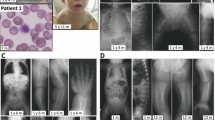
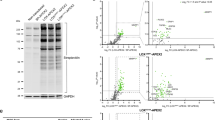
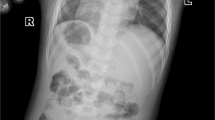
Abstract
The congenital disorders of glycosylation (CDG) are characterized by defects in N-linked glycan biosynthesis that result from mutations in genes encoding proteins directly involved in the glycosylation pathway. Here we describe two siblings with a fatal form of CDG caused by a mutation in the gene encoding COG-7, a subunit of the conserved oligomeric Golgi (COG) complex. The mutation impairs integrity of the COG complex and alters Golgi trafficking, resulting in disruption of multiple glycosylation pathways. These cases represent a new type of CDG in which the molecular defect lies in a protein that affects the trafficking and function of the glycosylation machinery.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Varki, A. Biological roles of oligosaccharides: all of the theories are correct. Glycobiology 3, 97–130 (1993).
Storrie, B. et al. Recycling of Golgi-resident glycosyltransferases through the ER reveals a novel pathway and provides an explanation for nocodazole-induced Golgi scattering. J. Cell Biol. 143, 1505–1521 (1998).
Marquardt, T. & Denecke, J. Congenital disorders of glycosylation: review of their molecular bases, clinical presentations and specific therapies. Eur. J. Pediatr. 162, 359–379 (2003).
Grunewald, S., Matthijs, G. & Jaeken, J. Congenital disorders of glycosylation: a review. Pediatr. Res. 52, 618–624 (2002).
Schauer, R. Biosynthesis and function of N- and O-substituted sialic acids. Glycobiology 1, 449–452 (1991).
Kim, S., Miura, Y., Etchison, J.R. & Freeze, H.H. Intact Golgi synthesize complex branched O-linked chains on glycoside primers: evidence for the functional continuity of seven glycosyltransferases and three sugar nucleotide transporters. Glycoconj. J. 18, 623–633 (2001).
Lippincott-Schwartz, J., Roberts, T.H. & Hirschberg, K. Secretory protein trafficking and organelle dynamics in living cells. Annu. Rev. Cell Dev. Biol. 16, 557–589 (2000).
Kingsley, D.M., Kozarsky, K.F., Segal, M. & Krieger, M. Three types of low density lipoprotein receptor-deficient mutant have pleiotropic defects in the synthesis of N-linked, O-linked, and lipid-linked carbohydrate chains. J. Cell Biol. 102, 1576–1585 (1986).
Ungar, D. et al. Characterization of a mammalian Golgi-localized protein complex, COG, that is required for normal Golgi morphology and function. J. Cell Biol. 157, 405–415 (2002).
Whyte, J.R.C. & Munro, S. Vesicle tethering complexes in membrane traffic. J. Cell Sci. 115, 2627–2637 (2002).
Podos, S.D., Reddy, P., Ashkenas, J. & Krieger, M. LDLC encodes a Brefeldin-A sensitive, peripheral Golgi protein required for normal Golgi function. J. Cell Biol. 127, 679–691 (1994).
Chatterton, J.E. et al. Expression cloning of LDLB, a gene essential for normal Golgi function and assembly of the ldlCp complex. Proc. Natl. Acad. Sci. USA 96, 915–920 (1999).
Walter, D.M., Paul, K.S. & Waters, M.G. Purification and characterization of a novel 13 S hetero-oligomeric protein complex that stimulates in vitro Golgi transport J. Biol. Chem. 273, 29565–29576 (1998).
Loh, E. & Hong, W. Sec34 is implicated in traffic from the endoplasmic reticulum to the Golgi and exists in a complex with GTC–90 and ldlBp. J. Biol. Chem. 277, 21955–21961 (2002).
Whyte, J.R. & Munro, S. The Sec34/35 Golgi transport complex is related to the exocyst, defining a family of complexes involved in multiple steps of membrane traffic. Dev. Cell 1, 527–537 (2001).
