Abstract
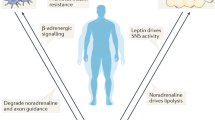
The relationship between stress and obesity remains elusive. In response to stress, some people lose weight, whereas others gain. Here we report that stress exaggerates diet-induced obesity through a peripheral mechanism in the abdominal white adipose tissue that is mediated by neuropeptide Y (NPY). Stressors such as exposure to cold or aggression lead to the release of NPY from sympathetic nerves, which in turn upregulates NPY and its Y2 receptors (NPY2R) in a glucocorticoid-dependent manner in the abdominal fat. This positive feedback response by NPY leads to the growth of abdominal fat. Release of NPY and activation of NPY2R stimulates fat angiogenesis, macrophage infiltration, and the proliferation and differentiation of new adipocytes, resulting in abdominal obesity and a metabolic syndrome-like condition. NPY, like stress, stimulates mouse and human fat growth, whereas pharmacological inhibition or fat-targeted knockdown of NPY2R is anti-angiogenic and anti-adipogenic, while reducing abdominal obesity and metabolic abnormalities. Thus, manipulations of NPY2R activity within fat tissue offer new ways to remodel fat and treat obesity and metabolic syndrome.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Change history
24 July 2007
Nat. Med. 13, 803-811 (2007); published online 1 July; corrected after print 24 July 2007. The version of this article initially published contained several typographical errors affecting figure citations, units of measure and figure legends, none of which change the scientific conclusions of the manuscript in any way. In addition, the authors incorrectly stated that they had no competing financial interests. A proper description of these competing interests, as is required by journal policy, has now been attached to the HTML version of the article, and the typographical errors have been corrected in the HTML and PDF versions.
References
Mikurube, H. et al. Association of change in the type of job with prevalence of components of the metabolic syndrome—special reference to job stress. Nippon Koshu Eisei Zasshi 52, 987–993 (2005).
Bowers, R.R. et al. Sympathetic innervation of white adipose tissue and its regulation of fat cell number. Am. J. Physiol. Regul. Integr. Comp. Physiol. 286, R1167–R1175 (2004).
Bachman, E.S. et al. βAR signaling required for diet-induced thermogenesis and obesity resistance. Science 297, 843–845 (2002).
Turtzo, L.C. & Lane, M.D. Completing the loop: neuron-adipocyte interactions and the control of energy homeostasis. Horm. Metab. Res. 34, 607–615 (2002).
Kalra, S.P. & Kalra, P.S. NPY and cohorts in regulating appetite, obesity and metabolic syndrome: beneficial effects of gene therapy. Neuropeptides 38, 201–211 (2004).
Mark, A.L., Correia, M.L., Rahmouni, K. & Haynes, W.G. Selective leptin resistance: a new concept in leptin physiology with cardiovascular implications. J. Hypertens. 20, 1245–1250 (2002).
Sainsbury, A., Schwarzer, C., Couzens, M. & Herzog, H. Y2 receptor deletion attenuates the type 2 diabetic syndrome of ob/ob mice. Diabetes 51, 3420–3427 (2002).
Bradley, R.L., Mansfield, J.P. & Maratos-Flier, E. Neuropeptides, including neuropeptide Y and melanocortins, mediate lipolysis in murine adipocytes. Obes. Res. 13, 653–661 (2005).
Lee, E.W. et al. Neuropeptide Y induces ischemic angiogenesis and restores function of ischemic skeletal muscles. J. Clin. Invest. 111, 1853–1862 (2003).
Ghersi, G., Chen, W., Lee, E.W. & Zukowska, Z. Critical role of dipeptidyl peptidase IV in neuropeptide Y-mediated endothelial cell migration in response to wounding. Peptides 22, 453–458 (2001).
Zukowska, Z., Grant, D.S. & Lee, E.W. Neuropeptide Y: a novel mechanism for ischemic angiogenesis. Trends Cardiovasc. Med. 13, 86–92 (2003).
