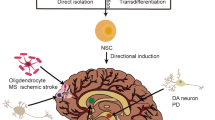
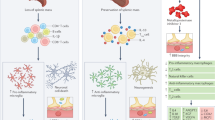
Abstract
Recent progress shows that neurons suitable for transplantation can be generated from stem cells in culture, and that the adult brain produces new neurons from its own stem cells in response to injury. These findings raise hope for the development of stem cell therapies in human neurodegenerative disorders. Before clinical trials are initiated, we need to know much more about how to control stem cell proliferation and differentiation into specific phenotypes, induce their integration into existing neural and synaptic circuits, and optimize functional recovery in animal models closely resembling the human disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Kordower, J.H. et al. Neuropathological evidence of graft survival and striatal reinnervation after the transplantation of fetal mesencephalic tissue in a patient with Parkinson's disease. N. Engl. J. Med. 332, 1118–1124 (1995).
Piccini, P. et al. Dopamine release from nigral transplants visualized in vivo in a Parkinson's patient. Nat. Neurosci. 2, 1137–1140 (1999).
Piccini, P. et al. Delayed recovery of movement-related cortical function in Parkinson's disease after striatal dopaminergic grafts. Ann. Neurol. 48, 689–695 (2000).
Lindvall, O. & Hagell, P. Clinical observations after neural transplantation in Parkinson's disease. Prog. Brain Res. 127, 299–320 (2000).
Polgar, S., Morris, M.E., Reilly, S., Bilney, B. & Sanberg, P.R. Reconstructive neurosurgery for Parkinson's disease: a systematic review and preliminary meta-analysis. Brain Res. Bull. 60, 1–24 (2003).
Freed, C.R. et al. Transplantation of embryonic dopamine neurons for severe Parkinson's disease. N. Engl. J. Med. 344, 710–719 (2001).
Olanow, C.W. et al. A double-blind controlled trial of bilateral fetal nigral transplantation in Parkinson's disease. Ann. Neurol. 54, 403–414 (2003).
Kordower, J.H. et al. Fetal nigral grafts survive and mediate clinical benefit in a patient with Parkinson's disease. Mov. Disord. 13, 383–393 (1998).
Kordower, J.H. et al. Functional fetal nigral grafts in a patient with Parkinson's disease: chemoanatomic, ultrastructural, and metabolic studies. J. Comp. Neurol. 370, 203–230 (1996).
Hagell, P. et al. Dyskinesias following neural transplantation in Parkinson's disease. Nat. Neurosci. 5, 627–628 (2002).
Ma, Y. et al. Dyskinesia after fetal cell transplantation for parkinsonism: a PET study. Ann. Neurol. 52, 628–634 (2002).
Isacson, O., Björklund, L.M. & Schumacher, J.M. Toward full restoration of synaptic and terminal function of the dopaminergic system in Parkinson's disease by stem cells. Ann. Neurol. 53, S135–S146 (2003).
Hagell, P. & Brundin, P. Cell survival and clinical outcome following intrastriatal transplantation in Parkinson disease. J. Neuropathol. Exp. Neurol. 60, 741–752 (2001).
Draper, J.S. et al. Recurrent gain of chromosomes 17q and 12 in cultured human embryonic stem cells. Nat. Biotechnol. 22, 53–54 (2004).
Amit, M. et al. Clonally derived human embryonic stem cell lines maintain pluripotency and proliferative potential for prolonged periods of culture. Dev. Biol. 227, 271–278 (2000).
Villa, A. et al. Long-term molecular and cellular stability of human neural stem cell lines. Exp. Cell. Res. 294, 559–570 (2004).
Roy, N.S. et al. Telomerase immortalization of neuronally restricted progenitor cells derived from the human fetal spinal cord. Nat. Biotechnol. 22, 297–305 (2004).
Song, H., Stevens, C.F. & Gage, F.H. Astroglia induce neurogenesis from adult neural stem cells. Nature 417, 39–44 (2002).
