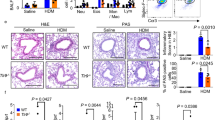
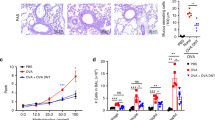
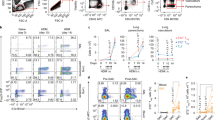
Abstract
Glucocorticoid-induced tumor necrosis factor receptor (GITR) on T cells and its natural ligand, GITRL, on accessory cells contribute to the control of immune homeostasis. Here we show that reverse signaling through GITRL after engagement by soluble GITR initiates the immunoregulatory pathway of tryptophan catabolism in mouse plasmacytoid dendritic cells, by means of noncanonical NF-κB–dependent induction of indoleamine 2,3-dioxygenase (IDO). The synthetic glucocorticoid dexamethasone administered in vivo activated IDO through the symmetric induction of GITR in CD4+ T cells and GITRL in plasmacytoid dendritic cells. The drug exerted IDO-dependent protection in a model of allergic airway inflammation. Modulation of tryptophan catabolism via the GITR-GITRL coreceptor system might represent an effective therapeutic target in immune regulation. Induction of IDO could be an important mechanism underlying the anti-inflammatory action of corticosteroids.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
van Essen, D., Kikutani, H. & Gray, D. CD40 ligand-transduced co-stimulation of T cells in the development of helper function. Nature 378, 620–623 (1995).
Eissner, G., Kolch, W. & Scheurich, P. Ligands working as receptors: reverse signaling by members of the TNF superfamily enhance the plasticity of the immune system. Cytokine Growth Factor Rev. 15, 353–366 (2004).
Grohmann, U. et al. CTLA-4–Ig regulates tryptophan catabolism in vivo. Nat. Immunol. 3, 1097–1101 (2002).
Fallarino, F. et al. Modulation of tryptophan catabolism by regulatory T cells. Nat. Immunol. 4, 1206–1212 (2003).
Orabona, C. et al. CD28 induces immunostimulatory signals in dendritic cells via CD80 and CD86. Nat. Immunol. 5, 1134–1142 (2004).
Mellor, A.L. & Munn, D.H. IDO expression by dendritic cells: tolerance and tryptophan catabolism. Nat. Rev. Immunol. 4, 762–774 (2004).
Nocentini, G. & Riccardi, C. GITR: a multifaceted regulator of immunity belonging to the tumor necrosis factor receptor superfamily. Eur. J. Immunol. 35, 1016–1022 (2005).
Shevach, E.M. & Stephens, G.L. The GITR–GITRL interaction: co-stimulation or contrasuppression of regulatory activity? Nat. Rev. Immunol. 6, 613–618 (2006).
Nocentini, G. et al. A new member of the tumor necrosis factor/nerve growth factor receptor family inhibits T cell receptor-induced apoptosis. Proc. Natl. Acad. Sci. USA 94, 6216–6221 (1997).
Tone, M. et al. Mouse glucocorticoid-induced tumor necrosis factor receptor ligand is costimulatory for T cells. Proc. Natl. Acad. Sci. USA 100, 15059–15064 (2003).
Kim, J.D. et al. Cloning and characterization of GITR ligand. Genes Immun. 4, 564–569 (2003).
Shimizu, J., Yamazaki, S., Takahashi, T., Ishida, Y. & Sakaguchi, S. Stimulation of CD25+CD4+ regulatory T cells through GITR breaks immunological self-tolerance. Nat. Immunol. 3, 135–142 (2002).
Kohm, A.P., Williams, J.S. & Miller, S.D. Cutting edge: ligation of the glucocorticoid-induced TNF receptor enhances autoreactive CD4+ T cell activation and experimental autoimmune encephalomyelitis. J. Immunol. 172, 4686–4690 (2004).
Stephens, G.L. et al. Engagement of glucocorticoid-induced TNFR family-related receptor on effector T cells by its ligand mediates resistance to suppression by CD4+CD25+ T cells. J. Immunol. 173, 5008–5020 (2004).
Ronchetti, S. et al. GITR, a member of the TNF receptor superfamily, is costimulatory to mouse T lymphocyte subpopulations. Eur. J. Immunol. 34, 613–622 (2004).
Ko, K. et al. Treatment of advanced tumors with agonistic anti-GITR mAb and its effects on tumor-infiltrating Foxp3+CD25+CD4+ regulatory T cells. J. Exp. Med. 202, 885–891 (2005).
