Abstract
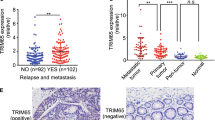
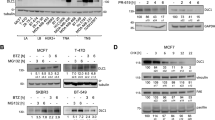
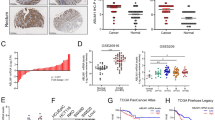
Metastasis is the primary cause of mortality from cancer, but the mechanisms leading to metastasis are poorly understood. In particular, relatively little is known about metastasis in cancers of mesenchymal origins, which are known as sarcomas. Approximately ten proteins have been characterized as 'metastasis suppressors', but how these proteins function and are regulated is, in general, not well understood. Gp78 (also known as AMFR or RNF45) is a RING finger E3 ubiquitin ligase that is integral to the endoplasmic reticulum (ER) and involved in ER-associated degradation (ERAD) of diverse substrates1,2,3,4,5,6. Here we report that expression of gp78 has a causal role in the metastasis of an aggressive human sarcoma and that this prometastatic activity requires the E3 activity of gp78. Further, gp78 associates with and targets the transmembrane metastasis suppressor, KAI1 (also known as CD82), for degradation. Suppression of gp78 increases KAI1 abundance and reduces the metastatic potential of tumor cells, an effect that is largely blocked by concomitant suppression of KAI1. An inverse relationship between these proteins was confirmed in a human sarcoma tissue microarray. Whereas most previous efforts have focused on genetic mechanisms for the loss of metastasis suppressor genes, our results provide new evidence for post-translational downregulation of a metastasis suppressor by its ubiquitin ligase, resulting in abrogation of its metastasis-suppressing effects.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Chen, B. et al. The activity of a human endoplasmic reticulum-associated degradation E3, gp78, requires its Cue domain, RING finger, and an E2-binding site. Proc. Natl. Acad. Sci. USA 103, 341–346 (2006).
Fang, S. et al. The tumor autocrine motility factor receptor, gp78, is a ubiquitin protein ligase implicated in degradation from the endoplasmic reticulum. Proc. Natl. Acad. Sci. USA 98, 14422–14427 (2001).
Lee, J.N., Song, B., DeBose-Boyd, R.A. & Ye, J. Sterol-regulated degradation of Insig-1 mediated by the membrane-bound ubiquitin ligase gp78. J. Biol. Chem. 281, 39308–39315 (2006).
Song, B.L., Sever, N. & DeBose-Boyd, R.A. Gp78, a membrane-anchored ubiquitin ligase, associates with Insig-1 and couples sterol-regulated ubiquitination to degradation of HMG CoA reductase. Mol. Cell 19, 829–840 (2005).
Shen, Y., Ballar, P. & Fang, S. Ubiquitin ligase gp78 increases solubility and facilitates degradation of the Z variant of α-1-antitrypsin. Biochem. Biophys. Res. Commun. 349, 1285–1293 (2006).
Liang, J.S. et al. Overexpression of the tumor autocrine motility factor receptor Gp78, a ubiquitin protein ligase, results in increased ubiquitinylation and decreased secretion of apolipoprotein B100 in HepG2 cells. J. Biol. Chem. 278, 23984–23988 (2003).
Shimizu, K. et al. The autocrine motility factor receptor gene encodes a novel type of seven transmembrane protein. FEBS Lett. 456, 295–300 (1999).
Watanabe, H. et al. Purification of human tumor cell autocrine motility factor and molecular cloning of its receptor. J. Biol. Chem. 266, 13442–13448 (1991).
Nabi, I.R., Watanabe, H. & Raz, A. Identification of B16–F1 melanoma autocrine motility-like factor receptor. Cancer Res. 50, 409–414 (1990).
Zhong, X. et al. AAA ATPase p97/valosin-containing protein interacts with gp78, a ubiquitin ligase for endoplasmic reticulum-associated degradation. J. Biol. Chem. 279, 45676–45684 (2004).
Kauffman, E.C., Robinson, V.L., Stadler, W.M., Sokoloff, M.H. & Rinker-Schaeffer, C.W. Metastasis suppression: the evolving role of metastasis suppressor genes for regulating cancer cell growth at the secondary site. J. Urol. 169, 1122–1133 (2003).
Khanna, C. et al. The membrane-cytoskeleton linker ezrin is necessary for osteosarcoma metastasis. Nat. Med. 10, 182–186 (2004).
Tonoli, H. & Barrett, J.C. CD82 metastasis suppressor gene: a potential target for new therapeutics? Trends Mol. Med. 11, 563–570 (2005).
Bass, R. et al. Regulation of urokinase receptor proteolytic function by the tetraspanin CD82. J. Biol. Chem. 280, 14811–14818 (2005).
Odintsova, E., Voortman, J., Gilbert, E. & Berditchevski, F. Tetraspanin CD82 regulates compartmentalisation and ligand-induced dimerization of EGFR. J. Cell Sci. 116, 4557–4566 (2003).
Ono, M., Handa, K., Withers, D.A. & Hakomori, S. Motility inhibition and apoptosis are induced by metastasis-suppressing gene product CD82 and its analogue CD9, with concurrent glycosylation. Cancer Res. 59, 2335–2339 (1999).