Wagner, J. et al. Induction of a midbrain dopaminergic phenotype in Nurr1-overexpressing neural stem cells by type 1 astrocytes. Nat. Biotechnol. 17, 653–659 (1999).
Zwaka, T.P. & Thomson, J.A. Homologous recombination in human embryonic stem cells. Nat. Biotechnol. 21, 319–321 (2003).
Zhao, M. et al. Evidence for neurogenesis in the adult mammalian substantia nigra. Proc. Natl. Acad. Sci. USA 100, 7925–7930 (2003).
Lie, D.C. et al. The adult substantia nigra contains progenitor cells with neurogenic potential. J. Neurosci. 22, 6639–6649 (2002).
Mao, L., Lau, Y.S., Petroske, E. & Wang, J.Q. Profound astrogenesis in the striatum of adult mice following nigrostriatal dopaminergic lesion by repeated MPTP administration. Dev. Brain Res. 131, 57–65 (2001).
Nowakowski, R.S. & Hayes, N.L. Stem cells: the promises and pitfalls. Neuropsychopharmacology 25, 799–804 (2001).
El-Khodor, B.F., Oo, T.F., Kholodilov, N. & Burke, R.E. Ectopic expression of cell cycle markers in models of induced programmed cell death in dopamine neurons of the rat substantia nigra pars compacta. Exp. Neurol. 179, 17–27 (2003).
Vitek, J.L. Deep brain stimulation for Parkinson's disease. A critical re-evaluation of STN versus GPi DBS. Stereotact. Funct. Neurosurg. 78, 119–131 (2002).
Winkler, C., Kirik, D., Björklund, A. & Dunnett, S.B. Transplantation in the rat model of Parkinson's disease: ectopic versus homotopic graft placement. Prog. Brain Res. 127, 233–265 (2000).
Mukhida, K., Baker, K.A., Sadi, D. & Mendez, I. Enhancement of sensorimotor behavioral recovery in hemiparkinsonian rats with intrastriatal, intranigral, and intrasubthalamic nucleus dopaminergic transplants. J. Neurosci. 21, 3521–3530 (2001).
Mendez, I. et al. Simultaneous intrastriatal and intranigral fetal dopaminergic grafts in patients with Parkinson disease: a pilot study. Report of three cases. J. Neurosurg. 96, 589–596 (2002).
Shim, J.W. et al. Enhanced in vitro midbrain dopamine neuron differentiation, dopaminergic function, neurite outgrowth, and 1-methyl-4-phenylpyridium resistance in mouse embryonic stem cells overexpressing Bcl-XL. J. Neurosci. 24, 843–852 (2004).
Kim, J.H. et al. Dopamine neurons derived from embryonic stem cells function in an animal model of Parkinson's disease. Nature 418, 50–56 (2002).
Chung, S. et al. Genetic engineering of mouse embryonic stem cells by Nurr1 enhances differentiation and maturation into dopaminergic neurons. Eur. J. Neurosci. 16, 1829–1838 (2002).
Moon, L.D., Asher, R.A., Rhodes, K.E. & Fawcett, J.W. Regeneration of CNS axons back to their target following treatment of adult rat brain with chondroitinase ABC. Nat. Neurosci. 4, 465–466 (2001).
Ourednik, J., Ourednik, V., Lynch, W.P., Schachner, M. & Snyder, E.Y. Neural stem cells display an inherent mechanism for rescuing dysfunctional neurons. Nat. Biotechnol. 20, 1103–1110 (2002).
Steece-Collier, K. et al. Embryonic mesencephalic grafts increase levodopa-induced forelimb hyperkinesia in parkinsonian rats. Mov. Disord. 18, 1442–1454 (2003).
Björklund, L.M. et al. Embryonic stem cells develop into functional dopaminergic neurons after transplantation in a Parkinson rat model. Proc. Natl. Acad. Sci. USA 99, 2344–2349 (2002).
Erdö, F. et al. Host-dependent tumorigenesis of embryonic stem cell transplantation in experimental stroke. J. Cereb. Blood Flow Metab. 23, 780–785 (2003).
