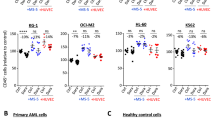
Abstract
The long-term survival of patients with acute myeloid leukemia (AML) is dismally poor. A permanent cure of AML requires elimination of leukemic stem cells (LSCs), the only cell type capable of initiating and maintaining the leukemic clonal hierarchy. We report a therapeutic approach using an activating monoclonal antibody directed to the adhesion molecule CD44. In vivo administration of this antibody to nonobese diabetic-severe combined immune-deficient mice transplanted with human AML markedly reduced leukemic repopulation. Absence of leukemia in serially transplanted mice demonstrated that AML LSCs are directly targeted. Mechanisms underlying this eradication included interference with transport to stem cell–supportive microenvironmental niches and alteration of AML-LSC fate, identifying CD44 as a key regulator of AML LSCs. The finding that AML LSCs require interaction with a niche to maintain their stem cell properties provides a therapeutic strategy to eliminate quiescent AML LSCs and may be applicable to other types of cancer stem cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Wang, J.C. & Dick, J.E. Cancer stem cells: lessons from leukemia. Trends Cell Biol. 15, 494–501 (2005).
Hope, K., Jin, L. & Dick, J.E. Acute myeloid leukemia originates from a hierarchy of leukemic stem cell classes that differ in self-renewal capacity. Nat. Immunol. 5, 738–743 (2004).
Lapidot, T. et al. A cell initiating human acute myeloid leukaemia after transplantation into SCID mice. Nature 367, 645–648 (1994).
Bonnet, D. & Dick, J.E. Human acute myeloid leukemia is organized as a hierarchy that originates from a primitive hematopoietic cell. Nat. Med. 3, 730–737 (1997).
Guan, Y. & Hogge, D.E. Proliferative status of primitive hematopoietic progenitors from patients with acute myelogenous leukemia (AML). Leukemia 14, 2135–2141 (2000).
Guzman, M.L. et al. Nuclear factor-κB is constitutively activated in primitive human acute myelogenous leukemia cells. Blood 98, 2301–2307 (2001).
Tallman, M.S. New strategies for the treatment of acute myeloid leukemia including antibodies and other novel agents. Hematology Am. Soc. Hematol. Educ. Program 2005, 143–150 (2005).
Wilson, A. & Trumpp, A. Bone-marrow haematopoietic-stem-cell niches. Nat. Rev. Immunol. 6, 93–106 (2006).
Nilsson, S.K. et al. Hyaluronan is synthesized by primitive hemopoietic cells, participates in their lodgment at the endosteum following transplantation, and is involved in the regulation of their proliferation and differentiation in vitro. Blood 101, 856–862 (2003).
Zhang, J. et al. Identification of the haematopoietic stem cell niche and control of the niche size. Nature 425, 836–841 (2003).
Calvi, L.M. et al. Osteoblastic cells regulate the haematopoietic stem cell niche. Nature 425, 841–846 (2003).
Cancelas, J.A. & Williams, D.A. Stem cell mobilization by β2-agonists. Nat. Med. 12, 278–279 (2006).
Lapidot, T., Dar, A. & Kollet, O. How do stem cells find their way home? Blood 106, 1901–1910 (2005).
Kortlepel, K., Bendall, L.J. & Gottlieb, D.J. Human acute myeloid leukaemia cells express adhesion proteins and bind to bone marrow fibroblast monolayers and extracellular matrix proteins. Leukemia 7, 1174–1179 (1993).
Reuss-Borst, M.A., Buhring, H.J., Klein, G. & Muller, C.A. Adhesion molecules on CD34+ hematopoietic cells in normal human bone marrow and leukemia. Ann. Hematol. 65, 169–174 (1992).
Liesveld, J.L. et al. Adhesive interactions of normal and leukemic human CD34+ myeloid progenitors: role of marrow stromal, fibroblast, and cytomatrix components. Exp. Hematol. 19, 63–70 (1991).
Tavor, S. et al. CXCR4 regulates migration and development of human acute myelogenous leukemia stem cells in transplanted NOD/SCID mice. Cancer Res. 64, 2817–2824 (2004).
Till, J.E. & McCulloch, E.A. Hemopoietic stem cell differentiation. Biochim. Biophys. Acta 605, 431–459 (1980).
Ponta, H., Sherman, L. & Herrlich, P.A. CD44: from adhesion molecules to signalling regulators. Nat. Rev. Mol. Cell Biol. 4, 33–45 (2003).
Bendall, L.J., Bradstock, K.F. & Gottlieb, D.J. Expression of CD44 variant exons in acute myeloid leukemia is more common and more complex than that observed in normal blood, bone marrow or CD34+ cells. Leukemia 14, 1239–1246 (2000).
Legras, S. et al. A strong expression of CD44-6v correlates with shorter survival of patients with acute myeloid leukemia. Blood 91, 3401–3413 (1998).
Avigdor, A. et al. CD44 and hyaluronic acid cooperate with SDF-1 in the trafficking of human CD34+ stem/progenitor cells to bone marrow. Blood 103, 2981–2989 (2004).
Turley, E.A., Noble, P.W. & Bourguignon, L.Y. Signaling properties of hyaluronan receptors. J. Biol. Chem. 277, 4589–4592 (2002).
Charrad, R.S. et al. Ligation of the CD44 adhesion molecule reverses blockage of differentiation in human acute myeloid leukemia. Nat. Med. 5, 669–676 (1999).
