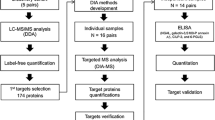
Abstract
We analyzed urinary polypeptides from individuals with neonatal ureteropelvic junction (UPJ) obstruction to predict which individuals with this condition will evolve toward obstruction that needs surgical correction. We identified polypeptides that enabled diagnosis of the severity of obstruction and validated these biomarkers in urine collected in a prospective blinded study. Using these noninvasive biomarkers, we were able to predict, several months in advance and with 94% precision, the clinical evolution of neonates with UPJ obstruction.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Hanash, S. Nature 422, 226–232 (2003).
Kolch, W., Mischak, H., Chalmers, M.J., Pitt, A. & Marshall, A.G. Rapid Commun. Mass Spectrom. 18, 2365–2366 (2004).
Haubitz, M. et al. Kidney Int. 67, 2313–2320 (2005).
Kolch, W., Neusüß, C., Pelzing, M. & Mischak, H. Mass Spectrom. Rev. 24, 959–977 (2005).
Csaicsich, D., Greenbaum, L.A. & Aufricht, C. Curr. Opin. Urol. 14, 213–217.
Chevalier, R.L. J. Urol. 172, 852–857 (2004).
Chevalier, R.L. & Peters, C.A. Pediatr. Nephrol. 18, 576–606 (2003).
Kaiser, T. et al. Blood 104, 340–349 (2004).
Theodorescu, D. et al. Electrophoresis 26, 2797–2808 (2005).
Eyre, D. Arthritis Res. 4, 30–35 (2002).
Bonnemann, C.G. et al. Proc. Natl. Acad. Sci. USA 97, 1212–1217 (2000).
Cochrane, A.L. et al. J. Am. Soc. Nephrol. 16, 3623–3630 (2005).
Wittke, S. et al. Electrophoresis 26, 1476–1487 (2005).
Acknowledgements
This work was supported by a grant from the Clinical Research Hospital Program from the French Ministry of Health (Programme Hospitalier de Recherche Clinique 2004) and sponsored by the University Hospital of Toulouse for regulatory and ethic submission. J.P.S. was supported by Inserm and the “Direction Régional de la Recherche” (Centre Hospitalier Universitaire de Toulouse) under the Interface program. We would like to thank P. Koester for his technical assistance during the MS/MS experiments.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Harald Mischak is founder and co-owner of Mosaiques Diagnostics, which developed the CE-MS technology and software solutions utilized in this manuscript.
Supplementary information
Supplementary Fig. 1
DMSA scintigraphy one year after surgery of an OP patient showing severely degraded function of one of the kidneys. (PDF 894 kb)
Supplementary Fig. 2
MS/MS (LIFT TOF-TOF) spectra of four potential biomarkers from human urine for UPJ-obstruction. (PDF 1584 kb)
Supplementary Table 1
Classification of different UPJ-obstruction patients. (PDF 20 kb)
Supplementary Table 2
Polypeptides of good discriminatory value identified by mass and migration time. (PDF 17 kb)
Supplementary Table 3
All UPJ patients used in the study as well as the healthy controls. (PDF 767 kb)
Supplementary Tables 4
Raw mass spectrometry data file (pivot table). (XLS 31990 kb)
Rights and permissions
About this article
Cite this article
Decramer, S., Wittke, S., Mischak, H. et al. Predicting the clinical outcome of congenital unilateral ureteropelvic junction obstruction in newborn by urinary proteome analysis. Nat Med 12, 398–400 (2006). https://doi.org/10.1038/nm1384
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1384
This article is cited by
-
Comparison of clean catch and bag urine using LC–MS/MS proteomics in infants
Pediatric Nephrology (2024)
-
Biomarker bei ureteropelviner Stenose
Monatsschrift Kinderheilkunde (2023)
-
Early detection of ureteropelvic junction obstruction in neonates with prenatal diagnosis of renal pelvis dilatation using 1H NMR urinary metabolomics
Scientific Reports (2022)
-
Management of antenatal hydronephrosis
Pediatric Nephrology (2020)
-
A single-center study to evaluate the efficacy of a fetal urine peptide signature predicting postnatal renal outcome in fetuses with posterior urethral valves
Pediatric Nephrology (2020)