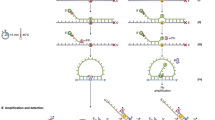
Abstract
PCR is widely employed as the initial DNA amplification step for genetic testing. However, a key limitation of PCR-based methods is the inability to selectively amplify low levels of mutations in a wild-type background. As a result, downstream assays are limited in their ability to identify subtle genetic changes that can have a profound impact in clinical decision-making and outcome. Here we describe co-amplification at lower denaturation temperature PCR (COLD-PCR), a novel form of PCR that amplifies minority alleles selectively from mixtures of wild-type and mutation-containing sequences irrespective of the mutation type or position on the sequence. We replaced regular PCR with COLD-PCR before sequencing or genotyping assays to improve mutation detection sensitivity by up to 100-fold and identified new mutations in the genes encoding p53, KRAS and epidermal growth factor in heterogeneous cancer samples that had been missed by the currently used methods. For clinically relevant microdeletions, COLD-PCR enabled exclusive amplification and isolation of the mutants. COLD-PCR will transform the capabilities of PCR-based genetic testing, including applications in cancer, infectious diseases and prenatal identification of fetal alleles in maternal blood.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Kobayashi, S. et al. EGFR mutation and resistance of non–small-cell lung cancer to gefitinib. N. Engl. J. Med. 352, 786–792 (2005).
Hoffmann, C. et al. DNA bar coding and pyrosequencing to identify rare HIV drug resistance mutations. Nucleic Acids Res. 35, e91 (2007).
Lo, Y.M. et al. Presence of fetal DNA in maternal plasma and serum. Lancet 350, 485–487 (1997).
Paez, J.G. et al. EGFR mutations in lung cancer: correlation with clinical response to gefitinib therapy. Science 304, 1497–1500 (2004).
Janne, P.A. et al. A rapid and sensitive enzymatic method for epidermal growth factor receptor mutation screening. Clin. Cancer Res. 12, 751–758 (2006).
Engelman, J.A. et al. Allelic dilution obscures detection of a biologically significant resistance mutation in EGFR-amplified lung cancer. J. Clin. Invest. 116, 2695–2706 (2006).
Diehl, F. et al. Detection and quantification of mutations in the plasma of patients with colorectal tumors. Proc. Natl. Acad. Sci. USA 102, 16368–16373 (2005).
Kimura, T. et al. Mutant DNA in plasma of lung cancer patients: potential for monitoring response to therapy. Ann. NY Acad. Sci. 1022, 55–60 (2004).
Li, J. et al. s-RT-MELT for rapid mutation scanning using enzymatic selection and real time DNA-melting: new potential for multiplex genetic analysis. Nucleic Acids Res. 35, e84 (2007).
Lipsky, R.H. et al. DNA melting analysis for detection of single nucleotide polymorphisms. Clin. Chem. 47, 635–644 (2001).
Liew, M. et al. Genotyping of single-nucleotide polymorphisms by high-resolution melting of small amplicons. Clin. Chem. 50, 1156–1164 (2004).
Yeung, A.T., Hattangadi, D., Blakesley, L. & Nicolas, E. Enzymatic mutation detection technologies. Biotechniques 38, 749–758 (2005).
Ogino, S. et al. Sensitive sequencing method for KRAS mutation detection by pyrosequencing. J. Mol. Diagn. 7, 413–421 (2005).
Huang, C. et al. Mutations in exon 7 and 8 of TP53 as poor prognostic factors in patients with non-small cell lung cancer. Oncogene 16, 2469–2477 (1998).
Huang, C.L. et al. Mutations of TP53 and K-ras genes as prognostic factors for non-small cell lung cancer. Int. J. Oncol. 12, 553–563 (1998).
Jackson, P.E. et al. Specific TP53 mutations detected in plasma and tumors of hepatocellular carcinoma patients by electrospray ionization mass spectrometry. Cancer Res. 61, 33–35 (2001).
