Abstract
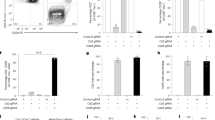
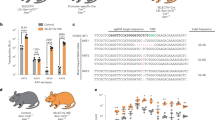
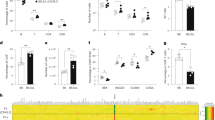
Conditional targeted cell ablation is a powerful approach for investigating the pathogenesis of human diseases and in vivo cellular functions. Intermedilysin (ILY) is a cytolytic pore-forming toxin secreted by Streptococcus intermedius that lyses human cells exclusively, owing to its receptor specificity for human CD59. We generated two transgenic mouse strains that express human CD59 either on erythrocytes (strain ThCD59RBC) or on endothelia (strain ThCD59END). Intravenous injection of ILY in ThCD59RBC mice induced acute intravascular hemolysis, leading to reduced nitric oxide bioavailability, increased platelet activation and rapid death. In ThCD59END mice, ILY induced rapid endothelial damage, leading to acute death and disseminated intravascular coagulation. Additionally, we show that human serum contains ILY-specific neutralizing antibodies not found in any other animal species. Together, these results suggest that this new rapid conditional targeted ILY-mediated cell ablation technique can be used in combination with any available transgenic expression system to study the physiologic role of specific cell populations.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Buch, T. et al. A Cre-inducible diphtheria toxin receptor mediates cell lineage ablation after toxin administration. Nat. Methods 2, 419–426 (2005).
Saito, M. et al. Diphtheria toxin receptor-mediated conditional and targeted cell ablation in transgenic mice. Nat. Biotechnol. 19, 746–750 (2001).
Luquet, S., Perez, F.A., Hnasko, T.S. & Palmiter, R.D. NPY/AgRP neurons are essential for feeding in adult mice but can be ablated in neonates. Science 310, 683–685 (2005).
Lahl, K. et al. Selective depletion of Foxp3+ regulatory T cells induces a scurfy-like disease. J. Exp. Med. 204, 57–63 (2007).
Collier, R.J. in ADP-Ribosylating Toxins and G proteins: Insights into Signal Transduction (eds. Moss, J. & Vaughan, M.) 3–19 (American Society for Microbiology, Washington, DC, 1990).
Nagamune, H. et al. Intermedilysin, a novel cytotoxin specific for human cells secreted by Streptococcus intermedius UNS46 isolated from a human liver abscess. Infect. Immun. 64, 3093–3100 (1996).
Giddings, K.S., Zhao, J., Sims, P.J. & Tweten, R.K. Human CD59 is a receptor for the cholesterol-dependent cytolysin intermedilysin. Nat. Struct. Mol. Biol. 11, 1173–1178 (2004).
Parker, M.W. & Feil, S.C. Pore-forming protein toxins: from structure to function. Prog. Biophys. Mol. Biol. 88, 91–142 (2005).
Whiley, R.A., Beighton, D., Winstanley, T.G., Fraser, H.Y. & Hardie, J.M. Streptococcus intermedius, Streptococcus constellatus, and Streptococcus anginosus (the Streptococcus milleri group): association with different body sites and clinical infections. J. Clin. Microbiol. 30, 243–244 (1992).
Sukeno, A. et al. Intermedilysin is essential for the invasion of hepatoma HepG2 cells by Streptococcus intermedius. Microbiol. Immunol. 49, 681–694 (2005).
Qin, X. et al. Deficiency of the mouse complement regulatory protein mCd59b results in spontaneous hemolytic anemia with platelet activation and progressive male infertility. Immunity 18, 217–227 (2003).
Kooyman, D.L. et al. In vivo transfer of GPI-linked complement restriction factors from erythrocytes to the endothelium. Science 269, 89–92 (1995).
Gladwin, M.T., Crawford, J.H. & Patel, R.P. The biochemistry of nitric oxide, nitrite, and hemoglobin: role in blood flow regulation. Free Radic. Biol. Med. 36, 707–717 (2004).
Loscalzo, J. Nitric oxide insufficiency, platelet activation, and arterial thrombosis. Circ. Res. 88, 756–762 (2001).
Minneci, P.C. et al. Hemolysis-associated endothelial dysfunction mediated by accelerated NO inactivation by decompartmentalized oxyhemoglobin. J. Clin. Invest. 115, 3409–3417 (2005).
van der Zee, P.M. et al. P-selectin- and CD63-exposing platelet microparticles reflect platelet activation in peripheral arterial disease and myocardial infarction. Clin. Chem. 52, 657–664 (2006).
