancer 4, 349–360 (2004).
Hideshima, T., Richardson, P.G. & Anderson, K.C. Targeting proteasome inhibition in hematologic malignancies. Rev. Clin. Exp. Hematol. 7, 191–204 (2003).
Rajkumar, S.V., Richardson, P.G., Hideshima, T. & Anderson, K.C. Proteasome inhibition as a novel therapeutic target in human cancer. J. Clin. Oncol. 23, 630–639 (2005).
Meister, S. et al. Extensive immunoglobulin production sensitizes myeloma cells for proteasome inhibition. Cancer Res. 67, 1783–1792 (2007).
Obeng, E.A. et al. Proteasome inhibitors induce a terminal unfolded protein response in multiple myeloma cells. Blood 107, 4907–4916 (2006).
Hibi, T. & Dosch, H.M. Limiting dilution analysis of the B cell compartment in human bone marrow. Eur. J. Immunol. 16, 139–145 (1986).
Lefkowith, J.B. & Gilkeson, G.S. Nephritogenic autoantibodies in lupus: current concepts and continuing controversies. Arthritis Rheum. 39, 894–903 (1996).
Hahn, B.H. Antibodies to DNA. N. Engl. J. Med. 338, 1359–1368 (1998).
Manz, R.A., Thiel, A. & Radbruch, A. Lifetime of plasma cells in the bone marrow. Nature 388, 133–134 (1997).
Radbruch, A. et al. Competence and competition: the challenge pf becoming a long-lived plasma cell. Nat. Rev. Immunol. 6, 741–750 (2006).
Nencioni, A. et al. Proteasome inhibitor bortezomib modulates TLR4-induced dendritic cell activation. Blood 108, 551–558 (2006).
Straube, C. et al. Bortezomib significantly impairs the immunostimulatory capacity of human myeloid blood dendritic cells. Leukemia 21, 1464–1471 (2007).
Oyadomari, S. & Mori, M. Roles of CHOP/GADD153 in endoplasmic reticulum stress. Cell Death Differ. 11, 381–389 (2004).
Moran, S.T. et al. Synergism between NF-κB1/p50 and Notch2 during the development of marginal zone B lymphocytes. J. Immunol. 179, 195–200 (2007).
Sasaki, Y. et al. Canonical NF-κB activity, dispensable for B cell development, replaces BAFF-receptor signals and promotes B cell proliferation upon activation. Immunity 24, 729–739 (2006).
Maseda, D., Meister, S., Neubert, K., Herrmann, M. & Voll, R.E. Proteasome inhibition drastically but reversibly impairs murine lymphocyte development. Cell Death Differ. 15, 600–612 (2008).
Schubert, U. et al. Rapid degradation of a large fraction of newly synthesized proteins by proteasomes. Nature 404, 770–774 (2000).
Gass, J.N., Gifford, N.M. & Brewer, J.W. Activation of an unfolded protein response during differentiation of antibody-secreting B cells. J. Biol. Chem. 277, 49047–49054 (2002).
Iwakoshi, N.N. et al. Plasma cell differentiation and the unfolded protein response intersect at the transcription factor XBP-1. Nat. Immunol. 4, 321–329 (2003).
Kaufman, R.J. Stress signaling from the lumen of the endoplasmic reticulum: coordination of gene transcriptional and translational controls. Genes Dev. 13, 1211–1233 (1999).
Patil, C. & Walter, P. Intracellular signaling from the endoplasmic reticulum to the nucleus: the unfolded protein response in yeast and mammals. Curr. Opin. Cell Biol. 13, 349–355 (2001).
Reimold, A.M. et al. Plasma cell differentiation requires the transcription factor XBP-1. Nature 412, 300–307 (2001).
van Anken, E. et al. Sequential waves of functionally related proteins are expressed when B cells prepare for antibody secretion. Immunity 18, 243–253 (2003).
Wiertz, E.J. et al. Sec61-mediated transfer of a membrane protein from the endoplasmic reticulum to the proteasome for destruction. Nature 384, 432–438 (1996).
Kim, R., Emi, M., Tanabe, K. & Murakami, S. Role of the unfolded protein response in cell death. Apoptosis 11, 5–13 (2006).
Nawrocki, S.T. et al. Bortezomib inhibits PKR-like endoplasmic reticulum (ER) kinase and induces apoptosis via ER stress in human pancreatic cancer cells. Cancer Res. 65, 11510–11519 (2005).
Nawrocki, S.T. et al. Bortezomib sensitizes pancreatic cancer cells to endoplasmic reticulum stress-mediated apoptosis. Cancer Res. 65, 11658–11666 (2005).
Schaumann, D.H., Tuischer, J., Ebell, W., Manz, R.A. & Lauster, R. VCAM-1–positive stromal cells from human bone marrow producing cytokines for B lineage progenitors and for plasma cells: SDF-1, flt3L and BAFF. Mol. Immunol. 44, 1606–1612 (2007).
Tokoyoda, K., Egawa, T., Sugiyama, T., Choi, B.I. & Nagasawa, T. Cellular niches controlling B lymphocyte behavior within bone marrow during development. Immunity 20, 707–718 (2004).
Klinman, D.M. & Steinberg, A.D. Inquiry into murine and human lupus. Immunol. Rev. 144, 157–193 (1995).