Koulu, M. et al. Neuropeptide Y and Y2-receptor are involved in development of diabetic retinopathy and retinal neovascularization. Ann. Med. 36, 232–240 (2004).
Kitlinska, J. et al. Differential effects of neuropeptide Y on the growth and vascularization of neural crest-derived tumors. Cancer Res. 65, 1719–1728 (2005).
Zukowska-Grojec, Z., Marks, E.S. & Haass, M. Neuropeptide Y is a potent vasoconstrictor and a cardiodepressant in rat. Am. J. Physiol. 253, H1234–H1239 (1987).
Li, L., Lee, E.W., Ji, H. & Zukowska, Z. Neuropeptide Y-induced acceleration of postangioplasty occlusion of rat carotid artery. Arterioscler. Thromb. Vasc. Biol. 23, 1204–1210 (2003).
Zukowska-Grojec, Z. & Neuropeptide, Y. A novel sympathetic stress hormone and more. Ann. NY Acad. Sci. 771, 219–233 (1995).
Zukowska-Grojec, Z. & Vaz, A.C. Role of neuropeptide Y (NPY) in cardiovascular responses to stress. Synapse 2, 293–298 (1988).
Morris, M.J. et al. Increases in plasma neuropeptide Y concentrations during sympathetic activation in man. J. Auton. Nerv. Syst. 17, 143–149 (1986).
Zukowska-Grojec, Z., Konarska, M. & McCarty, R. Differential plasma catecholamine and neuropeptide Y responses to acute stress in rats. Life Sci. 42, 1615–1624 (1988).
Dallman, M.F. et al. Chronic stress and obesity: a new view of “comfort food”. Proc. Natl. Acad. Sci. USA 100, 11696–11701 (2003).
Bray, G.A. The nutrient balance hypothesis: peptides, sympathetic activity, and food intake. Ann. NY Acad. Sci. 676, 223–241 (1993).
Yamada, T. et al. Signals from intra-abdominal fat modulate insulin and leptin sensitivity through different mechanisms: neuronal involvement in food-intake regulation. Cell Metab. 3, 223–229 (2006).
Bujalska, I.J., Walker, E.A., Tomlinson, J.W., Hewison, M. & Stewart, P.M. 11β-hydroxysteroid dehydrogenase type 1 in differentiating omental human preadipocytes: from de-activation to generation of cortisol. Endocr. Res. 28, 449–461 (2002).
Dodt, C., Lonnroth, P., Wellhoner, J.P., Fehm, H.L. & Elam, M. Sympathetic control of white adipose tissue in lean and obese humans. Acta Physiol. Scand. 177, 351–357 (2003).
Masuzaki, H. et al. Transgenic amplification of glucocorticoid action in adipose tissue causes high blood pressure in mice. J. Clin. Invest. 112, 83–90 (2003).
Jimenez, M. et al. β1/β2/β3-adrenoceptor knockout mice are obese and cold-sensitive but have normal lipolytic responses to fasting. FEBS Lett. 530, 37–40 (2002).
Troisi, R.J. et al. Relation of obesity and diet to sympathetic nervous system activity. Hypertension 17, 669–677 (1991).
Rosmond, R., Dallman, M.F. & Bjorntorp, P. Stress-related cortisol secretion in men: relationships with abdominal obesity and endocrine, metabolic and hemodynamic abnormalities. J. Clin. Endocrinol. Metab. 83, 1853–1859 (1998).
Bartness, T.J., Kay Song, C., Shi, H., Bowers, R.R. & Foster, M.T. Brain-adipose tissue cross talk. Proc. Nutr. Soc. 64, 53–64 (2005).
Hjemdahl, P. Stress and the metabolic syndrome: an interesting but enigmatic association. Circulation 106, 2634–2636 (2002).
Levenson, C.W. & Moore, J.B. Response of rat adrenal neuropeptide Y and tyrosine hydroxylase mRNA to acute stress is enhanced by long-term voluntary exercise. Neurosci. Lett. 242, 177–179 (1998).
Boswell, T. et al. NPY and galanin in a hibernator: hypothalamic gene expression and effects on feeding. Brain Res. Bull. 32, 379–384 (1993).
