Abstract
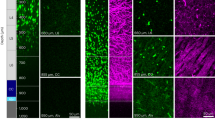
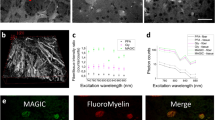
Here we describe a technique for measuring changes in Ca2+ in the cytosolic domain of mature compact myelin of live axons in the central nervous system (CNS). We label the myelin sheath of optic nerve and dorsal column axons by using the Ca2+ indicator X-rhod-1 coupled with DiOC6(3) to produce bright myelin counterstaining, thereby providing unambiguous identification of the myelin sheath for analysis of two-photon excited fluorescence. We present evidence for localization of the Ca2+ reporter to the cytosolic domain of myelin, obtained by using fluorescence lifetime, spectral measurements and Mn2+ quenching. Chemical ischemia increased myelinic X-rhod-1 fluorescence (∼50% after 30 min) in a manner dependent on extracellular Ca2+. Inhibiting Na+-dependent glutamate transporters (with TBOA) or glycine transporters (with sarcosine and ALX-1393) reduced the ischemia-induced increase in Ca2+. We show that myelinic N-methyl-D-aspartate (NMDA) receptors are activated by the two conventional coagonists glutamate and glycine, which are released by specific transporters under conditions of cellular Na+ loading and depolarization in injured white matter. This new technique facilitates detailed studies of living myelin, a vital component of the mammalian CNS.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Ranvier, L. & Weber, E. Leçons sur l'Histologie du Système Nerveux (Librairie F. Savy, Paris, 1878).
Virchow, R.L.K., Vogel, J. & Stiebel, S.F. Allgemeine Störungen der Ernährung und des Blutes (Erlangen, 1854).
Lazzarini, R.A. et al. (eds.) Myelin Biology and Disorders (Elsevier Academic, Amsterdam, 2004).
Taylor, C.M. et al. Proteomic mapping provides powerful insights into functional myelin biology. Proc. Natl. Acad. Sci. USA 101, 4643–4648 (2004).
Michailov, G.V. et al. Axonal neuregulin-1 regulates myelin sheath thickness. Science 304, 700–703 (2004).
Schlaepfer, W.W. Vesicular disruption of myelin simulated by exposure of nerve to calcium ionophore. Nature 265, 734–736 (1977).
Ellisman, M.H., Friedman, P.L. & Hamilton, W.J. Cytochemical localization of cations in myelinated nerve using TEM, HVEM, SEM and electron probe microanalysis. Scan. Electron Microsc. 1979, 793–800 (1979).
Micu, I. et al. NMDA receptors mediate calcium accumulation in central nervous system myelin during chemical ischaemia. Nature 439, 988–992 (2006).
Ness, J.K., Valentino, M., McIver, S.R. & Goldberg, M.P. Identification of oligodendrocytes in experimental disease models. Glia 50, 321–328 (2005).
Minta, A., Kao, J.P. & Tsien, R.Y. Fluorescent indicators for cytosolic calcium based on rhodamine and fluorescein chromophores. J. Biol. Chem. 264, 8171–8178 (1989).
Lakowicz, J.R. Principles of Fluorescence Spectroscopy (Plenum, New York, 1999).
Berridge, M.J., Lipp, P. & Bootman, M.D. The versatility and universality of calcium signalling. Nat. Rev. Mol. Cell Biol. 1, 11–21 (2000).
Dingledine, R., Borges, K., Bowie, D. & Traynelis, S.F. The glutamate receptor ion channels. Pharmacol. Rev. 51, 7–61 (1999).
Danbolt, N.C. Glutamate uptake. Prog. Neurobiol. 65, 1–105 (2001).
Aragon, C. & Lopez-Corcuera, B. Structure, function and regulation of glycine neurotransporters. Eur. J. Pharmacol. 479, 249–262 (2003).
Stys, P.K. White matter injury mechanisms. Curr. Mol. Med. 4, 113–130 (2004).
Alexander, S.P.H., Mathie, A. & Peters, J.A. Cell-surface transmitter transporters: glycine. Br. J. Pharmacol. 147, S148–S149 (2006).
Arundine, M. & Tymianski, M. Molecular mechanisms of calcium-dependent neurodegeneration in excitotoxicity. Cell Calcium 34, 325–337 (2003).
Mrsulja, B.J., Zalewski, A.A. & Coping, G. Ultracytochemical localization of ouabain-sensitive K+-dependent, p-nitrophenyl phosphatase in myelin. Brain Res. 343, 154–158 (1985).
Martenson, R.E. Myelin—Biology and Chemistry (CRC Press, Boca Raton, Florida, USA, 1992).
Cherksey, B., Durrie, R., Braun, P.E. & Sapirstein, V.S. In vitro analysis of ion channels in periaxolemmal-myelin and white matter clathrin coated vesicles: modulation by calcium and GTP gamma S. Neurochem. Res. 19, 1101–1106 (1994).
Ropte, S., Scheidt, P. & Friede, R.L. The intermediate dense line of the myelin sheath is preferentially accessible to cations and is stabilized by cations. J. Neurocytol. 19, 242–252 (1990).
