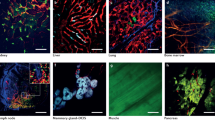
Abstract
A significant impediment to the widespread use of noninvasive in vivo vascular imaging techniques is the current lack of suitable intravital imaging probes. We describe here a new strategy to use viral nanoparticles as a platform for the multivalent display of fluorescent dyes to image tissues deep inside living organisms. The bioavailable cowpea mosaic virus (CPMV) can be fluorescently labeled to high densities with no measurable quenching, resulting in exceptionally bright particles with in vivo dispersion properties that allow high-resolution intravital imaging of vascular endothelium for periods of at least 72 h. We show that CPMV nanoparticles can be used to visualize the vasculature and blood flow in living mouse and chick embryos to a depth of up to 500 μm. Furthermore, we show that the intravital visualization of human fibrosarcoma-mediated tumor angiogenesis using fluorescent CPMV provides a means to identify arterial and venous vessels and to monitor the neovascularization of the tumor microenvironment.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
McDonald, D.M. & Choyke, P.L. Imaging of angiogenesis: from microscope to clinic. Nat. Med. 9, 713–725 (2003).
Yang, C.S. et al. Nanoparticle-based in vivo investigation on blood-brain barrier permeability following ischemia and reperfusion. Anal. Chem. 76, 4465–4471 (2004).
Josephson, L., Kircher, M.F., Mahmood, U., Tang, Y. & Weissleder, R. Near-infrared fluorescent nanoparticles as combined MR/optical imaging probes. Bioconjug. Chem. 13, 554–560 (2002).
Lee, P.J. & Peyman, G.A. Visualization of the retinal and choroidal microvasculature by fluorescent liposomes. Methods Enzymol. 373, 214–233 (2003).
Rizzo, V., Steinfeld, R., Kyriakides, C. & DeFouw, D.O. The microvascular unit of the 6-day chick chorioallantoic membrane: a fluorescent confocal microscopic and ultrastructural morphometric analysis of endothelial permselectivity. Microvasc. Res. 46, 320–332 (1993).
Jilani, S.M. et al. Selective binding of lectins to embryonic chicken vasculature. J. Histochem. Cytochem. 51, 597–604 (2003).
Pardanaud, L., Altmann, C., Kitos, P., Dieterlen-Lievre, F. & Buck, C.A. Vasculogenesis in the early quail blastodisc as studied with a monoclonal antibody recognizing endothelial cells. Development 100, 339–349 (1987).
Larson, D.R. et al. Water-soluble quantum dots for multiphoton fluorescence imaging in vivo. Science 300, 1434–1436 (2003).
Kirchner, C. et al. Cytotoxicity of colloidal CdSe and CdSe/ZnS nanoparticles. Nano Lett. 5, 331–338 (2005).
Goldbach, R. & Van Kammen, A. Structure replication and expression of the bipartite genome of Cowpea mosaic virus. in Molecular Plant Virology (ed. Davies, J.) 83–120 (CRC Press, Boca Raton, Florida, 1985).
Lomonossoff, G. & Shanks, M. The nucleotide sequence of Cowpea mosaic virus B RNA. EMBO J. 2, 2253–2258 (1983).
Porta, C. et al. Cowpea mosaic virus-based chimaeras. Effects of inserted peptides on the phenotype, host range, and transmissibility of the modified viruses. Virology 310, 50–63 (2003).
Chatterji, A. et al. Chemical conjugation of heterologous proteins on the surface of Cowpea mosaic virus. Bioconjug. Chem. 15, 807–813 (2004).
Lin, T. et al. The refined crystal structure of cowpea mosaic virus at 2.8 Å resolution. Virology 265, 20–34 (1999).
Brennan, F.R., Jones, T.D. & Hamilton, W.D. Cowpea mosaic virus as a vaccine carrier of heterologous antigens. Mol. Biotechnol. 17, 15–26 (2001).
Nicholas, B.L. et al. Characterization of the immune response to canine parvovirus induced by vaccination with chimaeric plant viruses. Vaccine 20, 2727–2734 (2002).
Wang, Q., Kaltgrad, E., Lin, T., Johnson, J.E. & Finn, M.G. Natural supramolecular building blocks. Wild-type cowpea mosaic virus. Chem. Biol. 9, 805–811 (2002).
Chatterji, A. et al. New addresses on an addressable virus nanoblock; uniquely reactive Lys residues on cowpea mosaic virus. Chem. Biol. 11, 855–863 (2004).
Gref, R. et al. Poly(ethylene glycol)-coated nanospheres: potential carriers for intravenous drug administration. Pharm. Biotechnol. 10, 167–198 (1997).
Seandel, M., Noack-Kunnmann, K., Zhu, D., Aimes, R.T. & Quigley, J.P. Growth factor-induced angiogenesis in vivo requires specific cleavage of fibrillar type I collagen. Blood 97, 2323–2332 (2001).
