Abstract
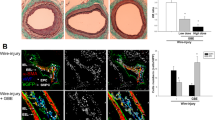
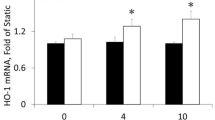
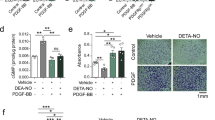
Heme oxygenase (HO-1, encoded by Hmox1) is an inducible protein activated in systemic inflammatory conditions by oxidant stress. Vascular injury is characterized by a local reparative process with inflammatory components, indicating a potential protective role for HO-1 in arterial wound repair. Here we report that HO-1 directly reduces vasoconstriction and inhibits cell proliferation during vascular injury. Expression of HO-1 in arteries stimulated vascular relaxation, mediated by guanylate cyclase and cGMP, independent of nitric oxide. The unexpected effects of HO-1 on vascular smooth muscle cell growth were mediated by cell-cycle arrest involving p21Cip1. HO-1 reduced the proliferative response to vascular injury in vivo; expression of HO-1 in pig arteries inhibited lesion formation and Hmox1−/− mice produced hyperplastic arteries compared with controls. Induction of the HO-1 pathway moderates the severity of vascular injury by at least two adaptive mechanisms independent of nitric oxide, and is a potential therapeutic target for diseases of the vasculature.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Maines, M.D. The heme oxygenase system: a regulator of second messenger gases. Annu. Rev. Pharmacol. Toxicol. 37, 517–554 (1997).
Soares, M.P. et al. Expression of heme oxygenase-1 can determine cardiac xenografts survival. Nature Med. 4, 1073–1077 (1998).
Hancock, W.W., Buelow, R., Sayegh, M.H. & Turka, L.A. Antibody-induced transplant arteriosclerosis is prevented by graft expression of anti-oxidant and anti-apoptotic genes. Nature Med. 4, 1392–1396 (1998).
Verma, A., Hirsch, D.J., Glatt, C.E., Ronnet, G.V. & Snyder, S.H. Carbon monoxide: putative neural messenger. Science 259, 381–384 (1993).
McCoubrey, W.K., Jr ., Huang, T.J. & Maines, M.D. Isolation and characterization of a cDNA from the rat brain that encodes hemoprotein heme oxygenase-3. Eur. J. Biochem. 247, 725–732 (1997).
Brouard, S. et al. Carbon monoxide generated by heme oxygenase 1 suppresses endothelial cell apoptosis. J. Exp. Med. 192, 1015–1026 (2000).
Otterbein, L.E. et al. Carbon monoxide has anti-inflammatory effects involving the mitogen-activated protein kinase pathway. Nature Med. 6, 422–428 (2000).
Otterbein, L.E., Mantell, L.L. & Choi, A.M. Carbon monoxide provides protection against hyperoxic lung injury. Am. J. Physiol. 276, 688–694 (1999).
Dore, S. et al. Bilirubin, formed by activation of heme oxygenase-2, protects neurons against oxidative stress injury. Proc. Natl. Acad. Sci. USA 96, 2445–2450 (1999).
Morita, T. et al. Smooth muscle cell-derived carbon monoxide is a regulator of vascular cGMP. Proc. Natl. Acad. Sci. USA 92, 1479 (1995).
Ohno, T. et al. Gene therapy for vascular smooth muscle cell proliferation after arterial injury. Science 265, 781–784 (1994).
Tanner, F.C. et al. Expression of cyclin-dependent kinase inhibitors in vascular disease. Circ. Res. 82, 396–403 (1998).
Yang, Z,-Y. et al. Role of the p21 cyclin-dependent kinase inhibitor in limiting intimal cell proliferation in response to arterial injury. Proc. Natl. Acad. Sci. USA 93, 7905–7910 (1996).
Morita, T. & Kourembanas, S. Endothelial cell expression of vasoconstrictors and growth factors is regulated by smooth muscle cell-derived carbon monoxide. J. Clin. Invest. 96, 2676–2682 (1995).
Marks, G., Brian, J., Nakatsu, K. & McLaughlin, B. Does carbon monoxide have a physiological function? Trend Pharmacol. Sci. 12, 185–188 (1991).
Sammut, I.A. et al. Carbon monoxide is a major contributor to the regulation of vascular tone in aortas expressing high levels of haeme oxygenase-1. Br. J. Pharmacol. 125, 1437–1444 (1998).
Suematsu, M. et al. Carbon monoxide: an endogenous modulator of sinusoidal tone in the perfused rat liver. J. Clin. Invest. 96, 2431–2437 (1995).
Foresti, R., Clark, J.E., Green, C.J. & Motterlini, R. Thiol compounds interact with nitric oxide in regulating heme oxygenase-1 induction in endothelial cells. Involvement of superoxide and peroxynitrite anions. J. Biol. Chem. 272, 18411–18417 (1997).
Durante, W., Kroll, M.H., Christodoulides, N., Peyton, K.J. & Schafer, A. Nitric oxide induces heme oxygenase-1 gene expression and carbon monoxide in production in vascular smooth muscle cells. Circ. Res. 80, 557–564 (1997).
