Abstract
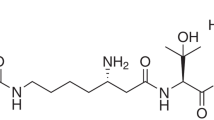
The rate of expansion of primary prostatic carcinoma is comparatively slow, with tumours frequently taking years or decades to reach clinically relevant size. We now report the presence of an endogenous inhibitor, derived from aqueous extracts of human prostate tissue, which blocks prostatic carcinoma cell proliferation in vitro and prevents subcutaneous tumour expansion in vivo. Purification and characterization revealed the inhibitor to be spermine, a polyamine known to be locally abundant in the prostate. These results suggest that endogenous polyamine can negatively regulate the growth of prostatic carcinoma cells at their primary site in vivo and may explain the slow rate of primary tumour expansion in the prostate.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Boring, C.C., Squires, T.S., Tong, T. & Montgomery, S. Cancer statistics, 1994. CA: Cancer J. Clinicians 44, 7–26 (1994).
Scott, R. Jr, Mutchnik, D.L., Laskowski, T.Z. & Schmalhorst, W.R. Cancer of the prostate in elderly men: Incidence, growth characteristics and clinical significance. J. Urol. 101, 602–607 (1969).
Stamey, T.A. et al. Localized prostate cancer. Relationship of tumor volume to clinical significance for treatment of prostate cancer. Cancer 71 (suppl. 3), 933–938 (1993).
Mc'Neal, J.E. Prostatic microcarcinomas in relation to cancer origin and the evolution to clinical cancer. Cancer 71 (suppl. 3), 984–991 (1993).
Rossi, M.C. & Zetter, B.R. Selective stimulation of prostatic carcinoma cell proliferation by transferrin. Proc. natn. Acad. Sci. U.SA. 89, 6197–6201 (1992).
Heston, W.D.W. Prostatic polyamines and polyamine targeting as a new approach to therapy of prostatic cancer. Cancer Surv. 11, 217–238 (1991).
Mann, T. Biochemistry of Semen and of the Male Reproductive Tract 193–200 (Wiley, New York, 1964).
Heby, O. & Andersson, G. Simplified micro-method for the quantitative analysis of putrescine, spermidine and spermine in the urine. J. Chromatogr. 145, 73–80 (1978).
Webber, M.M. & Chaproniere-Rickenberg, D. Spermine oxidation products are selectively toxic to fibroblasts in cultures of normal human prostatic epithelium. Cell Biol. int. Rep. 4, 185–193 (1980).
Allen, R.D. & Roberts, T.K. Role of spermine in the cytotoxic effects of seminal plasma. Am. J. repro. Immunol. 13, 4–8 (1987).
Coffino, P. & Poznanski, A. Killer polyamine? J. cell. Biochem. 45, 54–58 (1991).
Brunton, V.G., Grant, M.H. & Wallace, H.M. Mechanisms of spermine toxicity in baby-hamster kidney (BHK) cells. The role of amine oxidases and oxidative stress. Biochem. J. 280, 193–198 (1991).
Kameji, T. & Pegg, A.E. Inhibition of translation of mRNAs for ornithine decar-boxylase and S-adenosylmethionine decarboxylase by polyamines. J. biol. Chem. 262, 2427–2430 (1987).
Gahl, W.A. & Pitot, H.C. Reversal by aminoguanidine of the inhibition of proliferation of human fibroblasts by spermidine and spermine. Chem. Biol. Interact. 22, 91–98 (1978).
Isaacs, J.T., Isaacs, W.B., Feitz, W.F. & Scheres, J. Establishment and characterizationof seven Dunning rat prostatic cancer cell lines and their use in developing methods for predicting metastatic abilities of prostatic cancers. Prostate 9, 261–281 (1986).
Chackal-Roy, M., Niemeyer, C., Moore, M. & Zetter, B.R. Stimulation of humanprostatic carcinoma cell growth by factors present in human bone marrow. J.clin. Invest. 84, 43–50 (1989).
