Abstract
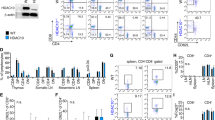
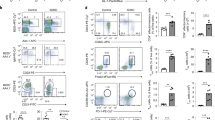
Histone/protein deacetylases (HDACs) regulate chromatin remodeling and gene expression as well as the functions of more than 50 transcription factors and nonhistone proteins. We found that administration of an HDAC inhibitor (HDACi) in vivo increased Foxp3 gene expression, as well as the production and suppressive function of regulatory T cells (Treg cells). Although Treg cells express multiple HDACs, HDAC9 proved particularly important in regulating Foxp3-dependent suppression. Optimal Treg function required acetylation of several lysines in the forkhead domain of Foxp3, and Foxp3 acetylation enhanced binding of Foxp3 to the Il2 promoter and suppressed endogenous IL-2 production. HDACi therapy in vivo enhanced Treg-mediated suppression of homeostatic proliferation, decreased inflammatory bowel disease through Treg-dependent effects, and, in conjunction with a short course of low-dose rapamycin, induced permanent, Treg-dependent cardiac and islet allograft survival and donor-specific allograft tolerance. Our data show that use of HDACi allows the beneficial pharmacologic enhancement of both the numbers and suppressive function of Foxp3+ Treg cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Lam, A.L., Pazin, D.E. & Sullivan, B.A. Control of gene expression and assembly of chromosomal subdomains by chromatin regulators with antagonistic functions. Chromosoma 114, 242–251 (2005).
Johnstone, R.W. Histone-deacetylase inhibitors: novel drugs for the treatment of cancer. Nat. Rev. Drug Discov. 1, 287–299 (2002).
McKinsey, T.A. & Olson, E.N. Cardiac histone acetylation–therapeutic opportunities abound. Trends Genet. 20, 206–213 (2004).
Matsuzaki, H. et al. Acetylation of Foxo1 alters its DNA-binding ability and sensitivity to phosphorylation. Proc. Natl. Acad. Sci. USA 102, 11278–11283 (2005).
Tian, L. et al. Reversible histone acetylation and deacetylation mediate genome-wide, promoter-dependent and locus-specific changes in gene expression during plant development. Genetics 169, 337–345 (2005).
Nusinzon, I. & Horvath, C.M. Histone deacetylases as transcriptional activators? Role reversal in inducible gene regulation. Sci. STKE 2005, re11 (2005).
Glozak, M.A., Sengupta, N., Zhang, X. & Seto, E. Acetylation and deacetylation of non-histone proteins. Gene 363, 15–23 (2005).
Gu, W. & Roeder, R.G. Activation of p53 sequence-specific DNA binding by acetylation of the p53 C-terminal domain. Cell 90, 595–606 (1997).
Reddy, P. et al. Histone deacetylase inhibitor suberoylanilide hydroxamic acid reduces acute graft-versus-host disease and preserves graft-versus-leukemia effect. Proc. Natl. Acad. Sci. USA 101, 3921–3926 (2004).
Yoshida, M., Kijima, M., Akita, M. & Beppu, T. Potent and specific inhibition of mammalian histone deacetylase both in vivo and in vitro by trichostatin A. J. Biol. Chem. 265, 17174–17179 (1990).
Bettelli, E. et al. Reciprocal developmental pathways for the generation of pathogenic effector TH17 and regulatory T cells. Nature 441, 235–238 (2006).
Brunkow, M.E. et al. Disruption of a new forkhead/winged-helix protein, scurfin, results in the fatal lymphoproliferative disorder of the scurfy mouse. Nat. Genet. 27, 68–73 (2001).
Wu, Y. et al. FOXP3 controls regulatory T cell function through cooperation with NFAT. Cell 126, 375–387 (2006).
Ono, M. et al. Foxp3 controls regulatory T-cell function by interacting with AML1/Runx1. Nature 446, 685–689 (2007).
Melgar, S., Karlsson, A. & Michaelsson, E. Acute colitis induced by dextran sulfate sodium progresses to chronicity in C57BL/6 but not in BALB/c mice: correlation between symptoms and inflammation. Am. J. Physiol. Gastrointest. Liver Physiol. 288, G1328–G1338 (2005).
Shintani, N. et al. Involvement of CD4+ T cells in the development of dextran sulfate sodium-induced experimental colitis and suppressive effect of IgG on their action. Gen. Pharmacol. 31, 477–481 (1998).