Kallio, J. et al. Enhanced exercise-induced GH secretion in subjects with Pro7 substitution in the prepro-NPY. J. Clin. Endocrinol. Metab. 86, 5348–5352 (2001).
Rupnick, M.A. et al. Adipose tissue mass can be regulated through the vasculature. Proc. Natl. Acad. Sci. USA 99, 10730–10735 (2002).
Brakenhielm, E. et al. Angiogenesis inhibitor, TNP-470, prevents diet-induced and genetic obesity in mice. Circ. Res. 94, 1579–1588 (2004).
Allen, J.M., Martin, J.B. & Heinrich, G. Neuropeptide Y gene expression in PC12 cells and its regulation by nerve growth factor: a model for developmental regulation. Brain Res. 427, 39–43 (1987).
Nankova, B. et al. Immobilization stress elevates gene expression for catecholamine biosynthetic enzymes and some neuropeptides in rat sympathetic ganglia: effects of adrenocorticotropin and glucocorticoids. Endocrinology 137, 5597–5604 (1996).
Li, L., Jonsson-Rylander, A.C., Abe, K. & Zukowska, Z. Chronic stress induces rapid occlusion of angioplasty-injured rat carotid artery by activating neuropeptide Y and its Y1 receptors. Arterioscler. Thromb. Vasc. Biol. 25, 2075–2080 (2005).
Schwarz, H., Villiger, P.M., von Kempis, J. & Lotz, M. Neuropeptide Y is an inducible gene in the human immune system. J. Neuroimmunol. 51, 53–61 (1994).
Prod'homme, T., Weber, M.S., Steinman, L. & Zamvil, S.S. A neuropeptide in immune-mediated inflammation, Y? Trends Immunol. 27, 164–167 (2006).
Guzik, T.J., Mangalat, D. & Korbut, R. Adipocytokines—novel link between inflammation and vascular function? J. Physiol. Pharmacol. 57, 505–528 (2006).
Suganami, T., Nishida, J. & Ogawa, Y. A paracrine loop between adipocytes and macrophages aggravates inflammatory changes: role of free fatty acids and tumor necrosis factor alpha. Arterioscler. Thromb. Vasc. Biol. 25, 2062–2068 (2005).
Steppan, C.M. et al. The hormone resistin links obesity to diabetes. Nature 409, 307–312 (2001).
Kozlowski, S. et al. Mechanism of sympathetic activation during prolonged physical exercise in dogs. The role of hepatic glucoreceptors. Pflugers Arch. 399, 63–67 (1983).
Boehm, S. & Huck, S. Receptors controlling transmitter release from sympathetic neurons in vitro. Prog. Neurobiol. 51, 225–242 (1997).
Lavebratt, C., Alpman, A., Persson, B., Arner, P. & Hoffstedt, J. Common neuropeptide Y2 receptor gene variant is protective against obesity among Swedish men. Int. J. Obes. 30, 453–459 (2006).
van Rossum, C.T., Pijl, H., Adan, R.A., Hoebee, B. & Seidell, J.C. Polymorphisms in the NPY and AGRP genes and body fatness in Dutch adults. Int. J. Obes. 30, 1522–1528 (2006).
Rotunda, A.M. & Kolodney, M.S. Mesotherapy and phosphatidylcholine injections: historical clarification and review. Dermatol. Surg. 32, 465–480 (2006).
Naveilhan, P., Svensson, L., Nystrom, S., Ekstrand, A.J. & Ernfors, P. Attenuation of hypercholesterolemia and hyperglycemia in ob/ob mice by NPY Y2 receptor ablation. Peptides 23, 1087–1091 (2002).