Winfield, J.B., Faiferman, I. & Koffler, D. Avidity of anti-DNA antibodies in serum and IgG glomerular eluates from patients with systemic lupus erythematosus. Association of high avidity antinative DNA antibody with glomerulonephritis. J. Clin. Invest. 59, 90–96 (1977).
Cambridge, G. et al. B cell depletion therapy in systemic lupus erythematosus: effect on autoantibody and antimicrobial antibody profiles. Arthritis Rheum. 54, 3612–3622 (2006).
Sfikakis, P.P. et al. Remission of proliferative lupus nephritis following B cell depletion therapy is preceded by down-regulation of the T cell costimulatory molecule CD40 ligand: an open-label trial. Arthritis Rheum. 52, 501–513 (2005).
Leandro, M.J., Edwards, J.C., Cambridge, G., Ehrenstein, M.R. & Isenberg, D.A. An open study of B lymphocyte depletion in systemic lupus erythematosus. Arthritis Rheum. 46, 2673–2677 (2002).
Emery, P. et al. The efficacy and safety of rituximab in patients with active rheumatoid arthritis despite methotrexate treatment: results of a phase IIB randomized, double-blind, placebo-controlled, dose-ranging trial. Arthritis Rheum. 54, 1390–1400 (2006).
Looney, R.J. et al. B cell depletion as a novel treatment for systemic lupus erythematosus: a phase I/II dose-escalation trial of rituximab. Arthritis Rheum. 50, 2580–2589 (2004).
Medina, F., Segundo, C., Campos-Caro, A., Gonzalez-Garcia, I. & Brieva, J.A. The heterogeneity shown by human plasma cells from tonsil, blood, and bone marrow reveals graded stages of increasing maturity, but local profiles of adhesion molecule expression. Blood 99, 2154–2161 (2002).
Benner, R., Van Oudenaren, A. & Koch, G. Induction of antibody formation in mouse bone marrow. in Immunological Methods Vol. II (eds. Pernis, B. & Lefkovits, I.) 247–262 (Academic Press, New York, 1981).
Wellmann, U., Letz, M., Schneider, A., Amann, K. & Winkler, T.H. An Ig μ-heavy chain transgene inhibits systemic lupus erythematosus immunopathology in autoimmune (NZB x NZW)F1 mice. Int. Immunol. 13, 1461–1469 (2001).
Acknowledgements
We are grateful to U. Appelt for expert cell sorting and M. Wiesener and F. Nimmerjahn for critical reading the manuscript. This work was supported by the Interdisciplinary Center for Clinical Research (project number N2) and the German Research Society (project VO673/31 and Collaborative Research Centers SFB 643; project B3, both to R.E.V.). Parts of this work were funded by an intramural grant from the ELAN fond, a Training Grant GK 592 from the German Research Society and Collaborative Research Centers SFB 423 (project Z2).
Author information
Authors and Affiliations
Contributions
K.N. designed experiments; performed animal studies, flow cytometry, ELISAs, ELISPOTs and quantitative real-time RT-PCR; analyzed data; generated figures and wrote the manuscript. S.M. assisted in performing flow cytometry and animal experiments and participated in discussions. K.M. performed and assisted with the BrdU experiments, provided antibodies and participated in discussions. F.W. performed RNA isolation and cDNA synthesis and participated in discussions. D.M. assisted with the animal studies. K.A. performed all histological analyses. C.W. contributed to the analyses of DC and T cell function. T.H.W. provided crucial ideas, participated in discussions and edited the manuscript. J.R.K. participated in discussions and edited the manuscript. R.A.M. crucially participated in design and analyses of the BrdU labeling experiments, provided antibodies, participated in discussions and edited the manuscript. R.E.V. provided crucial ideas, designed the study and experiments, supervised the study and wrote the manuscript together with K.N.
Corresponding author
Ethics declarations
Competing interests
R.E.V. holds stocks of Millenium Pharmaceuticals, the manufacturer of bortezomib (value of less than $15,000).
K.N., S.M., K.M., R.A.M., D.M., J.R.K. and R.E.V. are inventors listed on a patent application for the use of proteasome inhibitors to deplete plasma cells.
Supplementary information
Supplementary Text and Figures
Supplementary Figs. 1–9 and Supplementary Methods (PDF 227 kb)
Rights and permissions
About this article
Cite this article
Neubert, K., Meister, S., Moser, K. et al. The proteasome inhibitor bortezomib depletes plasma cells and protects mice with lupus-like disease from nephritis. Nat Med 14, 748–755 (2008). https://doi.org/10.1038/nm1763
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1763
This article is cited by
-
Large-scale computational modelling of the M1 and M2 synovial macrophages in rheumatoid arthritis
npj Systems Biology and Applications (2024)
-
A narrative review of potential drug treatments for nephritis in children with IgA vasculitis (HSP)
Clinical Rheumatology (2023)
-
Immune Mechanisms in Epileptogenesis: Update on Diagnosis and Treatment of Autoimmune Epilepsy Syndromes
Drugs (2023)
-
Targeted Small Molecules for Systemic Lupus Erythematosus: Drugs in the Pipeline
Drugs (2023)
-
Clinical research progress of novel biologics for the treatment of lupus nephritis
Clinical and Experimental Medicine (2023)