Elson, C.O. et al. Experimental models of inflammatory bowel disease reveal innate, adaptive, and regulatory mechanisms of host dialogue with the microbiota. Immunol. Rev. 206, 260–276 (2005).
Lee, I. et al. Recruitment of Foxp3+ T regulatory cells mediating allograft tolerance depends on the CCR4 chemokine receptor. J. Exp. Med. 201, 1037–1044 (2005).
Hricik, D.E. et al. Enzyme linked immunosorbent spot (ELISPOT) assay for interferon-gamma independently predicts renal function in kidney transplant recipients. Am. J. Transplant. 3, 878–884 (2003).
Choi, J.H. et al. Trichostatin A attenuates airway inflammation in mouse asthma model. Clin. Exp. Allergy 35, 89–96 (2005).
Moreira, J.M., Scheipers, P. & Sorensen, P. The histone deacetylase inhibitor Trichostatin A modulates CD4+ T cell responses. BMC Cancer 3, 30 (2003).
Bennett, C.L. et al. The immune dysregulation, polyendocrinopathy, enteropathy, X-linked syndrome (IPEX) is caused by mutations of FOXP3. Nat. Genet. 27, 20–21 (2001).
Hori, S., Nomura, T. & Sakaguchi, S. Control of regulatory T cell development by the transcription factor Foxp3. Science 299, 1057–1061 (2003).
Fontenot, J.D., Gavin, M.A. & Rudensky, A.Y. Foxp3 programs the development and function of CD4+CD25+ regulatory T cells. Nat. Immunol. 4, 330–336 (2003).
Khattri, R., Cox, T., Yasayko, S.A. & Ramsdell, F. An essential role for Scurfin in CD4+CD25+ T regulatory cells. Nat. Immunol. 4, 337–342 (2003).
Fisson, S. et al. Continuous activation of autoreactive CD4+ CD25+ regulatory T cells in the steady state. J. Exp. Med. 198, 737–746 (2003).
Chen, W. et al. Conversion of peripheral CD4+CD25- naive T cells to CD4+CD25+ regulatory T cells by TGF-beta induction of transcription factor Foxp3. J. Exp. Med. 198, 1875–1886 (2003).
Liang, S. et al. Conversion of CD4+ CD25- cells into CD4+ CD25+ regulatory T cells in vivo requires B7 costimulation, but not the thymus. J. Exp. Med. 201, 127–137 (2005).
Almeida, A.R., Zaragoza, B. & Freitas, A.A. Competition controls the rate of transition between the peripheral pools of CD4+CD25- and CD4+CD25+ T cells. Int. Immunol. 18, 1607–1613 (2006).
Minucci, S. & Pelicci, P.G. Histone deacetylase inhibitors and the promise of epigenetic (and more) treatments for cancer. Nat. Rev. Cancer 6, 38–51 (2006).
Chen, C., Rowell, E.A., Thomas, R.M., Hancock, W.W. & Wells, A.D. Transcriptional regulation by Foxp3 is associated with direct promoter occupancy and modulation of histone acetylation. J. Biol. Chem. 281, 36828–36834 (2006).
Li, B. et al. FOXP3 interactions with histone acetyltransferase and class II histone deacetylases are required for repression. Proc. Natl. Acad. Sci. USA 104, 4571–4576 (2007).
Parra, M., Kasler, H., McKinsey, T.A., Olson, E.N. & Verdin, E. Protein kinase D1 phosphorylates HDAC7 and induces its nuclear export after T-cell receptor activation. J. Biol. Chem. 280, 13762–13770 (2005).
Dequiedt, F. et al. Phosphorylation of histone deacetylase 7 by protein kinase D mediates T cell receptor-induced Nur77 expression and apoptosis. J. Exp. Med. 201, 793–804 (2005).
Zhang, C.L. et al. Class II histone deacetylases act as signal-responsive repressors of cardiac hypertrophy. Cell 110, 479–488 (2002).
Glauben, R. et al. Histone hyperacetylation is associated with amelioration of experimental colitis in mice. J. Immunol. 176, 5015–5022 (2006).
Wood, K.J. & Sakaguchi, S. Regulatory T cells in transplantation tolerance. Nat. Rev. Immunol. 3, 199–210 (2003).
