Abstract
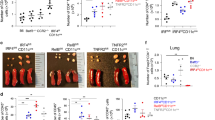
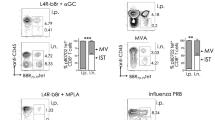
Bronchus-associated lymphoid tissue (BALT) is occasionally found in the lungs of mice and humans; however, its role in respiratory immunity is unknown. Here we show that mice lacking spleen, lymph nodes and Peyer's patches generate unexpectedly robust primary B- and T-cell responses to influenza, which seem to be initiated at sites of induced BALT (iBALT). Areas of iBALT have distinct B-cell follicles and T-cell areas, and support T and B-cell proliferation. The homeostatic chemokines CXCL13 and CCL21 are expressed independently of TNFα and lymphotoxin at sites of iBALT formation. In addition, mice with iBALT, but lacking peripheral lymphoid organs, clear influenza infection and survive higher doses of virus than do normal mice, indicating that immune responses generated in iBALT are not only protective, but potentially less pathologic, than systemic immune responses. Thus, iBALT functions as an inducible secondary lymphoid tissue for respiratory immune responses.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Goodnow, C.C. Chance encounters and organized rendezvous. Immunol. Rev. 156, 5–10 (1997).
Barker, C.F. & Billingham, R.E. The role of afferent lymphatics in the rejection of skin homografts. J. Exp. Med. 128, 197–221 (1968).
Zinkernagel, R.M. et al. Antigen localisation regulates immune responses in a dose- and time-dependent fashion: a geographical view of immune reactivity. Immunol. Rev. 156, 199–209 (1997).
Weninger, W., Crowley, M.A., Manjunath, N. & von Andrian, U.H. Migratory properties of naive, effector, and memory CD8+ T cells. J. Exp. Med. 194, 953–966 (2001).
Roman, E. et al. CD4 effector T cell subsets in the response to influenza: heterogeneity, migration, and function. J. Exp. Med. 196, 957–968 (2002).
Banks, T.A. et al. Lymphotoxin-α-deficient mice: effects on secondary lymphoid organ development and humoral immune responsiveness. J. Immunol. 155, 1685–1693 (1995).
de Togni, P. et al. Abnormal development of peripheral lymphoid organs in mice deficient in lymphotoxin. Science 264, 703–707 (1994).
Miyawaki, S. et al. A new mutation, aly, that induces a generalized lack of lymph nodes accompanied by immunodeficiency in mice. Eur. J. Immunol. 24, 429–434 (1994).
Shinkura, R. et al. Alymphoplasia is caused by a point mutation in the mouse gene encoding NFκB-inducing kinase. Nat. Genet. 22, 74–77 (1999).
Karrer, U. et al. On the key role of secondary organs in antiviral immune responses studied in alymphoplastic (aly/aly) and spleenless (Hox 11−/−) mutant mice. J. Exp. Med. 185, 2157–2170 (1997).
Rennert, P.D. et al. Essential role of lymph nodes in contact hypersensitivity revealed in lymphotoxin-α-deficient mice. J. Exp. Med. 193, 1227–1238 (2001).
Lakkis, F.G., Arakelov, A., Konieczny, B.T. & Inoue, Y. Immunological 'ignorance' of vascularized organ transplants in the absence of secondary lymphoid tissue. Nat. Med. 6, 686–688 (2000).
Lund, F.E. et al. Lymphotoxin-α-deficient mice make delayed, but effective, T and B cell responses to influenza. J. Immunol. 169, 5236–5243 (2002).
Lee, B.J., Santee, S., Von Gesjen, S., Ware, C.F. & Sarawar, S.R. Lymphotoxin-α-deficient mice can clear a productive infection with murine gammaherpesvirus 68 but fail to develop splenomegaly or lymphocytosis. J. Virol. 74, 2786–2792 (2000).
Feuerer, M. et al. Bone marrow as a priming site for T-cell responses to blood-borne antigen. Nat. Med. 9, 1151–1157 (2003).
Tripp, R.A., Topham, D.J., Watson, S.R. & Doherty, P.C. Bone marrow can function as a lymphoid organ during a primary immune response under conditions of disrupted lymphocyte trafficking. J. Immunol. 158, 3716–3720 (1997).
Bienenstock, J., Rudzik, O., Clancy, R.L. & Perey, D.Y. Bronchial lymphoid tissue. Adv. Exp. Med. Biol. 45, 47–56 (1974).
Delventhal, S., Hensel, A., Petzoldt, K. & Pabst, R. Effects of microbial stimulation on the number, size and activity of bronchus-associated lymphoid tissue (BALT) structures in the pig. Int. J. Exp. Path. 73, 351–357 (1992).
Tshering, T. & Pabst, R. Bronchus associated lymphoid tissue (BALT) is not present in normal adult lung but in different diseases. Pathobiol. 68, 1–8 (2000).
Chvatchko, Y., Kosco-Vilbois, M.H., Herren, S., Lefort, J. & Bonnefoy, J.-Y. Germinal center formation and local immunoglobulin E (IgE) production in the lung after an airway antigenic challenge. J. Exp. Med. 184, 2353–2360 (1996).
Chin, R.K. et al. Lymphotoxin pathway directs thymic Aire expression. Nat. Immunol. 4, 1121–1127 (2003).
Bienenstock, J. & Johnston, N. A morphologic study of rabbit bronchial lymphoid aggregates and lymphoepithelium. Lab. Invest. 35, 343–348 (1976).
Plesch, B.E.C., van der Brugge-Gamelkoorn, G.J. & van de Ende, M.B. Development of bronchus associated lymphoid tissue (BALT) in the rat, with special reference to T and B cells. Dev. Comp. Immunol. 7, 79–84 (1983).
