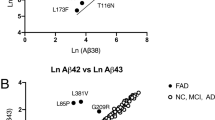
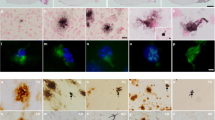
Abstract
Mutations in the presenilin 1 (PS1) and presenilin 2 (PS2) genes can cause Alzheimer's disease in affected members of the majority of early-onset familial Alzheimer's disease (FAD) pedigrees1–7. PS1 encodes an ubiquitously expressed, eight transmembrane protein1,8–11. PS1 is endoproteolytically processed to an amino-terminal derivative (∼27–28 kDa) and a carboxy-terminal derivative (∼17–18 kDa). These polypeptides accumulate to saturable levels in the brains of transgenic mice, independent of the expression of PS1 holoprotein12. We now document that, in the brains of transgenic mice, the absolute amounts of accumulated N- and C-terminal derivatives generated from the FAD-linked PS1 variants in which Glu replaces Ala at codon 246 (A246E) or Leu replaces Met at codon 146 (M146L) accumulate to a significantly higher degree (∼40–50%) than the fragments derived from wild-type PS1. Moreover, the FAD-linked ΔE9 PS1 variant, a polypeptide that is not subject to endoproteolytic cleavage in vivo, also accumulates in greater amounts than the fragments generated from wild-type human PS1. Thus, the metabolism of PS1 variants linked to FAD is fundamentally different from that of wild-type PS1 in vivo.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Sherrington, R. et. al. Cloning of a gene bearing missense mutations in early-onset familial Alzheimer's disease. Nature 375, 754–760 (1995).
Alzheimar's Disease Collaborative Group. The structure of the presenilin 1 (S182) gene and identification of six novel mutations in early onset AD families. Nature Genet. 11, 219–222 (1995).
Rogaev, E.I. et al. Familial Alzheimer's disease in kindreds with missense mutations in a gene on chromosome 1 related to the Alzheimer's disease type 3 gene. Nature 376, 775–778 (1995).
Levy-Lahad, E. et al. A familial Alzheimer's disease locus on chromosome 1. Science 269, 970–973 (1995).
Levy-Lahad, E. et al. Candidate gene for the chromosome 1 familial Alzheimer's disease locus. Science 269, 973–977 (1995).
Perez-tur, J. et al. A mutation in Alzheimer's disease destroying a splice acceptor site in the presenilin-1 gene. Neuroreport 7, 297–301 (1995).
Campion, D. et al. Mutations of the presenilin 1 gene in families with early-onset Alzheimer's disease. Hum. Mol. Genet. 4, 2373–2377 (1995).
Kovacs, D.M. et al. Alzheimer-associated presenilins 1 and 2: Neuronal expression in brain and localization to intracellular membranes in mammalian cells. Nature Med. 2, 224–229 (1996).
Lee, M.K. et al. Expression of presenilin 1 and 2 (PS1 and PS2) in human and murine tissues. J. Neurosci. 16, 75137525 (1996).
Li, X. & Greenwald, I. Membrane topology of the C. elegans SEL-12 presenilin. Neuron 17, 1015–1021 (1996).
Doan, A. et al. Protein topology of presenilin 1. Neuron 17, 1023–1030 (1996).
Thinakaran, G. et al. Endoproteolysis of presenilin 1 and accumulation of processed derivatives in vitro and in vivo. Neuron 17, 181–190 (1996).
Lah, J.J. et al. Light and electron microscopic localization of presenilin-1 in primate brain. J. Neurosci. 19, 1971–1980 (1997).
Levitan, D. et al. Assessment of normal and mutant human presenilin function in Caenorhabditis elegans. Proc. Natl. Acad. Sci. USA 93, 14940–14944 (1966).
Borchelt, D.R. et al. Familial Alzheimer's disease-linked presenilin 1 variants elevate Aβ1 - 42/1 -40 ratio in vitro and in vivo. Neuron 17, 1005–1013.
Duff, K. et al. Increased amyloid-β42(43) in brains of mice expressing mutant presenilin 1. Nature 383, 710–713.
Citron, M. et al. Mutant presenilins of Alzheimer's disease increase production of 42-residue amyloid β-protein in both transfected cells and transgenic mice. Nature Med. 3 67–72.
Iwatsubo, T. et al. Visualization of Aβ42(43) and Aβ40 in senile plaques with end-specific Aβ monoclonals: Evidence that an initially deposited species is Aβ42(43). Neuron 13, 45–53 (1994).
Lemere, C.A. et al. The E280A presenilin 1 Alzheimer mutation produces increased Aβ42 deposition and severe cerebellar pathology. Nature Med. 2, 1146–1150 (1996).
Lemere, C.A. et al. Sequence of deposition of heterogeneous amyloid β-peptides and ApoE in Down syndrome: Implications of initial events in amyloid plaque formation. Neurobiol. Disease 3, 16–32 (1996).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lee, M., Borchelt, D., Kim, G. et al. Hyperaccumulation of FAD-linked presenilin 1 variants in vivo. Nat Med 3, 756–760 (1997). https://doi.org/10.1038/nm0797-756
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nm0797-756
This article is cited by
-
Blockade of adenosine A2A receptors reverses early spatial memory defects in the APP/PS1 mouse model of Alzheimer’s disease by promoting synaptic plasticity of adult-born granule cells
Alzheimer's Research & Therapy (2023)
-
Murine Aβ over-production produces diffuse and compact Alzheimer-type amyloid deposits
Acta Neuropathologica Communications (2015)
-
Protection of Radial Glial-Like Cells in the Hippocampus of APP/PS1 Mice: a Novel Mechanism of Memantine in the Treatment of Alzheimer’s Disease
Molecular Neurobiology (2015)
-
Potent amyloidogenicity and pathogenicity of Aβ43
Nature Neuroscience (2011)
-
APP Transgenic Mice: Their Use and Limitations
NeuroMolecular Medicine (2011)