Waldmann, H., Adams, E., Fairchild, P. & Cobbold, S. Infectious tolerance and the long-term acceptance of transplanted tissue. Immunol. Rev. 212, 301–313 (2006).
Hancock, W.W., Buelow, R., Sayegh, M.H. & Turka, L.A. Antibody-induced transplant arteriosclerosis is prevented by graft expression of anti-oxidant and anti-apoptotic genes. Nat. Med. 4, 1392–1396 (1998).
Wang, L. et al. Permanent survival of fully MHC-mismatched islet allografts by targeting a single chemokine receptor pathway. J. Immunol. 175, 6311–6318 (2005).
Fukuzaki, T., Hancock, W.W., Monaco, A.P. & Maki, T. Indefinite survival of skin allografts in adult thymectomized, antilymphocyte serum-treated mice given bone marrow and thymus grafts of donor origin: tolerance induction by donor bone marrow and thymus. Transplantation 65, 1036–1043 (1998).
Wang, L. et al. B7–H3 promotes acute and chronic allograft rejection. Eur. J. Immunol. 35, 428–438 (2005).
Lee, I. et al. Blocking the monocyte chemoattractant protein-1/CCR2 chemokine pathway induces permanent survival of islet allografts through a programmed death-1 ligand-1-dependent mechanism. J. Immunol. 171, 6929–6935 (2003).
Workman, C.J. & Vignali, D.A. Negative regulation of T cell homeostasis by Lymphocyte Activation Gene-3 (CD223). J. Immunol. 174, 688–695 (2005).
Thornton, A.M. & Shevach, E.M. CD4+CD25+ immunoregulatory T cells suppress polyclonal T cell activation in vitro by inhibiting interleukin 2 production. J. Exp. Med. 188, 287–296 (1998).
Tao, R. et al. Differential effects of B and T lymphocyte attenuator and programmed death-1 on acceptance of partially versus fully MHC-mismatched cardiac allografts. J. Immunol. 175, 5774–5782 (2005).
Suchin, E.J. et al. Quantifying the frequency of alloreactive T cells in vivo: new answers to an old question. J. Immunol. 166, 973–981 (2001).
Wysocka, J., Reilly, P.T. & Herr, W. Loss of HCF-1-chromatin association precedes temperature-induced growth arrest of tsBN67 cells. Mol. Cell. Biol. 21, 3820–3829 (2001).
Zeng, L. et al. HDAC3 is crucial in shear- and VEGF-induced stem cell differentiation toward endothelial cells. J. Cell Biol. 174, 1059–1069 (2006).
Pear, W.S., Nolan, G.P., Scott, M.L. & Baltimore, D. Production of high-titer helper-free retroviruses by transient transfection. Proc. Natl. Acad. Sci. USA 90, 8392–8396 (1993).
Acknowledgements
This work was supported by a US National Institutes of Health grant to W.W.H. (R01 AI 54720).
Author information
Authors and Affiliations
Contributions
R.T. performed most of the transplant and in vitro studies, E.F.D.Z. performed colitis studies, E.O. performed Foxp3 mutagenesis, C.C. and A.D.W. performed ChIP studies, L.W. performed additional transplant and in vitro experiments, P.M.P. and L.A.T. performed peripheral conversion studies with Fox-GFP transgenic mice, B.L., M.I.G. and E.N.O. were instrumental in early stages of the project, and W.W.H. conceived, designed and directed the study and wrote the manuscript.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figs. 1–7 (PDF 594 kb)
Rights and permissions
About this article
Cite this article
Tao, R., de Zoeten, E., Özkaynak, E. et al. Deacetylase inhibition promotes the generation and function of regulatory T cells. Nat Med 13, 1299–1307 (2007). https://doi.org/10.1038/nm1652
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1652
This article is cited by
-
The regulation and differentiation of regulatory T cells and their dysfunction in autoimmune diseases
Nature Reviews Immunology (2024)
-
A CIC-related-epigenetic factors-based model associated with prediction, the tumor microenvironment and drug sensitivity in osteosarcoma
Scientific Reports (2024)
-
Epigenetic regulation in the tumor microenvironment: molecular mechanisms and therapeutic targets
Signal Transduction and Targeted Therapy (2023)
-
Regulatory T cells in the face of the intestinal microbiota
Nature Reviews Immunology (2023)
-
Gastrointestinal Tract, Microbiota and Multiple Sclerosis (MS) and the Link Between Gut Microbiota and CNS
Current Microbiology (2023)