Abstract
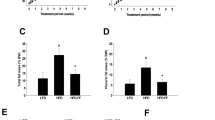
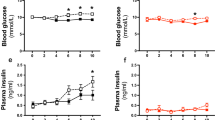
When CS7BI6 male mice are fed a high–fat diet, they develop significant fatty streak lesions in the aorta. Addition of tamoxifen (TMX) to a high–fat diet, equivalent to a dose of approximately 1 mg TMX per kg body weight per day, suppressed the diet–induced increase in the area of lipid staining in the aortic sinus of the mice by 88% and in the average number of lesions by 86%. The TMX–treated mice had 11% ± 5% less total plasma cholesterol, with most of the reduction in the high density lipoprotein fraction, whereas plasma triglycerides were significantly elevated, and circulating concentrations of 17β–estradiol and testosterone were unaffected. Both circulating and aortic concentrations of active and latent transforming growth factor–β (TGF–β) were substantially elevated by TMX. The inhibition of lesion formation may be due, at least in part, to cardiovascular protection by TGF–β.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Kirschenlohr, H.L., Metcalfe, J.C., Weissberg, P.L. & Grainger, D.J. Adult human aortic smooth muscle cells in culture produce active TGF-β. Am. J. Physiol. 265, C571–C576 (1993).
Grainger, D.J. et al. Proliferation of human smooth muscle cells promoted by lipoprotein (a). Science 260, 1655–1658 (1993).
Kojima, S., Harpel, P.C. & Rifkin, D.B. Lipoprotein (a) inhibits generation of transforming growth factor P: An endogenous inhibitor of smooth muscle cell migration. J.Cell.Biol 113, 1439–1445 (1991).
Grainger, D.J., Kemp, P.R., Liu, A.C., Lawn, R.M. & Metcalfe, J.C. Activation of transforming growth factor-β is inhibited in transgenic apolipoprotein (a) mice. Nature 370, 460–462 (1994).
Grainger, D.J. & Metcalfe, J.C. A pivotal role for TGF-β in atherogenesis? Biol. Rev. (in the press).
Grainger, D.J. et al. The serum concentration of active transforming growth factor-β is severely depressed in advanced atherosclerosis. Nature Med. 1, 74–79 (1995).
Paigen, B., Morrow, A., Brandon, C., Mitchell, D. & Holmes, P. Variation in susceptibility to atherosclerosis among inbred strains of mice. Atherosclerosis 57, 313–327 (1985).
Paigen, B., Ishida, B., Verstuyft, J., Winters, R.B. & Albee, D. Atherosclerosis susceptibility differences among progenitors of recombinant inbred strains of mice. Arteriosclerosis 10, 316–323 (1990).
Stewart-Philips, J.L. St Lough, J. Pathology of atherosclerosis in cholesterol-fed, susceptible mice. Atherosclerosis 90, 211–218 (1991).
Emeson, E.E. & Shen, M.-L. Accelerated atherosclerosis in hyperlipidemic C57B1/6 mice treated with cyclosporin A. Am. J. Pathol. 142, 1906–1915 (1993).
Grainger, D.J., Weissberg, P.L. & Metcalfe, J.C. Tamoxifen decreases the rate of proliferation of rat vascular smooth muscle cells in culture by inducing production of transforming growth factor p. Biochem. J. 294, 109–112 (1993).
Colletta, A.A. et al. Anti-oestrogens induce the secretion of active transforming growth factor p from human fetal fibroblasts. Br. J. Cancer 62, 405–409 (1990).
Colletta, A.A. et al. The growth inhibition of human breast cancer cells by a novel synthetic progestin involves the induction of transforming growth factor beta. J. din. Invest. 87, 277–283 (1991).
Butta, A. et al. Induction of transforming growth factor pi in human breast cancer in vivo following tamoxifen treatment. Cancer Res. 52, 4261–4264 (1992).
Grainger, D.J., Mosedale, D.E., Metcalfe, J.C., Weissberg, P.L. & Kemp, P.R. Active and total TGF-β in human sera, platelets and plasma. Clin. Chim. Acta 235, 11–31 (1995).
Kubo, C., Johnson, B.C., Day, N.K. & Good, R.A. Effects of calorie restriction on immunological functions and development of autoimmune-disease in NZB mice. Proc. Soc. exp. Biol. Med. 201, 192–199 (1992)
Bursting, S.D., Perkins, S.N. & Phang, J.M. Calorie restriction delays spontaneous tumorigenesis in p53-knockout transgenic mice. Proc. natn. Acad, Sci. U.S.A. 91, 7035–7040 (1994).
Williams, P.T., Krauss, R.M., Stefanick, M.X., Vranizan, K.M. & Wood, P.O. Effects of low-fat diet, calorie restriction and running on high-density-lipoprotein (HDL) subfraction concentrations in moderately overweight men. Metabolism 43, 655–663 (1994).
Paigen, B. et al. Ath-1, a gene determining atherosclerosis susceptibility and high density lipoprotein levels in mice. Proc. natn. Acad. Sci. U.S.A. 84, 3763–3767 (1987).
Paigen, B., Havens, M.B. & Morrow, A. Effect of 3-methylcholanthrene on the development of aortic lesions in mice. Cancer Res. 45, 3850–3855 (1985).
Furr, B.J.A. & Jordan, V.C. The pharmacology and clinical uses of tamoxifen. Pharmac. Ther. 25, 127–205 (1984).