Acknowledgements
We thank J.F. Mill, A.K. Myers and P.C. Fox for editorial comments, M. Czarnecka for assisting with graphical aspects of the figures, A.-M. Hageny for assistance with catecholamine assays, M.D. Lane (Johns Hopkins) for 3T3-L1 preadipocytes, W. Rasband for NIH ImageJ and plug-ins, M. Abramoff for VolumeJ plug-in, and F. Bunz (Johns Hopkins) for pAdTrack-Cre. This work was supported by US National Institutes of Health (NIH) grants HL067357 and HL055310 to Z.Z., NIH grant DE016050 and PSEF National Endowment Grant to S.B.B., a Predoctoral Mid-Atlantic Fellowship from the American Heart Association to L.K., and Slovak Research and Development Agency grant APVV0148-06 to R.K.
Author information
Authors and Affiliations
Contributions
L.E.K. carried out most of the experiments (as her PhD thesis work); developed stress models; established adipocyte-endothelial-neuronal co-cultures; together with S.T.F., adapted the MRI technique to the assessment of fat volumes; prepared most of the figures; and wrote major parts of the manuscript. J.U.T. contributed to many experiments using stress models, and assisted with harvesting tissues and adenoviral vectors. J.B.K. designed primers for RT-PCR; carried out molecular analyses; assisted with adipocyte-endothelial-neuronal co-cultures; and made major contributions to experimental design, data analyses, interpretation and presentation, and manuscript writing. L.L. performed NPY ELISA measurements, developed and carried out immunocytochemistry protocols, and contributed to data analyses and interpretation. S.B.B. provided human fat tissue derived from plastic surgeries that he had performed, together with M.D.J.; established a xenograft model of human fat growth in nude mice; and made major conceptual contribution to the clinical significance of the study for fat grafting and remodeling. M.D.J. contributed to all aspects dealing with human fat, and was instrumental in the adenoviral work by providing the vectors, measuring viral titers and training L.E.K. in experimental techniques. E.W.L. started the project and carried out experiments on genetically obese B6.V-Lepob/J mice. M.S.B. carried out resistin analyses and contributed to discussions of stress effects on inflammation and metabolic syndrome. H.H. developed and provided the Npy−/− and Npy2rlox/lox mice for the study, trained J.U.T. in preparing adenoviral vectors, and contributed to discussion of the results. S.T.F. developed the MRI protocol for analyzing fat and contributed to discussion of the results. R.K. supervised catecholamine assays and contributed to analyses, interpretation and discussion of the data dealing with glucocorticoids and adrenergic system. Z.Z. developed the idea for and supervised the study, designed protocols, developed collaborations and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
Z.Z., L.E.K., S.B.B., M.D.J. and E.W.L. are listed as the inventors on the patent application “Compositions and Methods for Lipomodeling”, PCT/US2006/021873, filed 14 December 2006 by Georgetown University and based on the work described in this paper.
Supplementary information
Supplementary Fig. 1
Npy and Npy2r expression and actions in adipocytes and endothelial cells in vitro and in vivo. (PDF 428 kb)
Supplementary Fig. 2
Metabolic and neurohormonal effects of chronic stress, HFS and intra-fat Npy2r inhibition or deletion. (PDF 378 kb)
Rights and permissions
About this article
Cite this article
Kuo, L., Kitlinska, J., Tilan, J. et al. Neuropeptide Y acts directly in the periphery on fat tissue and mediates stress-induced obesity and metabolic syndrome. Nat Med 13, 803–811 (2007). https://doi.org/10.1038/nm1611
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1611
This article is cited by
-
Neuropeptide Y directly reduced apoptosis of granulosa cells, and the expression of NPY and its receptors in PCOS subjects
Journal of Ovarian Research (2023)
-
The evolving functions of the vasculature in regulating adipose tissue biology in health and obesity
Nature Reviews Endocrinology (2023)
-
NPY1R exerts inhibitory action on estradiol-stimulated growth and predicts endocrine sensitivity and better survival in ER-positive breast cancer
Scientific Reports (2022)
-
Nutrigenetic variants and response to diet/lifestyle intervention in obese subjects: a pilot study
Acta Diabetologica (2022)
-
Activation of Neuropeptide Y2 Receptor Can Inhibit Global Cerebral Ischemia-Induced Brain Injury
NeuroMolecular Medicine (2022)