Seo, S.K. et al. 4–1BB-mediated immunotherapy of rheumatoid arthritis. Nat. Med. 10, 1088–1094 (2004).
Asselin-Paturel, C. et al. Mouse type I IFN-producing cells are immature APCs with plasmacytoid morphology. Nat. Immunol. 2, 1144–1150 (2001).
Colonna, M., Trinchieri, G. & Liu, Y.J. Plasmacytoid dendritic cells in immunity. Nat. Immunol. 5, 1219–1226 (2004).
Ochando, J.C. et al. Alloantigen-presenting plasmacytoid dendritic cells mediate tolerance to vascularized grafts. Nat. Immunol. 7, 652–662 (2006).
Mellor, A.L. et al. Cutting edge: induced indoleamine 2,3 dioxygenase expression in dendritic cell subsets suppresses T cell clonal expansion. J. Immunol. 171, 1652–1655 (2003).
Fallarino, F. et al. Murine plasmacytoid dendritic cells initiate the immunosuppressive pathway of tryptophan catabolism in response to CD200 receptor engagement. J. Immunol. 173, 3748–3754 (2004).
Fallarino, F. & Puccetti, P. Toll-like receptor 9-mediated induction of the immunosuppressive pathway of tryptophan catabolism. Eur. J. Immunol. 36, 8–11 (2006).
Fallarino, F. et al. Ligand and cytokine dependence of the immunosuppressive pathway of tryptophan catabolism in plasmacytoid dendritic cells. Int. Immunol. 17, 1429–1438 (2005).
Muller, A.J., DuHadaway, J.B., Donover, P.S., Sutanto-Ward, E. & Prendergast, G.C. Inhibition of indoleamine 2,3-dioxygenase, an immunoregulatory target of the cancer suppression gene Bin1, potentiates cancer chemotherapy. Nat. Med. 11, 312–319 (2005).
Lawrence, T., Bebien, M., Liu, G.Y., Nizet, V. & Karin, M. IKKα limits macrophage NF-κB activation and contributes to the resolution of inflammation. Nature 434, 1138–1143 (2005).
Bonizzi, G. & Karin, M. The two NF-κB activation pathways and their role in innate and adaptive immunity. Trends Immunol. 25, 280–288 (2004).
Kinoshita, D. et al. Essential role of IκB kinase α in thymic organogenesis required for the establishment of self-tolerance. J. Immunol. 176, 3995–4002 (2006).
Chen, X., Murakami, T., Oppenheim, J.J. & Howard, O.M. Differential response of murine CD4+CD25+ and CD4+CD25− T cells to dexamethasone-induced cell death. Eur. J. Immunol. 34, 859–869 (2004).
Romani, L. Immunity to fungal infections. Nat. Rev. Immunol. 4, 1–23 (2004).
Judson, M.A. & Stevens, D.A. Current pharmacotherapy of allergic bronchopulmonary aspergillosis. Expert Opin. Pharmacother. 2, 1065–1071 (2001).
Montagnoli, C. et al. Immunity and tolerance to Aspergillus involve functionally distinct regulatory T cells and tryptophan catabolism. J. Immunol. 176, 1712–1723 (2006).
Romani, L. et al. Thymosin α1 activates dendritic cell tryptophan catabolism and establishes a regulatory environment for balance of inflammation and tolerance. Blood 108, 2265–2274 (2006).
Romani, L. & Puccetti, P. Protective tolerance to fungi: the role of IL-10 and tryptophan catabolism. Trends Microbiol. 14, 183–189 (2006).
Bluestone, J.A. & Tang, Q. How do CD4+CD25+ regulatory T cells control autoimmunity? Curr. Opin. Immunol. 17, 638–642 (2005).
Grohmann, U., Fallarino, F. & Puccetti, P. Tolerance, DCs and tryptophan: much ado about IDO. Trends Immunol. 24, 242–248 (2003).
Orabona, C. et al. Towards the identification of a tolerogenic signature in IDO-competent dendritic cells. Blood 107, 2846–2854 (2006).
Gurtner, G.J., Newberry, R.D., Schloemann, S.R., McDonald, K.G. & Stenson, W.F. Inhibition of indoleamine 2,3-dioxygenase augments trinitrobenzene sulfonic acid colitis in mice. Gastroenterology 125, 1762–1773 (2003).