Shao, Z.M., Wu, J., Shen, Z.Z. & Nguyen, M. TP53 mutation in plasma DNA and its prognostic value in breast cancer patients. Clin. Cancer Res. 7, 2222–2227 (2001).
Mayall, F., Jacobson, G., Wilkins, R. & Chang, B. Mutations of TP53 gene can be detected in the plasma of patients with large bowel carcinoma. J. Clin. Pathol. 51, 611–613 (1998).
Silva, J.M. et al. Tumor DNA in plasma at diagnosis of breast cancer patients is a valuable predictor of disease-free survival. Clin. Cancer Res. 8, 3761–3766 (2002).
Gonzalez, R. et al. Microsatellite alterations and TP53 mutations in plasma DNA of small-cell lung cancer patients: follow-up study and prognostic significance. Ann. Oncol. 11, 1097–1104 (2000).
Eberhard, D.A. et al. Mutations in the epidermal growth factor receptor and in KRAS are predictive and prognostic indicators in patients with non-small-cell lung cancer treated with chemotherapy alone and in combination with erlotinib. J. Clin. Oncol. 23, 5900–5909 (2005).
Thomas, R.K. et al. Sensitive mutation detection in heterogeneous cancer specimens by massively parallel picoliter reactor sequencing. Nat. Med. 12, 852–855 (2006).
Thomas, R.K. et al. High-throughput oncogene mutation profiling in human cancer. Nat. Genet. 39, 347–351 (2007).
Sun, X., Hung, K., Wu, L., Sidransky, D. & Guo, B. Detection of tumor mutations in the presence of excess amounts of normal DNA. Nat. Biotechnol. 20, 186–189 (2002).
Fuery, C.J. et al. Detection of rare mutant alleles by restriction endonuclease-mediated selective-PCR: assay design and optimization. Clin. Chem. 46, 620–624 (2000).
Belinsky, S.A. et al. Gene promoter methylation in plasma and sputum increases with lung cancer risk. Clin. Cancer Res. 11, 6505–6511 (2005).
Greenman, C. Patterns of somatic mutation in human cancer genomes. Nature 446, 153–158 (2007).
Acknowledgements
We gratefully acknowledge the assistance of A. Brown at the Harvard Partners Center for Genetics and Genomics High Throughput Genotyping Facility and of M. Miri, F. Haluska and P. Janne in obtaining specimens from the Massachusetts General Hospital Tumor Bank and Dana Farber Cancer Institute. We also acknowledge B. Price and A. D'Andrea for valuable comments on the manuscript. This work was supported by training grant 5 T32 CA09078 (J.L.) and US National Institutes of Health grants CA111994-01 and CA115439-01.
Author information
Authors and Affiliations
Contributions
J.L. and L.W., experimental design; H.M. and M.H.K., clinical considerations and rationale; R.B., modeling; G.M.M., project setup, experimental design and manuscript preparation.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figs. 1–8 and Supplementary Table 1 (PDF 883 kb)
Rights and permissions
About this article
Cite this article
Li, J., Wang, L., Mamon, H. et al. Replacing PCR with COLD-PCR enriches variant DNA sequences and redefines the sensitivity of genetic testing. Nat Med 14, 579–584 (2008). https://doi.org/10.1038/nm1708
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1708
This article is cited by
-
Detection of low-frequency mutations in clinical samples by increasing mutation abundance via the excision of wild-type sequences
Nature Biomedical Engineering (2023)
-
Limitations and opportunities of technologies for the analysis of cell-free DNA in cancer diagnostics
Nature Biomedical Engineering (2022)
-
Selective extraction of low-abundance BRAF V600E mutation from plasma, urine, and sputum using ion-tagged oligonucleotides and magnetic ionic liquids
Analytical and Bioanalytical Chemistry (2022)
-
Enhanced asymmetric blocked qPCR method for affordable detection of point mutations in KRAS oncogene
Analytical and Bioanalytical Chemistry (2021)
-
Somatic mutation detection efficiency in EGFR: a comparison between high resolution melting analysis and Sanger sequencing
BMC Cancer (2020)