Celano, P., Baylin, S.B. & Casero, R. Jr., Polyamines differentially modulate the transcription of growth-associated genes in human colon carcinoma cells. J.biol. Chem. 264, 8922–8927 (1989).
Pegg, A.E. Polyamine metabolism and its importance in neoplastic growth and as a target for chemotherapy. Cancer Res. 48, 759–774 (1988).
Hibshoosh, H., Johnson, M. & Weinstein, I.B. Effects of overexpression of or-nithine decarboxylase (ODC) on growth control and oncogene-induced celltransformation. Oncogene 6, 739–743 (1991).
Moshier, J.A., Dosescu, J., Skunca, M. & Luk, G.D. Transformation of NIH/3T3 cells by ornithine decarboxylase overexpression. Cancer Res. 53, 2618–2622 (1993).
Auvinen, M., Paasinen, A., Andersson, L.C. & Holtta, E. Ornithine decarboxy lase activity is critical for cell transformation. Nature 360, 355–358 (1992).
Zaneveld, L.J.D. & Tauber, P.F. The Prostatic Cell: Structure and Function, Part A, 265–277 (Liss, New York, 1981).
Bernacki, R.J., Bergeron, R.J. & Porter, C.W. Antitumor activity of N,N′-bis(ethyl)spermine homologues against human MALME-3 melanoma xenografts. Cancer Res. 52, 2424–2430 (1992).
Porter, C.W., Bernacki, R.J., Miller, J. & Bergeron, R.J. Antitumor activity of N1,N11-bis(ethyl)norspermine against human melanoma xenografts and possi ble biochemical correlates of drug action. Cancer Res. 53, 581–586 (1993).
Davidson, N.E., Mank, A.R., Prestigiacomo, L.J., Bergeron, R.J. & Casero, R.A. Growth inhibition of hormone-responsive and resistant human breast cancer cells in culture by N1,N12-bis(ethyl)spermine. Cancer Res. 53, 2071–2075 (1993).
Danzin, C., Jung, M.J., Grove, J. & Bey, P. Effect of alpha-difluoromethylor- nithine, an enzyme-activated inhibitor of ornithine decarboxylase, on polyamine levels in rat tissues. Life Sci. 24, 519–524 (1979).
Janne, J., Raina, A. & Sjimes, M. Spermidine and spermine in rat tissues at differ ent ages. Actaphysiol. Scand. 62, 352–358 (1964).
Kabra, P.M., Lee, H.K., Lubich, W.P. & Morton, L.J. Solid-phase extraction and determination of dansyl derivatives of unconjugated and acetylated polyamines by reversed-phase liquid chromatography: improved separation systems for polyamines in cerebrospinal fluid, urine and tissue. J. Chromatogr. 380, 19–32 (1986).
Carmichael, J., DeGraff, W.G., Gazdar, A.F., Minna, J.D. & Mitchell, J.B. Evaluation of a tetrazolium-based semiautomated colorimetric assay: Assessment of nemo-sensitivity testing. Cancer Res. 47, 936–942 (1987).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Smith, R., Litwin, M., Lu, Y. et al. Identification of an endogenous inhibitor of prostatic carcinoma cell growth. Nat Med 1, 1040–1045 (1995). https://doi.org/10.1038/nm1095-1040
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nm1095-1040
This article is cited by
-
High resolution magic angle spinning MRS in prostate cancer
Magnetic Resonance Materials in Physics, Biology and Medicine (2022)
-
Developments in proton MR spectroscopic imaging of prostate cancer
Magnetic Resonance Materials in Physics, Biology and Medicine (2022)
-
Metabolomic imaging of prostate cancer with magnetic resonance spectroscopy and mass spectrometry
European Journal of Nuclear Medicine and Molecular Imaging (2013)
-
A decade in prostate cancer: from NMR to metabolomics
Nature Reviews Urology (2011)
-
Two-dimensional MR spectroscopy of healthy and cancerous prostates in vivo
Magnetic Resonance Materials in Physics, Biology and Medicine (2008)