Lev-Ram, V. & Ellisman, M.H. Axonal activation-induced calcium transients in myelinating Schwann cells, sources, and mechanisms. J. Neurosci. 15, 2628–2637 (1995).
Hildebrand, C. & Hahn, R. Relation between myelin sheath thickness and axon size in spinal cord white matter of some vertebrate species. J. Neurol. Sci. 38, 421–434 (1978).
Simpson, P.B., Challiss, R.A. & Nahorski, S.R. Divalent cation entry in cultured rat cerebellar granule cells measured using Mn2+ quench of fura 2 fluorescence. Eur. J. Neurosci. 7, 831–840 (1995).
Li, S., Mealing, G.A., Morley, P. & Stys, P.K. Novel injury mechanism in anoxia and trauma of spinal cord white matter: glutamate release via reverse Na+-dependent glutamate transport. J. Neurosci. 19, RC16 (1999).
Spike, R.C., Watt, C., Zafra, F. & Todd, A.J. An ultrastructural study of the glycine transporter GLYT2 and its association with glycine in the superficial laminae of the rat spinal dorsal horn. Neuroscience 77, 543–551 (1997).
Ren, Y., Ridsdale, A., Coderre, E. & Stys, P.K. Calcium imaging in live rat optic nerve myelinated axons in vitro using confocal laser microscopy. J. Neurosci. Methods 102, 165–176 (2000).
Verbny, Y., Zhang, C.L. & Chiu, S.Y. Coupling of calcium homeostasis to axonal sodium in axons of mouse optic nerve. J. Neurophysiol. 88, 802–816 (2002).
Ouardouz, M. et al. Depolarization-induced Ca2+ release in ischemic spinal cord white matter involves L-type Ca2+ channel activation of ryanodine receptors. Neuron 40, 53–63 (2003).
Acknowledgements
We thank J. Wang and C. Morris for providing HEK293 cells for fluorescence lifetime and spectral measurements. This work was supported by grants from the National Institute of Neurological Disorders and Stroke (NINDS), the Canadian Institutes of Health Research (CIHR), the Canadian Institute for Photonic Innovations, and the Intramural Research Program of the National Institutes of Health, NINDS (operating); CIHR 90408 and the Heart and Stroke Foundation Center for Stroke Recovery (equipment); and the generosity of private donors. P.K.S. is supported by a Heart and Stroke Foundation of Ontario Career Investigator Award.
Author information
Authors and Affiliations
Contributions
I.M., experimental design, two-photon calcium imaging, fluorescence lifetime measurements, numerical data analysis, in-place immunohistochemistry, confocal microscopy, and a major role in the writing of the article. A.R. and L.Z., two-photon microscopy, fluorescence lifetime measurements and associated numerical data analysis, and editing and revision of the article. J.W. and J.M., electron microscopy and image interpretation. C.A.B. and S.B.A., electron probe microanalysis, analysis of data and scientific discussion. P.K.S., experimental concept and design, coordination of experiments, data analysis and interpretation, and writing of the article. All authors were involved with the analysis and interpretation of their respective data and contributed to the writing of the article.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
In order to exclude the remote possibility that the failure of Mn2+ to quench X-rhod-1 fluorescence in resting, non-ischemic myelin was due to the inability of this cation to permeate uninjured myelin altogether, electron probe microanalysis was carried out on individual myelin sheaths from control optic nerves and those exposed to Mn2+-containing CSF (without ischemia, but both sets pre-treated with the same solutions normally used for dye loading). (PDF 326 kb)
Supplementary Fig. 2
X-rhod-1 emission spectrum in various micro-environments. (PDF 92 kb)
Supplementary Fig. 3
Effects of glutamate or glycine transport inhibition on myelinic and oligodendroglial Ca2+ levels in uninjured control optic nerves and dorsal columns. (PDF 110 kb)
Supplementary Fig. 4
Comparison of myelin integrity by electron microscopy in optic nerves maintained in normal Ca2+-CSF vs. nerves exposed to Ca2+-free loading solution indicated that the low Ca2+ exposure did not alter myelin structure to any appreciable degree. (PDF 782 kb)
Rights and permissions
About this article
Cite this article
Micu, I., Ridsdale, A., Zhang, L. et al. Real-time measurement of free Ca2+ changes in CNS myelin by two-photon microscopy. Nat Med 13, 874–879 (2007). https://doi.org/10.1038/nm1568
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1568
This article is cited by
-
Copper ions, prion protein and Aβ modulate Ca levels in central nervous system myelin in an NMDA receptor-dependent manner
Molecular Brain (2022)
-
Evaluation of spinal cord protective threshold of serum memantine, an NMDA receptor antagonist, in a rabbit model of paraplegia
Indian Journal of Thoracic and Cardiovascular Surgery (2020)
-
Vesicular glutamate release from central axons contributes to myelin damage
Nature Communications (2018)
-
Association between chronic stress-induced structural abnormalities in Ranvier nodes and reduced oligodendrocyte activity in major depression
Scientific Reports (2016)