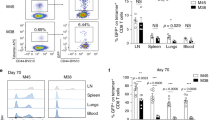
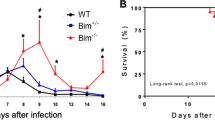
Abstract
In this study, we document the development of stable, antigen-independent CD8+ T cell memory after drug-induced cure of a chronic infection. By establishing a system for drug cure of chronic Trypanosoma cruzi infection, we present the first extensively documented case of total parasite clearance after drug treatment of this infection. Cure resulted in the emergence of a stable, parasite-specific CD8+ T cell population with the characteristics of central memory cells, based upon expression of CD62L, CCR7, CD127, CD122, Bcl-2 and a reduced immediate in vivo CTL function. CD8+ T cells from treated and cured mice also expanded more rapidly and provided greater protection following challenge than those from chronically infected mice. These results show that complete pathogen clearance results in stable, antigen-independent and protective T cell memory, despite the potentially exhausting effects of prior long-term exposure to antigen in this chronic infection.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Grayson, J.M., Zajac, A.J., Altman, J.D. & Ahmed, R. Cutting edge: increased expression of Bcl-2 in antigen-specific memory CD8+ T cells. J. Immunol. 164, 3950–3954 (2000).
Wherry, E.J., Barber, D.L., Kaech, S.M., Blattman, J.N. & Ahmed, R. Antigen-independent memory CD8 T cells do not develop during chronic viral infection. Proc. Natl. Acad. Sci. USA 101, 16004–16009 (2004).
Boyman, O., Purton, J.F., Surh, C.D. & Sprent, J. Cytokines and T-cell homeostasis. Curr. Opin. Immunol. 19, 320–326 (2007).
Sallusto, F., Lenig, D., Forster, R., Lipp, M. & Lanzavecchia, A. Two subsets of memory T lymphocytes with distinct homing potentials and effector functions. Nature 401, 708–712 (1999).
Jameson, S.C. Maintaining the norm: T-cell homeostasis. Nat. Rev. Immunol. 2, 547–556 (2002).
Becker, T.C. et al. Interleukin 15 is required for proliferative renewal of virus-specific memory CD8 T cells. J. Exp. Med. 195, 1541–1548 (2002).
Goldrath, A.W. et al. Cytokine requirements for acute and basal homeostatic proliferation of naive and memory CD8+ T cells. J. Exp. Med. 195, 1515–1522 (2002).
Zaph, C., Uzonna, J., Beverley, S.M. & Scott, P. Central memory T cells mediate long-term immunity to Leishmania major in the absence of persistent parasites. Nat. Med. 10, 1104–1110 (2004).
Cush, S.S., Anderson, K.M., Ravneberg, D.H., Weslow-Schmidt, J.L. & Flano, E. Memory generation and maintenance of CD8+ T cell function during viral persistence. J. Immunol. 179, 141–153 (2007).
Bengsch, B. et al. Analysis of CD127 and KLRG1 expression on hepatitis C virus–specific CD8+ T cells reveals the existence of different memory T-cell subsets in the peripheral blood and liver. J. Virol. 81, 945–953 (2007).
Shin, H., Blackburn, S.D., Blattman, J.N. & Wherry, E.J. Viral antigen and extensive division maintain virus-specific CD8 T cells during chronic infection. J. Exp. Med. 204, 941–949 (2007).
Day, C.L. et al. PD-1 expression on HIV-specific T cells is associated with T-cell exhaustion and disease progression. Nature 443, 350–354 (2006).
Barber, D.L. et al. Restoring function in exhausted CD8 T cells during chronic viral infection. Nature 439, 682–687 (2006).
Rehermann, B. Chronic infections with hepatotropic viruses: mechanisms of impairment of cellular immune responses. Semin. Liver Dis. 27, 152–160 (2007).
Shin, H. & Wherry, E.J. CD8 T cell dysfunction during chronic viral infection. Curr. Opin. Immunol. 19, 408–415 (2007).
Brooks, D.G. et al. Interleukin-10 determines viral clearance or persistence in vivo. Nat. Med. 12, 1301–1309 (2006).
Ejrnaes, M. et al. Resolution of a chronic viral infection after interleukin-10 receptor blockade. J. Exp. Med. 203, 2461–2472 (2006).