Rowlands, M.G., Parr, I.B., McCague, R., Jarman, M. & Goddard, P.M. Variation of the inhibition of calmodulin dependent cyclic AMP phosphodiesterase amongst analogues of tamoxifen. Biochem. Pharmacol. 40, 283–289 (1990).
0 Brian, C.A., Liskamp, R.M., Solomon, D.H. & Weinstein, I.E. Triphenylethylenes: A new class of protein kinase C inhibitors. J. natn. Cancer Inst. 76, 1243–1246 (1986).
Bertelli, G. et al. Adjuvant tamoxifen in primary breast cancer: Influence on plasma lipids and antithrombin III levels. Breast Cancer Res. Treat. 12, 307–310 (1988).
Caleffi, M. et al. Effect of tamoxifen on estrogen binding, lipid and lipoprotein concentrations and blood clotting parameters in premenopausal women with breast pain. J. Endocrinol. 119, 335–339 (1988).
Love, R.R. et al. Effects of tamoxifen therapy on lipid and lipoprotein levels in postmenopausal patients with node-negative breast cancer. J. natn. Cancer Inst. 82, 1327–1332 (1990).
Shewmon, D.A. et al. Tamoxifen and estrogen lower circulating lipoprotein (a) concentrations in healthy postmenopausal women. Arteriosclerosis 14, 1586–1593 (1994).
Shanahan, C.M., Weissberg, P.L. & Metcalfe, J.C. Isolation of gene markers of differentiated and proliferating vascular smooth muscle cells. Circ. Res 73, 193–204 (1993).
Giachelli, C., Bae, N., Lombardi, D., Majesky, M. & Schwartz, S. Molecular cloning and characterisation of 2B7, a rat mRNA which distinguishes smooth muscle cell phenotypes in vitro and is identical to osteopontin. Biochem. biophys. Res. Comm. 177, 867–873 (1991).
Giachelli, C. et al. Osteopontin is elevated during neointima formation in rat arteries and is a novel component of human atherosclerotic plaques. J. Clin. Invest 92, 1686–1696 (1993).
Shanahan, C.M., Gary, N.R.B. Metcalfe, J.C. & Weissberg, P.L. High expression of genes for calcification-regulating proteins in human atherosclerotic plaques. J. din. Invest. 93, 2393–1402 (1994).
Paigen, B., Holmes, P., Mitchell, D. & Albee, D. Comparison of atherosclerotic lesions and HDL-lipld levels in male, female and testosterone-treated female mice from strains C57B16, BALB/c and C3H. Atherosclerosis 64, 215–221 (1987).
Gordon, D.J. & Rifkind, B.M. Current concepts: High density lipoprotein the clinical implications of recent studies. NewEngl. J. Med. 321, 1311–1316 (1989).
Paszty, C., Maeda, N., Verstuyft, J. & Rubin, E.M. Apolipoprotein Al transgene corrects apolipoprotein E deficiency-induced atherosclerosis in mice. J. din. Invest. 94, 899–903 (1994).
Plump, A.S., Scott, C.J. & Breslow, J.L. Human apolipoprotein A-l gene expression increases high density lipoprotein and suppresses atherosclerosis in the apolipoprotein E-deflcient mouse. Proc. natn. Acad. Sci. U.SA. 91, 9607–9611 (1994).
Plump, A.S. et al. Severe hypercholesterolaemia and atherosclerosis in apolipoprotein E-deficient mice created by homologous recombination in ES cells. Cell 71, 343–353 (1992).
MacDonald, C.C. & Stewart, H.J. Fatal myocardial infarction in the Scottish adjuvant tamoxifen trial. Br. Med. J. 303, 435–437 (1991).
van Leeuwen, F.E. et al. Risk of endometrial cancer after tamoxifen treatment of breast cancer. Lancet 343, 448–452 (1994).
Allain, C.A., Poon, L.S., Chan, C.S.G. Richmond, W. & Fu, P.C. Enzymatic determination of total serum cholesterol. Clin. Chem. 20, 470–479 (1974).
Yokode, M., Hammer, R.E., Ishibashi, S., Brown, M.S. & Goldstein, J.L. Diet-induced hypercholesterolemia in mice: Prevention by overexpression of LDL receptors. Science 250, 1273–1275 (1990).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Grainger, D., Witchell, C. & Metcalfe, J. Tamoxifen elevates transforming growth factor–β and suppresses diet–induced formation of lipid lesions in mouse aorta. Nat Med 1, 1067–1073 (1995). https://doi.org/10.1038/nm1095-1067
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nm1095-1067
This article is cited by
-
Clinically used selective estrogen receptor modulators affect different steps of macrophage-specific reverse cholesterol transport
Scientific Reports (2016)
-
Tamoxifen and raloxifene suppress the proliferation of estrogen receptor-negative cells through inhibition of glutamine uptake
Cancer Chemotherapy and Pharmacology (2011)
-
Association of TGFB1 -509 C>T polymorphism with breast cancer: evidence from a meta-analysis involving 23,579 subjects
Breast Cancer Research and Treatment (2010)
-
Involvement of osteopontin as a core protein in cholesterol gallstone formation
Journal of Hepato-Biliary-Pancreatic Surgery (2009)
-
Transforming growth factors-β are not good biomarkers of chemopreventive efficacy in a preclinical breast cancer model system
Breast Cancer Research (2000)