Kondziolka, D. et al. Transplantation of cultured human neuronal cells for patients with stroke. Neurology 55, 565–569 (2000).
Meltzer, C.C. et al. Serial [18F]fluorodeoxyglucose positron emission tomography after human neuronal implantation for stroke. Neurosurgery 49, 586–591 (2001).
Nelson, P.T. et al. Clonal human (hNT) neuron grafts for stroke therapy: neuropathology in a patient 27 months after implantation. Am. J. Pathol. 160, 1201–1206 (2002).
Alvarez-Dolado, M. et al. Fusion of bone-marrow-derived cells with Purkinje neurons, cardiomyocytes and hepatocytes. Nature 425, 968–973 (2003).
Weimann, J.M., Johansson, C.B., Trejo, A. & Blau, H.M. Stable reprogrammed heterokaryons form spontaneously in Purkinje neurons after bone marrow transplant. Nat. Cell Biol. 5, 959–966 (2003).
Parent, J.M., Vexler, Z.S., Gong, C., Derugin, N. & Ferriero, D.M. Rat forebrain neurogenesis and striatal neuron replacement after focal stroke. Ann. Neurol. 52, 802–813 (2002).
Arvidsson, A., Collin, T., Kirik, D., Kokaia, Z. & Lindvall, O. Neuronal replacement from endogenous precursors in the adult brain after stroke. Nat. Med. 8, 963–970 (2002).
Jin, K. et al. Directed migration of neuronal precursors into the ischemic cerebral cortex and striatum. Mol. Cell Neurosci. 24, 171–189 (2003).
Jin, K., Mao, X.O., Sun, Y., Xie, L. & Greenberg, D.A. Stem cell factor stimulates neurogenesis in vitro and in vivo. J. Clin. Invest. 110, 311–319 (2002).
Yoshimura, S. et al. FGF-2 regulation of neurogenesis in adult hippocampus after brain injury. Proc. Natl. Acad. Sci. USA 98, 5874–5879 (2001).
Teramoto, T., Qiu, J., Plumier, J.C. & Moskowitz, M.A. EGF amplifies the replacement of parvalbumin-expressing striatal interneurons after ischemia. J. Clin. Invest. 111, 1125–1132 (2003).
Shingo, T., Sorokan, S.T., Shimazaki, T. & Weiss, S. Erythropoietin regulates the in vitro and in vivo production of neuronal progenitors by mammalian forebrain neural stem cells. J. Neurosci. 21, 9733–9743 (2001).
Gustafsson, E. et al. Anterograde delivery of brain-derived neurotrophic factor to striatum via nigral transduction of recombinant adeno-associated virus increases neuronal death but promotes neurogenic response following stroke. Eur. J. Neurosci. 17, 2667–2678 (2003).
Chmielnicki, E., Benraiss, A., Economides, A.N. & Goldman, S.A. Adenovirally expressed noggin and brain-derived neurotrophic factor cooperate to induce new medium spiny neurons from resident progenitor cells in the adult striatal ventricular zone. J. Neurosci. 24, 2133–2142 (2004).
Ekdahl, C.T. et al. Caspase-mediated death of newly formed neurons in the adult rat dentate gyrus following status epilepticus. Eur. J. Neurosci. 16, 1463–1471 (2002).
Ekdahl, C.T., Claasen, J.-H., Bonde, S., Kokaia, Z. & Lindvall, O. Inflammation is detrimental for neurogenesis in adult brain. Proc. Natl. Acad. Sci. USA 203, 13632–13637 (2003).
Monje, M.L., Toda, H. & Palmer, T.D. Inflammatory blockade restores adult hippocampal neurogenesis. Science 302, 1760–1765 (2003).
Nakatomi, H. et al. Regeneration of hippocampal pyramidal neurons after ischemic brain injury by recruitment of endogenous neural progenitors. Cell 110, 429–441 (2002).