Farkas, R.M. et al. The Drosophila COG5 homologue is required for cytokinesis, cell elongation, and assembly of specialized Golgi architecture during spermatogenesis. Mol. Biol. Cell 14, 190–200 (2003).
Suvorova, E.S., Kurten, R.C. & Lupashin, V.V. Identification of a human orthologue of Sec34p as a component of the cis-Golgi vesicle tethering machinery. J. Biol. Chem. 276, 22810–22818 (2001).
Freeze, H.H. Update and perspectives on congenital disorders of glycosylation. Glycobiology 11, 129R–143R (2001).
Wopereis, S. et al. Apolipoprotein C–III isofocusing in the diagnosis of genetic defects in O-glycan biosynthesis. Clin. Chem. 49, 1839–1845 (2003).
Oka, T., Ungar, D., Hughson, F.M. & Krieger, M. The COG and COPI complexes interact to control the abundance of GEARs, a subset of Golgi integral membrane proteins. Mol. Biol. Cell 15, 2423–2435 (2004).
Olkkonen, V.M. & Ikonen, E. Genetic defects of intracellular-membrane transport. N. Engl. J. Med. 343, 1095–1104 (2000).
Nichols, W.C. et al. Mutations in the ER-Golgi intermediate compartment protein ERGIC-53 cause combined deficiency of coagulation factors V and VIII. Cell 93, 61–70 (1998).
Gedeon, A.K. et al. Identification of the gene (SEDL) causing X-linked spondyloepiphyseal dysplasia tarda. Nat. Genet. 22, 400–404 (1999).
Zhang, B. et al. Bleeding due to disruption of a cargo-specific ER-to-Golgi transport complex. Nat. Genet. 34, 220–225 (2003).
Blixt, O. et al. Efficient chemoenzymatic synthesis of O-linked sialyl oligosaccharides. J. Am. Chem. Soc. 124, 5739–5746 (2002).
Steet, R.A., Melancon, P. & Kuchta, R.D. 3′-Azidothymidine potentially inhibits the biosynthesis of highly branched N-linked oligosaccharides and poly–N-acetyllactosamine chains in cells. J. Biol. Chem. 275, 26812–26820 (2000).
Ory, D.S., Neugeboren, B.A. & Mulligan, R.C. A stable human-derived packaging cell line for production of high titer retrovirus/vesicular stomatitis virus G pseudotypes. Proc. Natl. Acad. Sci. USA 93, 11400–11406 (1996).
Ju, T., Cummings, R.D. & Canfield, W.M. Purification, characterization, and subunit structure of rat Core1β1,3-galactosyltransferase. J. Biol. Chem. 277, 169–177 (2002).
Gillespie, W., Kelm, S. & Paulson, J.C. Cloning and expression of the Galβ1,3GalNAc α2,3–sialyltransferase. J. Biol. Chem. 267, 21004–21010 (1992).
Acknowledgements
We thank D. Ungar, T. Oka and V. Lupashin for purified COG antibodies; M. Ichikawa for technical support; B. Hayes and J. Kawakami for sugar nucleotide pool measurements; M. Fukuda for ST3Gal-I–GFP construct; D. Ory for assistance in preparing the retrovirus; and J. Sijstermans and S. van der Meer for clinical evaluation of the patients. This work was supported by grants RO1 DK55615 (H.H.F.), R37 CA08759 (S.K.), GM59115 (M.K.), U54 GM62116 and R24 GM61894, and by the March of Dimes Foundation (H.H.F.)
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Wu, X., Steet, R., Bohorov, O. et al. Mutation of the COG complex subunit gene COG7 causes a lethal congenital disorder. Nat Med 10, 518–523 (2004). https://doi.org/10.1038/nm1041
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1041
This article is cited by
-
Cardiac complications of congenital disorders of glycosylation (CDG): a systematic review of the literature
Journal of Inherited Metabolic Disease (2017)
-
Liver involvement in congenital disorders of glycosylation (CDG). A systematic review of the literature
Journal of Inherited Metabolic Disease (2017)
-
Golgi post‐translational modifications and associated diseases
Journal of Inherited Metabolic Disease (2015)
-
Skin manifestations in CDG
Journal of Inherited Metabolic Disease (2014)