Hayashi, T. et al. Inhibition of experimental asthma by indoleamine 2,3-dioxygenase. J. Clin. Invest. 114, 270–279 (2004).
de Heer, H.J. et al. Essential role of lung plasmacytoid dendritic cells in preventing asthmatic reactions to harmless inhaled antigen. J. Exp. Med. 200, 89–98 (2004).
Mellor, A.L. et al. Cutting edge: CpG oligonucleotides induce splenic CD19+ dendritic cells to acquire potent indoleamine 2,3-dioxygenase-dependent T cell regulatory functions via IFN type 1 signaling. J. Immunol. 175, 5601–5605 (2005).
Wingender, G. et al. Systemic application of CpG-rich DNA suppresses adaptive T cell immunity via induction of IDO. Eur. J. Immunol. 36, 12–20 (2006).
Yang, C.H., Murti, A. & Pfeffer, L.M. Interferon induces NF-κB-inducing kinase/tumor necrosis factor receptor-associated factor-dependent NF-κB activation to promote cell survival. J. Biol. Chem. 280, 31530–31536 (2005).
Bonizzi, G. et al. Activation of IKKα target genes depends on recognition of specific κB binding sites by RelB:p52 dimers. EMBO J. 23, 4202–4210 (2004).
Gilliet, M. & Liu, Y.J. Generation of human CD8 T regulatory cells by CD40 ligand-activated plasmacytoid dendritic cells. J. Exp. Med. 195, 695–704 (2002).
Ito, T. et al. Plasmacytoid dendritic cells prime IL-10-producing T regulatory cells by inducible costimulator ligand. J. Exp. Med. 204, 105–115 (2007).
Hwu, P. et al. Indoleamine 2,3-dioxygenase production by human dendritic cells results in the inhibition of T cell proliferation. J. Immunol. 164, 3596–3599 (2000).
Fallarino, F. et al. The combined effects of tryptophan starvation and tryptophan catabolites down-regulate T cell receptor ζ-chain and induce a regulatory phenotype in naive T cells. J. Immunol. 176, 6752–6761 (2006).
Karagiannidis, C. et al. Glucocorticoids upregulate FOXP3 expression and regulatory T cells in asthma. J. Allergy Clin. Immunol. 114, 1425–1433 (2004).
Vacca, C. et al. CD40 ligation prevents onset of tolerogenic properties in human dendritic cells treated with CTLA-4-Ig. Microbes Infect. 7, 1040–1048 (2005).
Acknowledgements
We thank P. Mosci for maintaining the mutant strains of mice and performing histopathology; and G. Andrielli for digital art and image editing. Supported by the Italian Association for Cancer Research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
GITR-Ig mediates processing of p100 to generate p52. (PDF 171 kb)
Supplementary Fig. 2
Kinetic immunoblot analysis of NIK, IKKα and IKKβ expression in pDCs treated with specific siRNAs (+) in one experiment representative of three. (PDF 196 kb)
Supplementary Fig. 3
NIK and IKKα are required for IFN-α induction by GITR-Ig. (PDF 21 kb)
Supplementary Fig. 4
Dexamethasone in vivo up-regulates GITR and GITRL. (PDF 83 kb)
Supplementary Fig. 5
Cytokine production in vitro in response to a range of GITR-Ig concentrations by pDCs from mice treated or not with dexamethasone (dex). (PDF 23 kb)
Supplementary Fig. 6
GITR-Ig activates noncanonical NF-κB signaling in Ifnar−/− mice. (PDF 242 kb)
Rights and permissions
About this article
Cite this article
Grohmann, U., Volpi, C., Fallarino, F. et al. Reverse signaling through GITR ligand enables dexamethasone to activate IDO in allergy. Nat Med 13, 579–586 (2007). https://doi.org/10.1038/nm1563
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1563
This article is cited by
-
GITRL impairs hepatocyte repopulation by liver progenitor cells to aggravate inflammation and fibrosis by GITR+CD8+ T lymphocytes in CDE Mice
Cell Death & Disease (2024)
-
Immune regulation through tryptophan metabolism
Experimental & Molecular Medicine (2023)
-
GITR/GITRL reverse signalling modulates the proliferation of hepatic progenitor cells by recruiting ANXA2 to phosphorylate ERK1/2 and Akt
Cell Death & Disease (2022)
-
The role of indoleamine 2,3‐dioxygenase in allergic disorders
Molecular Biology Reports (2022)
-
3-hydroxy-L-kynurenamine is an immunomodulatory biogenic amine
Nature Communications (2021)