Johnson, J., Lin, T. & Lomonossoff, G. Presentation of heterologous peptides on plant viruses: genetics, structure, and function. Annu. Rev. Phytopathol. 35, 67–86 (1997).
Lin, T., Porta, C., Lomonossoff, G. & Johnson, J.E. Structure-based design of peptide presentation on a viral surface: the crystal structure of a plant/animal virus chimera at 2.8 Å resolution. Fold. Des. 1, 179–187 (1996).
Lomonossoff, G.P. & Johnson, J.E. The synthesis and structure of comovirus capsids. Prog. Biophys. Mol. Biol. 55, 107–137 (1991).
Rae, C. et al. Systemic trafficking of plant virus nanoparticles in mice via the oral route. Virology 343, 224–235 (2005).
Tsourkas, A. et al. In vivo imaging of activated endothelium using an anti-VCAM-1 magnetooptical probe. Bioconjug. Chem. 16, 576–581 (2005).
White, J.G., Amos, W.B. & Fordham, M. An evaluation of confocal versus conventional imaging of biological structures by fluorescence light microscopy. J. Cell Biol. 105, 41–48 (1987).
Dessens, J.T. & Lomonossoff, G.P. Cauliflower mosaic virus 35S promoter-controlled DNA copies of cowpea mosaic virus RNAs are infectious on plants. J. Gen. Virol. 74, 889–892 (1993).
Jones, E.A. et al. Dynamic in vivo imaging of postimplantation mammalian embryos using whole embryo culture. Genesis 34, 228–235 (2002).
Zijlstra, A. et al. Collagenolysis-dependent angiogenesis mediated by matrix metalloproteinase-13 (collagenase-3). J. Biol. Chem. 279, 27633–27645 (2004).
Hahm, B., Arbour, N. & Oldstone, M.B. Measles virus interacts with human SLAM receptor on dendritic cells to cause immunosuppression. Virology 323, 292–302 (2004).
Sayle, R.A. & Milner-White, E.J. RASMOL: biomolecular graphics for all. Trends Biochem. Sci. 20, 374 (1995).
Schwede, T., Kopp, J., Guex, N. & Peitsch, M.C. SWISS-MODEL: an automated protein homology-modeling server. Nucleic Acids Res. 31, 3381–3385 (2003).
Reddy, V.S. et al. Virus Particle Explorer (VIPER), a website for virus capsid structures and their computational analyses. J. Virol. 75, 11943–11947 (2001).
Acknowledgements
This study was supported by US National Institutes of Health grants R01 HL65738 and R21 HL72270 (to H.S.), AI47823, CA112075 and N01-CO-27181 (to M.M.), R01 CA55852 and R01 CA105412 (to J.Q.) and NSERC PDF-313420-2005 (to J.L.). The authors thank M. Wood (Scripps Microscopy Core Facility), X. Zhang, E. Rockenstein and N. MacLean (University of California San Diego) for their technical assistance.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
A patent has been filed to cover the intellectual property described in this manuscript. This patent application has furthermore been exclusively licensed to a private third party.
Supplementary information
Supplementary Fig. 1
Cell adhesion and uptake is inhibited by coating of CPMV with PEG. (PDF 129 kb)
Supplementary Fig. 2
CPMV uptake is eliminated in chick embryos and reduced significantly in adult mice by PEG coating. (PDF 142 kb)
Supplementary Video 1
Fluorescent timelapse imaging of yolk sac of 11.5 d mouse embryo at 10 magnification 1 h after CPMV-A555 perfusion. (AVI 8067 kb)
Supplementary Video 2
Fluorescent timelapse imaging of chick embryo CAM vasculature at 20 magnification 30 min after CPMV-A555 perfusion. (AVI 3675 kb)
Supplementary Video 3
Fluorescent timelapse imaging of chick embryo CAM vasculature at 10 magnification 30 min after CPMV-A555 (left) and CPMV-PEG-FITC (right) coperfusion, duration; 11 s. (AVI 5813 kb)
Supplementary Video 4
Fluorescent timelapse imaging of internal vasculature of 11.5 d mouse embryo at 10 magnification 1 h after CPMV-A555 perfusion. (AVI 10674 kb)
Rights and permissions
About this article
Cite this article
Lewis, J., Destito, G., Zijlstra, A. et al. Viral nanoparticles as tools for intravital vascular imaging. Nat Med 12, 354–360 (2006). https://doi.org/10.1038/nm1368
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1368
This article is cited by
-
Self-assembled Viral Nanoparticles as Targeted Anticancer Vehicles
Biotechnology and Bioprocess Engineering (2021)
-
Nano drug delivery systems in upper gastrointestinal cancer therapy
Nano Convergence (2020)
-
Design and biosynthesis of functional protein nanostructures
Science China Life Sciences (2020)
-
Multiphoton imaging reveals that nanosecond pulsed electric fields collapse tumor and normal vascular perfusion in human glioblastoma xenografts
Scientific Reports (2016)
-
Intracellular delivery of antibodies by chimeric Sesbania mosaic virus (SeMV) virus like particles
Scientific Reports (2016)