Ravanat, C. et al. Cross-reactivity of human molecular markers for detection of prethrombotic states in various animal species. Blood Coagul. Fibrinolysis 6, 446–455 (1995).
Rother, R.P., Bell, L., Hillmen, P. & Gladwin, M.T. The clinical sequelae of intravascular hemolysis and extracellular plasma hemoglobin: a novel mechanism of human disease. J. Am. Med. Assoc. 293, 1653–1662 (2005).
Kadl, A. & Leitinger, N. The role of endothelial cells in the resolution of acute inflammation. Antioxid. Redox Signal. 7, 1744–1754 (2005).
Jancar, S. & Sanchez Crespo, M. Immune complex-mediated tissue injury: a multistep paradigm. Trends Immunol. 26, 48–55 (2005).
Homer, K.A., Grootveld, M.C., Hawkes, J., Naughton, D.P. & Beighton, D. Degradation of hyaluronate by Streptococcus intermedius strain UNS 35. J. Med. Microbiol. 41, 414–422 (1994).
Sierig, G., Cywes, C., Wessels, M.R. & Ashbaugh, C.D. Cytotoxic effects of streptolysin o and streptolysin s enhance the virulence of poorly encapsulated group a streptococci. Infect. Immun. 71, 446–455 (2003).
Polekhina, G., Giddings, K.S., Tweten, R.K. & Parker, M.W. Insights into the action of the superfamily of cholesterol-dependent cytolysins from studies of intermedilysin. Proc. Natl. Acad. Sci. USA 102, 600–605 (2005).
Danishpajooh, I.O. et al. Nitric oxide inhibits methionine synthase activity in vivo and disrupts carbon flow through the folate pathway. J. Biol. Chem. 276, 27296–27303 (2001).
Brouchet, L. et al. Estradiol accelerates reendothelialization in mouse carotid artery through estrogen receptor-alpha but not estrogen receptor-beta. Circulation 103, 423–428 (2001).
Foteinos, G., Afzal, A.R., Mandal, K., Jahangiri, M. & Xu, Q. Anti-heat shock protein 60 autoantibodies induce atherosclerosis in apolipoprotein E-deficient mice via endothelial damage. Circulation 112, 1206–1213 (2005).
Denis, C. et al. A mouse model of severe von Willebrand disease: defects in hemostasis and thrombosis. Proc. Natl. Acad. Sci. USA 95, 9524–9529 (1998).
Isermann, B. et al. Endothelium-specific loss of murine thrombomodulin disrupts the protein C anticoagulant pathway and causes juvenile-onset thrombosis. J. Clin. Invest. 108, 537–546 (2001).
Dixon, W.J. & Mood, A.M. A method for obtaining and analyzing sensitive data. J. Am. Stat. Assoc. 43, 109–126 (1948).
Acknowledgements
This work was supported by US National Institutes of Health grants RO1 DK060979 (J.A.H.) and RO1 AI061174 (X.Q.) and by a Scientist Development grant from the American Heart Association (0435483N; X.Q.). We are grateful to G.W. Byrne for providing the vector that contains the hemoglobin promoter and a locus control region, to P.J. Cowan for providing the vector that contains the ICAM-2 promoter, to M. Fan for comments, to G. Kunos for critical reading of the manuscript and to the Brigham and Women's Hospital Editorial Service for editorial assistance.
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figs. 1–3, Supplementary Table 1 and Supplementary Methods (PDF 768 kb)
Rights and permissions
About this article
Cite this article
Hu, W., Ferris, S., Tweten, R. et al. Rapid conditional targeted ablation of cells expressing human CD59 in transgenic mice by intermedilysin. Nat Med 14, 98–103 (2008). https://doi.org/10.1038/nm1674
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1674
This article is cited by
-
Distinct fate, dynamics and niches of renal macrophages of bone marrow or embryonic origins
Nature Communications (2020)
-
Versatile cell ablation tools and their applications to study loss of cell functions
Cellular and Molecular Life Sciences (2019)
-
Complement and HIV-I infection/HIV-associated neurocognitive disorders
Journal of NeuroVirology (2014)
-
Application of a novel inhibitor of human CD59 for the enhancement of complement-dependent cytolysis on cancer cells
Cellular & Molecular Immunology (2011)