Zhang, R.L., Zhang, Z.G., Zhang, L. & Chopp, M. Proliferation and differentiation of progenitor cells in the cortex and the subventricular zone in the adult rat after focal cerebral ischemia. Neuroscience 105, 33–41 (2001).
Magavi, S.S., Leavitt, B.R. & Macklis, J.D. Induction of neurogenesis in the neocortex of adult mice. Nature 405, 951–955 (2000).
Eriksson, P.S. et al. Neurogenesis in the adult human hippocampus. Nat. Med. 4, 1313–1317 (1998).
Sanai, N. et al. Unique astrocyte ribbon in adult human brain contains neural stem cells but lacks chain migration. Nature 427, 740–744 (2004).
Nunes, M.C. et al. Identification and isolation of multipotential neural progenitor cells from the subcortical white matter of the adult human brain. Nat. Med. 9, 439–447 (2003).
Palmer, T.D., Willhoite, A.R. & Gage, F.H. Vascular niche for adult hippocampal neurogenesis. J. Comp. Neurol. 425, 479–494 (2000).
Sun, Y. et al. VEGF-induced neuroprotection, neurogenesis, and angiogenesis after focal cerebral ischemia. J. Clin. Invest. 111, 1843–1851 (2003).
Zhang, H., Vutskits, L., Pepper, M.S. & Kiss, J.Z. VEGF is a chemoattractant for FGF-2-stimulated neural progenitors. J. Cell Biol. 163, 1375–1384 (2003).
Park, K.I., Teng, Y.D. & Snyder, E.Y. The injured brain interacts reciprocally with neural stem cells supported by scaffolds to reconstitute lost tissue. Nat. Biotechnol. 20, 1111–1117 (2002).
Hoehn, M. et al. Monitoring of implanted stem cell migration in vivo: a highly resolved in vivo magnetic resonance imaging investigation of experimental stroke in rat. Proc. Natl. Acad. Sci. USA 99, 16267–16272 (2002).
Pluchino, S. et al. Injection of adult neurospheres induces recovery in a chronic model of multiple sclerosis. Nature 422, 688–694 (2003).
Gates, M.A., Fricker-Gates, R.A. & Macklis, J.D. Reconstruction of cortical circuitry. Prog. Brain Res. 127, 115–156 (2000).
Fricker-Gates, R.A., Shin, J.J., Tai, C.C., Catapano, L.A. & Macklis, J.D. Late-stage immature neocortical neurons reconstruct interhemispheric connections and form synaptic contacts with increased efficiency in adult mouse cortex undergoing targeted neurodegeneration. J. Neurosci. 22, 4045–4056 (2002).
Nógrádi, A. & Vrbová, G. Improved motor function of denervated rat hindlimb muscles induced by embryonic spinal cord grafts. Eur. J. Neurosci. 8, 2198–2203 (1996).
Clowry, G., Sieradzan, K. & Vrbová, G. Transplants of embryonic motoneurones to adult spinal cord: survival and innervation abilities. Trends Neurosci. 14, 355–357 (1991).
Garbuzova-Davis, S. et al. Positive effect of transplantation of hNT neurons (NTera 2/D1 cell-line) in a model of familial amyotrophic lateral sclerosis. Exp. Neurol. 174, 169–180 (2002).
Garbuzova-Davis, S. et al. Intravenous administration of human umbilical cord blood cells in a mouse model of amyotrophic lateral sclerosis: distribution, migration, and differentiation. J. Hematother. Stem Cell Res. 12, 255–270 (2003).
Kerr, D.A. et al. Human embryonic germ cell derivatives facilitate motor recovery of rats with diffuse motor neuron injury. J. Neurosci. 23, 5131–5140 (2003).
Dunnett, S.B., Nathwani, F. & Björklund, A. The integration and function of striatal grafts. Prog. Brain Res. 127, 345–380 (2000).
Kendall, A.L. et al. Functional integration of striatal allografts in a primate model of Huntington's disease. Nat. Med. 4, 727–729 (1998).