Vezys, V. et al. Continuous recruitment of naive T cells contributes to heterogeneity of antiviral CD8 T cells during persistent infection. J. Exp. Med. 203, 2263–2269 (2006).
Prata, A. Clinical and epidemiological aspects of Chagas disease. Lancet Infect. Dis. 1, 92–100 (2001).
WHO Expert Committee. Control of Chagas disease. (WHO Technical Report Series 905) 120 (World Health Organization, Geneva, 2002).
Tarleton, R.L. Depletion of CD8+ T cells increases susceptibility and reverses vaccine-induced immunity in mice infected with Trypanosoma cruzi. J. Immunol. 144, 717–724 (1990).
Tarleton, R.L., Koller, B.H., Latour, A. & Postan, M. Susceptibility of β2-microglobulin–deficient mice to Trypanosoma cruzi infection. Nature 356, 338–340 (1992).
Tarleton, R.L., Sun, J., Zhang, L. & Postan, M. Depletion of T-cell subpopulations results in exacerbation of myocarditis and parasitism in experimental Chagas disease. Infect. Immun. 62, 1820–1829 (1994).
Martin, D.L. & Tarleton, R.L. Antigen-specific T cells maintain an effector memory phenotype during persistent Trypanosoma cruzi infection. J. Immunol. 174, 1594–1601 (2005).
Martin, D.L. et al. CD8+ T-cell responses to Trypanosoma cruzi are highly focused on strain-variant trans-sialidase epitopes. PLoS Pathog. 2, e77 (2006).
Laucella, S.A. et al. Frequency of interferon-γ–producing T cells specific for Trypanosoma cruzi inversely correlates with disease severity in chronic human Chagas disease. J. Infect. Dis. 189, 909–918 (2004).
Albareda, M.C. et al. Trypanosoma cruzi modulates the profile of memory CD8+ T cells in chronic Chagas' disease patients. Int. Immunol. 18, 465–471 (2006).
Rodriques Coura, J. & de Castro, S.L. A critical review on Chagas disease chemotherapy. Mem. Inst. Oswaldo Cruz 97, 3–24 (2002).
Zajac, A.J. et al. Viral immune evasion due to persistence of activated T cells without effector function. J. Exp. Med. 188, 2205–2213 (1998).
Zhang, J.-Y. et al. PD-1 up-regulation is correlated with HIV-specific memory CD8+ T-cell exhaustion in typical progressors but not in long-term nonprogressors. Blood 109, 4671–4678 (2007).
Penna, A. et al. Dysfunction and functional restoration of HCV-specific CD8 responses in chronic hepatitis C virus infection. Hepatology 45, 588–601 (2007).
Zhang, L. & Tarleton, R.L. Parasite persistence correlates with disease severity and localization in chronic Chagas' disease. J. Infect. Dis. 180, 480–486 (1999).
Dutton, R.W., Bradley, L.M. & Swain, S.L. T cell memory. Annu. Rev. Immunol. 16, 201–223 (1998).
Voehringer, D., Koschella, M. & Pircher, H. Lack of proliferative capacity of human effector and memory T cells expressing killer cell lectinlike receptor G1 (KLRG1). Blood 100, 3698–3702 (2002).
Sallusto, F., Geginat, J. & Lanzavecchia, A. Central memory and effector memory T cell subsets: function, generation and maintenance. Annu. Rev. Immunol. 22, 745–763 (2004).
Gallimore, A., Dumrese, T., Hengartner, H., Zinkernagel, R.M. & Rammensee, H.G. Protective immunity does not correlate with the hierarchy of virus-specific cytotoxic T cell responses to naturally processed peptides. J. Exp. Med. 187, 1647–1657 (1998).
Jabbari, A. & Harty, J.T. Secondary memory CD8+ T cells are more protective but slower to acquire a central-memory phenotype. J. Exp. Med. 203, 919–932 (2006).
Wherry, E.J., Blattman, J.N., Murali-Krishna, K., van der Most, R. & Ahmed, R. Viral persistence alters CD8 T-cell immunodominance and tissue distribution and results in distinct stages of functional impairment. J. Virol. 77, 4911–4927 (2003).