Charrad, R.S. et al. Effects of anti-CD44 monoclonal antibodies on differentiation and apoptosis of human myeloid leukemia cell lines. Blood 99, 290–299 (2002).
Gadhoum, Z. et al. CD44: a new means to inhibit acute myeloid leukemia cell proliferation via P27Kip1. Blood 103, 1059–1068 (2004).
Mohle, R. et al. The chemokine receptor CXCR-4 is expressed on CD34+ hematopoietic progenitors and leukemic cells and mediates transendothelial migration induced by stromal cell–derived factor-1. Blood 91, 4523–4530 (1998).
Mazurier, F., Doedens, M., Gan, O.I. & Dick, J.E. Rapid myeloerythroid repopulation after intrafemoral transplantation of NOD-SCID mice reveals a new class of human stem cells. Nat. Med. 9, 959–963 (2003).
Mazurier, F., Gan, O., McKenzie, J., Doedens, M. & Dick, J. Lentivector-mediated clonal tracking reveals intrinsic heterogeneity in the human hematopoietic stem cell compartment and culture-induced stem cell impairment. Blood 103, 545–552 (2004).
Madlambayan, G.J. et al. Dynamic changes in cellular and microenvironmental composition can be controlled to elicit in vitro human hematopoietic stem cell expansion. Exp. Hematol. 33, 1229–1239 (2005).
Peled, A. et al. Dependence of human stem cell engraftment and repopulation of NOD/SCID mice on CXCR4. Science 283, 845–848 (1999).
Kim, C.F. et al. Identification of bronchioalveolar stem cells in normal lung and lung cancer. Cell 121, 823–835 (2005).
Singh, S. et al. Identification of a cancer stem cell in human brain tumors. Cancer Res. 63, 5281–5288 (2003).
Al-Hajj, M., Wicha, M., Morrison, S.J. & Clarke, M.F. Prospective identification of tumorigenic breast cancer cells. Proc. Natl. Acad. Sci. USA 100, 3983–3988 (2003).
Liotta, L.A. & Kohn, E.C. The microenvironment of the tumour-host interface. Nature 411, 375–379 (2001).
Woerner, S.M. et al. Expression of CD44 splice variants in normal, dysplastic, and neoplastic cervical epithelium. Clin. Cancer Res. 1, 1125–1132 (1995).
Jo, D.Y., Rafii, S., Hamada, T. & Moore, M.A. Chemotaxis of primitive hematopoietic cells in response to stromal cell–derived factor-1. J. Clin. Invest. 105, 101–111 (2000).
Acknowledgements
We gratefully acknowledge M. Minden for providing AML samples, and members of the Dick laboratory and T. Lapidot for critical comments on the manuscript. This work was supported by a Canadian Institutes of Health Research studentship (K.J.H.) and fellowship (L.J.), grants from the Leukemia and Lymphoma Society, Fondation de France, Association pour la Recherche sur le Cancer (F.S.-J.) and grants from the National Cancer Institute of Canada with funds from the Canadian Cancer Society and the Terry Fox Foundation, Canadian Institutes of Health Research, Ontario Cancer Research Network, Genome Canada through the Ontario Genomics Institute, and a Canada Research Chair (J.E.D.). Research support (F.S.-J.) and H90 mAb production were provided by MAT Biopharma.
Author information
Authors and Affiliations
Contributions
The study was designed by L.J., K.J.H., F.S.-J. and J.E.D.; experiments were performed by L.J., K.J.H. and Q.Z.; and all authors contributed to writing the paper.
Corresponding author
Ethics declarations
Competing interests
F.S.-J. received some research support from MAT BioPharma, which also provided the purified monoclonal antibody used in the the study.
Supplementary information
Supplementary Fig. 1
Distinct characteristics of human cells in NOD-SCID mice transplanted with human AML or normal cord blood cells. (PDF 314 kb)
Supplementary Fig. 2
Increase in differentiated granulocytic cells induced by CD44 ligation in vivo. (PDF 106 kb)
Supplementary Fig. 3
Selective inhibitory effect of H90 on SL-ICs. (PDF 102 kb)
Supplementary Fig. 4
Effect of H90 on adhesion of primitive AML and normal BM cells to hyaluronan. (PDF 109 kb)
Supplementary Table 1
In vitro differentiation. (PDF 53 kb)
Rights and permissions
About this article
Cite this article
Jin, L., Hope, K., Zhai, Q. et al. Targeting of CD44 eradicates human acute myeloid leukemic stem cells. Nat Med 12, 1167–1174 (2006). https://doi.org/10.1038/nm1483
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1483
This article is cited by
-
Induction of DNMT1-dependent demethylation of SHP-1 by the natural flavonoid compound Baicalein overcame Imatinib-resistance in CML CD34+ cells
Cell Communication and Signaling (2023)
-
Exploring the dynamic interplay between cancer stem cells and the tumor microenvironment: implications for novel therapeutic strategies
Journal of Translational Medicine (2023)
-
Cancer Stem Cells are Actually Stem Cells with Disordered Differentiation: the Monophyletic Origin of Cancer
Stem Cell Reviews and Reports (2023)
-
PDP1 is a key metabolic gatekeeper and modulator of drug resistance in FLT3-ITD-positive acute myeloid leukemia
Leukemia (2023)
-
A primary hierarchically organized patient-derived model enables in depth interrogation of stemness driven by the coding and non-coding genome
Leukemia (2022)