Palfi, S. et al. Fetal striatal allografts reverse cognitive deficits in a primate model of Huntington disease. Nat. Med. 4, 963–966 (1998).
Freeman, T.B. et al. Transplanted fetal striatum in Huntington's disease: phenotypic development and lack of pathology. Proc. Natl. Acad. Sci. USA 97, 13877–13882 (2000).
Bachoud-Levi, A.C. et al. Motor and cognitive improvements in patients with Huntington's disease after neural transplantation. Lancet 356, 1975–1979 (2000).
Hauser, R.A. et al. Bilateral human fetal striatal transplantation in Huntington's disease. Neurology 58, 687–695 (2002).
Gaura, V. et al. Striatal neural grafting improves cortical metabolism in Huntington's disease patients. Brain 127, 65–72 (2004).
Dinsmore, J. et al. Embryonic stem cells differentiated in vitro as a novel source of cells for transplantation. Cell Transplant. 5, 131–143 (1996).
Lescaudron, L., Unni, D. & Dunbar, G.L. Autologous adult bone marrow stem cell transplantation in an animal model of Huntington's disease: behavioral and morphological outcomes. Int. J. Neurosci. 113, 945–956 (2003).
Fricker, R.A. et al. Site-specific migration and neuronal differentiation of human neural progenitor cells after transplantation in the adult rat brain. J. Neurosci. 19, 5990–6005 (1999).
Englund, U., Björklund, A. & Wictorin, K. Migration patterns and phenotypic differentiation of long-term expanded human neural progenitor cells after transplantation into the adult rat brain. Dev. Brain Res. 134, 123–141 (2002).
Curtis, M.A. et al. Increased cell proliferation and neurogenesis in the adult human Huntington's disease brain. Proc. Natl. Acad. Sci. USA 100, 9023–9027 (2003).
Kawasaki, H. et al. Induction of midbrain dopaminergic neurons from ES cells by stromal cell-derived inducing activity. Neuron 28, 31–40 (2000).
Barberi, T. et al. Neural subtype specification of fertilization and nuclear transfer embryonic stem cells and application in parkinsonian mice. Nat. Biotechnol. 21, 1200–1207 (2003).
Kawasaki, H. et al. Generation of dopaminergic neurons and pigmented epithelia from primate ES cells by stromal cell-derived inducing activity. Proc. Natl. Acad. Sci. USA 99, 1580–1585 (2002).
Carvey, P.M. et al. A clonal line of mesencephalic progenitor cells converted to dopamine neurons by hematopoietic cytokines: a source of cells for transplantation in Parkinson's disease. Exp. Neurol. 171, 98–108 (2001).
Storch, A. et al. Long-term proliferation and dopaminergic differentiation of human mesencephalic neural precursor cells. Exp. Neurol. 170, 317–325 (2001).
Studer, L. et al. Enhanced proliferation, survival, and dopaminergic differentiation of CNS precursors in lowered oxygen. J. Neurosci. 20, 7377–7383 (2000).
Studer, L., Tabar, V. & McKay, R.D. Transplantation of expanded mesencephalic precursors leads to recovery in parkinsonian rats. Nat. Neurosci. 1, 290–295 (1998).
Yan, J., Studer, L. & McKay, R.D. Ascorbic acid increases the yield of dopaminergic neurons derived from basic fibroblast growth factor expanded mesencephalic precursors. J. Neurochem. 76, 307–311 (2001).
Daadi, M.M. & Weiss, S. Generation of tyrosine hydroxylase-producing neurons from precursors of the embryonic and adult forebrain. J. Neurosci. 19, 4484–4497 (1999).
Jiang, Y. et al. Neuroectodermal differentiation from mouse multipotent adult progenitor cells. Proc. Natl. Acad. Sci. USA 100, 11854–11860 (2003).
Jiang, Y. et al. Pluripotency of mesenchymal stem cells derived from adult marrow. Nature 418, 41–49 (2002).
