Abstract
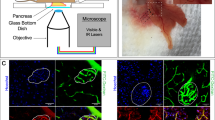
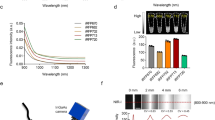
Advanced imaging techniques have become a valuable tool in the study of complex biological processes at the cellular level in biomedical research. Here, we introduce a new technical platform for noninvasive in vivo fluorescence imaging of pancreatic islets using the anterior chamber of the eye as a natural body window. Islets transplanted into the mouse eye engrafted on the iris, became vascularized, retained cellular composition, responded to stimulation and reverted diabetes. Laser-scanning microscopy allowed repetitive in vivo imaging of islet vascularization, beta cell function and death at cellular resolution. Our results thus establish the basis for noninvasive in vivo investigations of complex cellular processes, like beta cell stimulus-response coupling, which can be performed longitudinally under both physiological and pathological conditions.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Wajchenberg, B.L. Beta cell failure in diabetes and preservation by clinical treatment. Endocr. Rev. 28, 187–218 (2007).
Berggren, P.O. & Leibiger, I.B. Novel aspects on signal transduction in the pancreatic beta cell. Nutr. Metab. Cardiovasc. Dis. 16 Suppl 1, S7–S10 (2006).
Vetterlein, F., Petho, A. & Schmidt, G. Morphometric investigation of the microvascular system of pancreatic exocrine and endocrine tissue in the rat. Microvasc. Res. 34, 231–238 (1987).
Woods, S.C. & Porte, D., Jr. Neural control of the endocrine pancreas. Physiol. Rev. 54, 596–619 (1974).
Rahier, J., Goebbels, R.M. & Henquin, J.C. Cellular composition of the human diabetic pancreas. Diabetologia 24, 366–371 (1983).
Köhler, M. et al. Imaging of pancreatic beta cell signal transduction. Curr. Med. Chem.-Immun. Endoc. & Metab. Agents 4, 281–299 (2004).
Speier, S. & Rupnik, M. A novel approach to in situ characterization of pancreatic beta cells. Pflugers Arch. 446, 553–558 (2003).
Ricordi, C. Islet transplantation: a brave new world. Diabetes 52, 1595–1603 (2003).
Paty, B.W., Bonner-Weir, S., Laughlin, M.R., McEwan, A.J. & Shapiro, A.M. Toward development of imaging modalities for islets after transplantation: insights from the National Institutes of Health Workshop on Beta Cell Imaging. Transplantation 77, 1133–1137 (2004).
Menger, M.D., Yamauchi, J. & Vollmar, B. Revascularization and microcirculation of freely grafted islets of Langerhans. World J. Surg. 25, 509–515 (2001).
Porksen, N. et al. Coordinate pulsatile insulin secretion by chronic intraportally transplanted islets in the isolated perfused rat liver. J. Clin. Invest. 94, 219–227 (1994).
Meier, J.J. et al. Intrahepatic transplanted islets in humans secrete insulin in a coordinate pulsatile manner directly into the liver. Diabetes 55, 2324–2332 (2006).
Adeghate, E. & Donath, T. Morphological findings in long-term pancreatic tissue transplants in the anterior eye chamber of rats. Pancreas 5, 298–305 (1990).
Hoffer, B., Seiger, A., Ljungberg, T. & Olson, L. Electrophysiological and cytological studies of brain homografts in the anterior chamber of the eye: maturation of cerebellar cortex in oculo. Brain Res. 79, 165–184 (1974).
Katoh, N. et al. Target-specific innervation by autonomic and sensory nerve fibers in hairy fetal skin transplanted into the anterior eye chamber of adult rat. Cell Tissue Res. 266, 259–263 (1991).
Adeghate, E., Ponery, A.S., Ahmed, I. & Donath, T. Comparative morphology and biochemistry of pancreatic tissue fragments transplanted into the anterior eye chamber and subcutaneous regions of the rat. Eur. J. Morphol. 39, 257–268 (2001).
Menger, M.D., Vajkoczy, P., Leiderer, R., Jager, S. & Messmer, K. Influence of experimental hyperglycemia on microvascular blood perfusion of pancreatic islet isografts. J. Clin. Invest. 90, 1361–1369 (1992).
Szkudelski, T. The mechanism of alloxan and streptozotocin action in B cells of the rat pancreas. Physiol. Res. 50, 537–546 (2001).
Lipp, P. & Niggli, E. Ratiometric confocal Ca2+ measurements with visible wavelength indicators in isolated cardiac myocytes. Cell Calcium 14, 359–372 (1993).
Quesada, I., Nadal, A. & Soria, B. Different effects of tolbutamide and diazoxide in alpha, beta, and delta cells within intact islets of Langerhans. Diabetes 48, 2390–2397 (1999).
Valdeolmillos, M., Santos, R.M., Contreras, D., Soria, B. & Rosario, L.M. Glucose-induced oscillations of intracellular Ca2+ concentration resembling bursting electrical activity in single mouse islets of Langerhans. FEBS Lett. 259, 19–23 (1989).