Harmsen, A. et al. Organogenesis of nasal associated lymphoid tissue (NALT) occurs independently of lymphotoxin-α (LTα) and retinoic acid receptor-related orphan receptor-g, but the organization of NALT is LTα-dependent. J. Immunol. 168, 986–990 (2002).
Sun, Z. et al. Requirement for RORγ in thymocyte survival and lymphoid organ development. Science 288, 2369–2373 (2000).
Matsumoto, M. et al. Role of lymphotoxin and the type 1 TNF receptor in the formation of germinal centers. Science 271, 1289–1291 (1996).
Fu, Y.-X., Huang, G., Matsumoto, M., Molina, H. & Chaplin, D.D. Independent signals regulate development of primary and secondary follicular structure in spleen and mesenteric lymph node. Proc. Natl. Acad. Sci. 94, 5739–5743 (1997).
Ngo, V.N. et al. Lymphotoxin α/β and tumor necrosis factor are required for stromal cell expression of homing chemokines in B and T cell areas of the spleen. J. Exp. Med. 189, 403–412 (1999).
Gunn, M.D. et al. Mice lacking expression of secondary lymphoid organ chemokine have defects in lymphocyte homing and dendritic cell localization. J. Exp. Med. 189, 451–460 (1999).
Nakano, H. & Gunn, M.D. Gene duplications at the chemokine locus on mouse chromosome 4: multiple strain-specific haplotypes and the deletion of secondary lymphoid-organ chemokine and EBI-1 ligand chemokine genes in the plt mutation. J. Immunol. 166, 361–369 (2001).
Hussell, T., Pennycook, A. & Openshaw, P.J. Inhibition of tumor necrosis factor reduces the severity of virus-specific lung immunopathology. Eur. J. Immunol. 31, 2566–2573 (2001).
Constant, S.L. et al. Resident lung antigen-presenting cells have the capacity to promote Th2 T cell differentiation in situ. J. Clin. Invest. 110, 1441–1448 (2002).
Chen, H.D. et al. Memory CD8+ T cells in heterologous antiviral immunity and immunopathology in the lung. Nat. Immunol. 2, 1067–1076 (2001).
Brimnes, M.K., Bonifaz, L., Steinman, R.M. & Moran, T.M. Influenza virus-induced dendritic cell maturation is associated with the induction of strong T cell immunity to a coadministered, normally nonimmunogenic protein. J. Exp. Med. 198, 133–144 (2003).
Hogg, J.C. et al. The nature of small-airway obstruction in chronic obstructive pulmonary disease. N. Engl. J. Med. 350, 2645–2653 (2004).
Xu, B. et al. Lymphocyte homing to bronchus-associated lymphoid tissue (BALT) is mediated by L-selectin/PNAd, α4β1 integrin/VCAM-1, and LFA-1 adhesion pathways. J. Exp. Med. 197, 1255–1267 (2003).
Ansel, K.M. et al. A chemokine-driven positive feedback loop organizes lymphoid follicles. Nature 406, 309–314 (2000).
Futterer, A., Mink, K., Luz, A., Kosco-Vilbois, M.H. & Pfeffer, K. The lymphotoxin b receptor controls organogenesis and affinity maturation in peripheral lymphoid tissues. Immunity 9, 59–70 (1998).
Kang, H.S. et al. Lymphotoxin is required for maintaining physiological levels of serum IgE that minimizes Th1-mediated airway inflammation. J. Exp. Med. 198, 1643–1652 (2003).
Lo, J.C. et al. Differential regulation of CCL21 in lymphoid/nonlymphoid tissues for effectively attracting T cells to peripheral tissues. J. Clin. Invest. 112, 1495–1505 (2003).
Guo, Z. et al. Cutting edge: membrane lymphotoxin regulates CD8+ T cell-mediated intestinal allograft rejection. J. Immunol. 167, 4796–4800 (2001).
Acknowledgements
This work was supported by Trudeau Institute and US National Institutes of Health grants HL69409 and HL63925 (T.D.R.), HL63925 and AI57158 (D.L.W.) and AI50844 and HL63925 (F.E.L.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Generation of BM chimeric mice. (PDF 513 kb)
Supplementary Fig. 2
Splenectomized Rorγ−/− mice rapidly generate influenza-specific CD8 responses after infection. (PDF 72 kb)
Supplementary Fig. 3
Splenectomized Rorγ−/− mice rapidly generate influenza-specific antibody responses after infection. (PDF 42 kb)
Supplementary Fig. 4
Germinal center B cells are generated in mice lacking all peripheral lymphoid organs. (PDF 83 kb)
Supplementary Fig. 5
Expression of CXCL13 and CCL21 is induced in the lung after influenza infection. (PDF 394 kb)
Rights and permissions
About this article
Cite this article
Moyron-Quiroz, J., Rangel-Moreno, J., Kusser, K. et al. Role of inducible bronchus associated lymphoid tissue (iBALT) in respiratory immunity. Nat Med 10, 927–934 (2004). https://doi.org/10.1038/nm1091
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1091
This article is cited by
-
Meningeal inflammation as a driver of cortical grey matter pathology and clinical progression in multiple sclerosis
Nature Reviews Neurology (2023)
-
The roles of tertiary lymphoid structures in chronic diseases
Nature Reviews Nephrology (2023)
-
An interferon-integrated mucosal vaccine provides pan-sarbecovirus protection in small animal models
Nature Communications (2023)
-
The case for BALT in human respiratory immunity
Nature Immunology (2023)
-
Induction of bronchus-associated lymphoid tissue is an early life adaptation for promoting human B cell immunity
Nature Immunology (2023)