Schoenfeld, N., Bauer, M.K. & Grimm, S. The metastasis suppressor gene C33/CD82/KAI1 induces apoptosis through reactive oxygen intermediates. FASEB J. 18, 158–160 (2004).
Bandyopadhyay, S. et al. Interaction of KAI1 on tumor cells with DARC on vascular endothelium leads to metastasis suppression. Nat. Med. 12, 933–938 (2006).
Baek, S.H. et al. Exchange of N-CoR corepressor and Tip60 coactivator complexes links gene expression by NF-κB and β-amyloid precursor protein. Cell 110, 55–67 (2002).
Duriez, C., Falette, N., Cortes, U., Moyret-Lalle, C. & Puisieux, A. Absence of p53-dependent induction of the metastatic suppressor KAI1 gene after DNA damage. Oncogene 19, 2461–2464 (2000).
Kim, J.H. et al. Roles of sumoylation of a reptin chromatin-remodelling complex in cancer metastasis. Nat. Cell Biol. 8, 631–639 (2006).
Kim, J.H. et al. Transcriptional regulation of a metastasis suppressor gene by Tip60 and β-catenin complexes. Nature 434, 921–926 (2005).
Marreiros, A. et al. KAI1 promoter activity is dependent on p53, junB and AP2: evidence for a possible mechanism underlying loss of KAI1 expression in cancer cells. Oncogene 24, 637–649 (2005).
Berger, J.C., Vander Griend, D.J., Robinson, V.L., Hickson, J.A. & Rinker-Schaeffer, C.W. Metastasis suppressor genes: from gene identification to protein function and regulation. Cancer Biol. Ther. 4, 805–812 (2005).
Houle, C.D. et al. Loss of expression and altered localization of KAI1 and CD9 protein are associated with epithelial ovarian cancer progression. Gynecol. Oncol. 86, 69–78 (2002).
Hammond, C. et al. The tetraspan protein CD82 is a resident of MHC class II compartments where it associates with HLA-DR, -DM, and -DO molecules. J. Immunol. 161, 3282–3291 (1998).
Bruce, B. et al. Expression of the cytoskeleton linker protein ezrin in human cancers. Clin. Exp. Metastasis 24, 69–78 (2007).
Acknowledgements
We thank members of the Laboratory of Protein Dynamics and Signaling for helpful discussions, and we thank S.H. Hong for assistance in the caspase activation and imaging assays. Some experiments were performed with the help A.H. Chan and E. Sum. We also thank the following people for materials and reagents: K. Baird and P. Meltzer (NCI) for the human sarcoma tissue microarray, J.C. Barrett (Novartis) and M. Custer (NCI) for the KAI1 cDNA and antibody, P. Cresswell (Yale University) for the MaP.CD82 antibody, R. Gemmill (University of Colorado) for the Trc8 cDNA, M. Hochstrasser and L. Wang (Yale University) for the TEB4 cDNA, R. Kopito (Stanford University) for GFPu, and G. Melillo (NCI) for 786-O cells. We are indebted to members of the Tissue Array Research Program and to S. Lockett and members of the Image Analysis Laboratory for their assistance. This research is supported by the Intramural Research Program of the US National Institutes of Health, National Cancer Institute, Center for Cancer Research. Y.C.T. and A.M.W. dedicate this study to a wonderful mentor and colleague, C. Pickart, who was an inspiration for this research.
Author information
Authors and Affiliations
Contributions
Y.C.T. designed and carried out experiments, interpreted data and wrote the paper. A.M. performed animal studies. J.M.M., Z.K and B.C. developed reagents and performed experiments. M.Z. and T.V. carried out mass spectrometric analysis. S.M.H. helped to analyze the histopathology and tissue microarray data. L.J.H. helped to initiate this project and contributed to experimental design. C.K. contributed to directing the project, oversaw animal studies and wrote the paper. A.M.W. initiated and directed the project and wrote the paper.
Corresponding authors
Supplementary information
Supplementary Text and Figures
Supplementary Figs. 1–11 (PDF 451 kb)
Rights and permissions
About this article
Cite this article
Tsai, Y., Mendoza, A., Mariano, J. et al. The ubiquitin ligase gp78 promotes sarcoma metastasis by targeting KAI1 for degradation. Nat Med 13, 1504–1509 (2007). https://doi.org/10.1038/nm1686
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1686
This article is cited by
-
Unveiling the dark side of glucose-regulated protein 78 (GRP78) in cancers and other human pathology: a systematic review
Molecular Medicine (2023)
-
Estrogens drive the endoplasmic reticulum-associated degradation and promote proto-oncogene c-Myc expression in prostate cancer cells by androgen receptor/estrogen receptor signaling
Journal of Cell Communication and Signaling (2023)
-
Ubiquitination of NLRP3 by gp78/Insig-1 restrains NLRP3 inflammasome activation
Cell Death & Differentiation (2022)
-
Osteosarcoma
Nature Reviews Disease Primers (2022)
-
Basal Gp78-dependent mitophagy promotes mitochondrial health and limits mitochondrial ROS
Cellular and Molecular Life Sciences (2022)