Mathis, D., Vence, L. & Benoist, C. Beta cell death during progression to diabetes. Nature 414, 792–798 (2001).
Butler, A.E. et al. Beta cell deficit and increased beta cell apoptosis in humans with type 2 diabetes. Diabetes 52, 102–110 (2003).
Boersma, H.H. et al. Past, present, and future of annexin A5: from protein discovery to clinical applications. J. Nucl. Med. 46, 2035–2050 (2005).
Medarova, Z., Bonner-Weir, S., Lipes, M. & Moore, A. Imaging beta cell death with a near-infrared probe. Diabetes 54, 1780–1788 (2005).
Nyqvist, D., Köhler, M., Wahlstedt, H. & Berggren, P.O. Donor islet endothelial cells participate in formation of functional vessels within pancreatic islet grafts. Diabetes 54, 2287–2293 (2005).
Strebel, A., Harr, T., Bachmann, F., Wernli, M. & Erb, P. Green fluorescent protein as a novel tool to measure apoptosis and necrosis. Cytometry 43, 126–133 (2001).
Nikolova, G. et al. The vascular basement membrane: a niche for insulin gene expression and beta cell proliferation. Dev. Cell 10, 397–405 (2006).
Berney, T. et al. Endotoxin-mediated delayed islet graft function is associated with increased intra-islet cytokine production and islet cell apoptosis. Transplantation 71, 125–132 (2001).
Köhler, M. et al. On-line monitoring of apoptosis in insulin-secreting cells. Diabetes 52, 2943–2950 (2003).
Acknowledgements
This study was supported by grant DK-58508 and DK-075487 (to A.C.) from the US National Institutes of Health, Juvenile Diabetes Research Foundation International grant 3-2007-73 (to S.S.) and 4-2004-361, the Swedish Research Council, the Novo Nordisk Foundation, Karolinska Institutet, the Swedish Diabetes Association, The Family Knut and Alice Wallberg Foundation, Eurodia (FP6-518153), European Foundation for the Study of Diabetes, the European Foundation for the Study of Diabetes–Lilly Research Program, Berth von Kantzow's Foundation, the Family Erling-Persson Foundation and the Diabetes Research Institute Foundation.
Author information
Authors and Affiliations
Contributions
S.S., D.N. and A.C. developed the experimental transplantation platform. S.S., D.N., O.C., J.Y., R.D.M., A.P., T.M., M.K., B.L. and A.C. did the experiments. J.W. was responsible for generating the transgenic mice. C.R. was involved in designing the transplantation protocols and writing the manuscript. S.S., D.N., I.B.L. and P.-O.B. were responsible for designing the overall experimental plan and writing the manuscript. P.-O.B. was the originator of the idea of using the anterior chamber of the eye for noninvasive in vivo imaging of pancreatic islet cell biology.
Corresponding authors
Supplementary information
Supplementary Text and Figures
Supplementary Figure 1 and Supplementary Methods (PDF 103 kb)
Supplementary Video 1
Visualization of beta cells and blood vessels in engrafted islets in the anterior chamber of the eye. The movie shows a representative animated 3D reconstruction of the vascularization of an engrafted RIP-GFP islet in the anterior chamber of the eye. Intravenously applied Texas Red fluorescence (red) and GFP fluorescence of beta-cells (green) are observed. (MOV 2224 kb)
Supplementary Video 2
Ca2+ dye loading of islet grafts in the anterior chamber of the eye. The movie shows a representative animated three-dimensional reconstruction of two pancreatic islets engrafted in the anterior chamber of the eye. The reflection image of the islets is displayed in gray scale. Fura-Red and Fluo-4 loading of the islets in red and green, respectively. (MOV 2083 kb)
Supplementary Video 3
In vivo recording of cytoplasmic free Ca2+ concentration changes. The movie shows a representative experiment with cells of a pancreatic islet engrafted in the anterior chamber of the eye loaded with Fluo-4 (green) and Fura-Red (red). Systemic stimulation with 1 mg/kg glibenclamide is applied at 3 min. (MOV 2010 kb)
Rights and permissions
About this article
Cite this article
Speier, S., Nyqvist, D., Cabrera, O. et al. Noninvasive in vivo imaging of pancreatic islet cell biology. Nat Med 14, 574–578 (2008). https://doi.org/10.1038/nm1701
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1701
This article is cited by
-
Quantitative 3D OPT and LSFM datasets of pancreata from mice with streptozotocin-induced diabetes
Scientific Data (2022)
-
Unravelling innervation of pancreatic islets
Diabetologia (2022)
-
Intravital imaging of islet Ca2+ dynamics reveals enhanced β cell connectivity after bariatric surgery in mice
Nature Communications (2021)
-
Improved in vivo imaging method for individual islets across the mouse pancreas reveals a heterogeneous insulin secretion response to glucose
Scientific Reports (2021)
-
Topologically selective islet vulnerability and self-sustained downregulation of markers for β-cell maturity in streptozotocin-induced diabetes
Communications Biology (2020)