Li, Y. et al. Intracerebral transplantation of bone marrow stromal cells in a 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine mouse model of Parkinson's disease. Neurosci. Lett. 316, 67–70 (2001).
Chiba, S., Iwasaki, Y., Sekino, H. & Suzuki, N. Transplantation of motoneuron-enriched neural cells derived from mouse embryonic stem cells improves motor function of hemiplegic mice. Cell Transplant. 12, 457–468 (2003).
Mujtaba, T. et al. Lineage-restricted neural precursors can be isolated from both the mouse neural tube and cultured ES cells. Dev. Biol. 214, 113–127 (1999).
Renoncourt, Y., Carroll, P., Filippi, P., Arce, V. & Alonso, S. Neurons derived in vitro from ES cells express homeoproteins characteristic of motoneurons and interneurons. Mech. Dev. 79, 185–197 (1998).
Wichterle, H., Lieberam, I., Porter, J.A. & Jessell, T.M. Directed differentiation of embryonic stem cells into motor neurons. Cell 110, 385–397 (2002).
Kalyani, A., Hobson, K. & Rao, M.S. Neuroepithelial stem cells from the embryonic spinal cord: isolation, characterization, and clonal analysis. Dev. Biol. 186, 202–223 (1997).
Kalyani, A.J., Piper, D., Mujtaba, T., Lucero, M.T. & Rao, M.S. Spinal cord neuronal precursors generate multiple neuronal phenotypes in culture. J. Neurosci. 18, 7856–7868 (1998).
Mayer-Proschel, M., Kalyani, A.J., Mujtaba, T. & Rao, M.S. Isolation of lineage-restricted neuronal precursors from multipotent neuroepithelial stem cells. Neuron 19, 773–785 (1997).
Li, R. et al. Motoneuron differentiation of immortalized human spinal cord cell lines. J. Neurosci. Res. 59, 342–352 (2000).
Wu, P. et al. Region-specific generation of cholinergic neurons from fetal human neural stem cells grafted in adult rat. Nat. Neurosci. 5, 1271–1278 (2002).
Willing, A.E. et al. hNT neurons delay onset of motor deficits in a model of amyotrophic lateral sclerosis. Brain Res. Bull. 56, 525–530 (2001).
Garbuzova-Davis, S. et al. Intraspinal implantation of hNT neurons into SOD1 mice with apparent motor deficit. Amyotroph. Lateral Scler. Other Motor Neuron Disord. 2, 175–180 (2001).
Acknowledgements
We thank Bengt Mattsson for illustrations. Our own work was supported by grants from the Swedish Research Council, Swedish Foundation for Strategic Research, the Kock, Söderberg, Crafoord and Segerfalk Foundations, EU (BIO04-CT98-0530 and QLK3-CT-2001-02120), Foundation La Caixa, and Spanish Ministry of Science and Technology (MCYT SAF2001-1038-C02-02). The Lund Stem Cell Center is supported by a Center of Excellence grant in life sciences from the Swedish Foundation for Strategic Research.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Lindvall, O., Kokaia, Z. & Martinez-Serrano, A. Stem cell therapy for human neurodegenerative disorders–how to make it work. Nat Med 10 (Suppl 7), S42–S50 (2004). https://doi.org/10.1038/nm1064
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1064
This article is cited by
-
Functional nanoparticle-enabled non-genetic neuromodulation
Journal of Nanobiotechnology (2023)
-
KLF2 Regulates Neural Differentiation of Dental Pulp-derived Stem Cells by Modulating Autophagy and Mitophagy
Stem Cell Reviews and Reports (2023)
-
Mesenchymal stromal cell treatment attenuates repetitive mild traumatic brain injury-induced persistent cognitive deficits via suppressing ferroptosis
Journal of Neuroinflammation (2022)
-
Cell membrane-camouflaged inorganic nanoparticles for cancer therapy
Journal of Nanobiotechnology (2022)
-
Neurospheres: a potential in vitro model for the study of central nervous system disorders
Molecular Biology Reports (2021)