Sevilla, N. et al. Immunosuppression and resultant viral persistence by specific viral targeting of dendritic cells. J. Exp. Med. 192, 1249–1260 (2000).
Yao, Z.Q., King, E., Prayther, D., Yin, D. & Moorman, J. T cell dysfunction by hepatitis C virus core protein involves PD-1/PDL-1 signaling. Viral Immunol. 20, 276–287 (2007).
Seder, R.A. & Ahmed, R. Similarities and differences in CD4+ and CD8+ effector and memory T cell generation. Nat. Immunol. 4, 835–842 (2003).
Homann, D., Teyton, L. & Oldstone, M.B. Differential regulation of antiviral T-cell immunity results in stable CD8+ but declining CD4+ T-cell memory. Nat. Med. 7, 913–919 (2001).
Belkaid, Y., Piccirillo, C.A., Mendez, S., Shevach, E.M. & Sacks, D.L. CD4+CD25+ regulatory T cells control Leishmania major persistence and immunity. Nature 420, 502–507 (2002).
Urbina, J.A. et al. Parasitological cure of acute and chronic experimental Chagas disease using the long-acting experimental triazole TAK-187. Activity against drug-resistant Trypanosoma cruzi strains. Int. J. Antimicrob. Agents 21, 39–48 (2003).
Romanha, A.J. et al. Experimental chemotherapy against Trypanosoma cruzi infection: essential role of endogenous interferon-γ in mediating parasitologic cure. J. Infect. Dis. 186, 823–828 (2002).
Engel, J.C., Doyle, P.S., Hsieh, I. & McKerrow, J.H. Cysteine protease inhibitors cure an experimental Trypanosoma cruzi infection. J. Exp. Med. 188, 725–734 (1998).
Viotti, R., Vigliano, C., Armenti, H. & Segura, E. Treatment of chronic Chagas' disease with benznidazole: clinical and serologic evolution of patients with long-term follow-up. Am. Heart J. 127, 151–162 (1994).
Cancado, J.R. Long term evaluation of etiological treatment of Chagas disease with benznidazole. Rev. Inst. Med. Trop. Sao Paulo 44, 29–37 (2002).
Cummings, K.L. & Tarleton, R.L. Rapid quantitation of Trypanosoma cruzi in host tissue by real-time PCR. Mol. Biochem. Parasitol. 129, 53–59 (2003).
Acknowledgements
We thank J. Nelson of the Center for Tropical and Emerging Global Diseases Flow Cytometry Facility at the University of Georgia for technical assistance, C. Boehlke and G. Cooley for assistance with the parasites, M. Collins for critical reading of the manuscript, V. Alvarez and J. Cola Fernandes Rodrigues for assistance with the figures, and the Tetramer Core Facility (Emory University) for synthesis of MHC class I tetramers. The ELC-immunoglobulin chimera was a gift from K. Klonowski (University of Georgia). This work was supported by US National Institutes of Health grants AI-22070 and AI-33106 to R.L.T.
Author information
Authors and Affiliations
Contributions
J.M.B. designed and performed experiments and wrote the manuscript, L.M.B. performed experiments and assisted in writing of the manuscript and R.L.T. designed experiments and wrote the manuscript.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figs. 1 and 2 (PDF 72 kb)
Rights and permissions
About this article
Cite this article
Bustamante, J., Bixby, L. & Tarleton, R. Drug-induced cure drives conversion to a stable and protective CD8+ T central memory response in chronic Chagas disease. Nat Med 14, 542–550 (2008). https://doi.org/10.1038/nm1744
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1744
This article is cited by
-
Vaccine-linked chemotherapy induces IL-17 production and reduces cardiac pathology during acute Trypanosoma cruzi infection
Scientific Reports (2021)
-
Proteasome inhibition for treatment of leishmaniasis, Chagas disease and sleeping sickness
Nature (2016)
-
CD8+ T cells in Trypanosoma cruzi infection
Seminars in Immunopathology (2015)
-
Frequency of IFNγ-producing T cells correlates with seroreactivity and activated T cells during canine Trypanosoma cruzi infection
Veterinary Research (2014)
-
Perpetual expression of PAMPs necessary for optimal immune control and clearance of a persistent pathogen
Nature Communications (2013)