Hartsfield, C.L., Alam, J., Cook, J.L. & Choi, A.M. Regulation of heme oxygenase-1 gene expression in vascular smooth muscle cells by nitric oxide. Am. J. Physiol. 273, L980–L988 (1997).
Yet, S.F. et al. Induction of heme oxygenase-1 expression in vascular smooth muscle cells. A link to endotoxic shock. J. Biol. Chem. 272, 4295–4301 (1997).
Motterlini, R., Foresti, R., Intaglietta, M. & Winslow, R.M. NO-mediated activation of heme oxygenase: endogenous cytoprotection against oxidative stress to endothelium. Am. J. Physiol. 270, H107–114 (1996).
Abraham, N.G. et al. Transfection of the human heme oxygenase gene into rabbit coronary microvessel endothelial cells: protective effect against heme and hemoglobin toxicity. Proc. Natl. Acad. Sci USA 92, 6798–6802 (1995).
Morita, T., Mitsialis, S.A., Koike, H., Liu, Y.X. & Kourembanas, S. Carbon monoxide controls the proliferation of hypoxic vascular smooth muscle cells. J. Biol. Chem. 272, 32804–32809 (1997).
Tanner, F.C. et al. Nitric oxide modulates expression of cell cycle regulatory proteins. A cytostatic strategy for inhibition of human vascular smooth muscle cell proliferation. Circulation 101, 1982–1989 (2000).
Lee, P.J., Alam, J., Wiegan, G.W. & Choi, A.M. Overexpression of heme oxygenase-1 in human pulmonary epithelial cells results in cell growth arrest and increased resistance to hyperoxia. Proc. Natl. Acad. Sci. USA 93, 10393–10398 (1996).
Wagner, C., Durante, W., Christodoulides, N., Hellums, J. & Schafer, A. Hemodynamic forces induce the expression of heme oxygenase in cultured vascular smooth muscle cells. J. Clin. Invest. 100, 589–596 (1997).
Wang, L.-J., Lee, T.-S., Lee, F.-Y., Pai, R.-C. & Chau, L.-Y. Expression of heme oxygenase-1 in atherosclerotic lesions. Am. J. Pathol. 152, 711–720 (1998).
Yet, S.F. et al. Hypoxia induces severe right ventricular dilatation and infarction in heme oxygenase-1 null mice. J. Clin. Invest. 103, 23–29 (1999).
Brugarolas, J., Bronson, R.T. & Jacks, T. p21 is a critical CDK2 regulator essential for proliferation control in Rb-deficient cells. J. Cell Biol. 141, 503–514 (1998).
Brugarolas, J. et al. Radiation-induced cell cycle arrest compromised by p21 deficiency. Nature 377, 552–557 (1995).
Gunther, S., Alexander R.W., Atkinson W.J. & Gimbrone M.A. Functional angiotensin II receptors in cultured vascular smooth muscle cells. J. Cell Biol. 192, 289–298 (1982).
Layne, M.D. et al. Aortic carboxypeptidase-like protein, a novel protein with discoidin and carboxypeptidase-like domains, is up-regulated during vascular smooth muscle cell differentiation. J. Biol. Chem. 273, 15654–15660 (1998).
Yoshinaga, T., Sassa, S. & Kappas, A. Purification and properties of bovine spleen heme oxygenase. Amino acid composition and sites of action of inhibitors of heme oxidation. J. Biol. Chem. 257, 7778–7785 (1982).
Wolin, M.S. et al. Guanylate cyclase from bovine lung. A kinetic analysis of the purified soluble enzyme by protoporphyrin IX, heme, and nitrosyl-heme. J. Biol. Chem. 257, 13312–13318 (1982).
Zakhary, R. et al. Heme oxygenase 2: Endothelial and neuronal localization and role in endothelium-dependent relaxation. Proc. Natl. Acad. Sci. USA 93, 795–798 (1996).
Acknowledgements
H.J.D. was supported by a grant from NATO (NWO) and the Intercardiologisch Instituut Nederland (ICIN).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Duckers, H., Boehm, M., True, A. et al. Heme oxygenase-1 protects against vascular constriction and proliferation. Nat Med 7, 693–698 (2001). https://doi.org/10.1038/89068
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/89068
This article is cited by
-
Heme Oxygenase 1 in Vertebrates: Friend and Foe
Cell Biochemistry and Biophysics (2022)
-
A tough nitric oxide-eluting hydrogel coating suppresses neointimal hyperplasia on vascular stent
Nature Communications (2021)
-
Activation of heme oxygenase-1 by Ginkgo biloba extract differentially modulates endothelial and smooth muscle-like progenitor cells for vascular repair
Scientific Reports (2019)
-
Macrophages are exploited from an innate wound healing response to facilitate cancer metastasis
Nature Communications (2018)
-
Genetic ablation of Bach1 gene enhances recovery from hyperoxic lung injury in newborn mice via transient upregulation of inflammatory genes
Pediatric Research (2017)