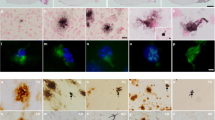
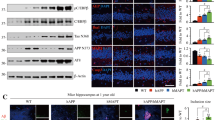
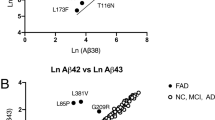
Abstract
Familial Alzheimer disease mutations of presenilin 1 (PS-1) enhance the generation of Aβ1–42, indicating that PS-1 is involved in amyloidogenesis. However, PS-1 transgenic mice have failed to show amyloid plaques in their brains. Because PS-1 mutations facilitate apoptotic neuronal death in vitro , we did careful quantitative studies in PS-1 transgenic mice and found that neurodegeneration was significantly accelerated in mice older than 13 months (aged mice) with familial Alzheimer disease mutant PS-1, without amyloid plaque formation. However, there were significantly more neurons containing intracellularly deposited Aβ42 in aged mutant transgenic mice. Our data indicate that the pathogenic role of the PS-1 mutation is upstream of the amyloid cascade.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Sherrington, R. et al. Cloning of a gene bearing missense mutations in early-onset familial Alzheimer's disease. Nature 375, 754–760 (1995).
Cruts, M., Van Broeckhoven, C. Presenilin mutations in Alzheimer's disease. Hum. Mutat. 11, 183 –190 (1998).
Kamimura, K. et al. Familial Alzheimer's disease genes in Japanese. J. Neurol. Sci. 160, 76–81 (1998).
Scheuner, D. et al. The amyloid β-protein deposited in the senile plaques of Alzheimer's disease is increased in vivo by the presenilin 1 and 2 and APP mutations linked to familial Alzheimer's disease. Nature Med. 2, 864–870 ( 1996).
Lemere, C. A. et al. The E280A presenilin-1 Alzheimer mutation produces increased Aβ42 deposition and severe cerebellar pathology. Nature Med. 2, 1146–1150 ( 1996).
Duff, K. et al. Increased amyloid-β42(43) in brains of mice expressing mutant presenilin 1. Nature 383,710– 713 (1996).
Borchelt, D. R. et al. Familial Alzheimer's disease-linked presenilin 1 variants elevate Aβ1-42/1-40 ratio in vitro and in vivo. Neuron 17, 1005–1013 ( 1996).
Citron, M. et al. Mutant presenilins of Alzheimer's disease increase production of 42-residue amyloid β-protein in both transfected cells and transgenic mice. Nature Med. 3, 67– 72 (1997).
Guo, Q. et al. Expression of mutant PS1 in PC12 cells increases their susceptibility to apoptosis by trophic factor withdrawal or by Aβ administration in vitro. J. Neurosci. 17, 4212– 4222 (1997).
Wolozin, B. et al. Participation of presenilin 2 in apoptosis: Enhanced basal activity conferred by an Alzheimer mutation. Science 274, 1710–1713 (1996).
Gallyas, F. et al. Four modified silver methods for thick sections of formaldehyde-fixed perikarya of all neurons, microglia cells and capillaries. J. Neurosci. Methods 50, 159–164 ( 1993).
Gallyas, F. et al. Formation of "dark" (argyrophilic) neurons of various origin proceeds with a common mechanism of biophysical nature (a novel hypothesis). Acta Neuropathol. 83, 504– 509 (1992).
Hardy, J. Amyloid, the presenilins and Alzheimer's disease. Trends Neurosci. 20, 154–159 ( 1997).
Neve, R. L., Robakis, N. K. Alzheimer's disease: a re-examination of the amyloid hypothesis. Trends Neurosci. 21, 15–19 (1998).
Games, D. et al. Alzheimer-type neuropathology in transgenic mice overexpressing V717F β-amyloid precursor protein. Nature 373, 523–527 (1995).
Irizarry, M. C. et al. APPSw transgenic mice develop age-related Aβ deposits and neuropil abnormalities, but no neuronal loss in CA1. J. Neuropathol. Exp. Neurol. 56, 965–973 (1997).
Borchelt, D. R. et al. Accelerated amyloid deposition in the brains of transgenic mice coexpressing mutant presenilin1 and amyloid precursor protein. Neuron 19, 939–945 ( 1997).
Holcomb, L. et al. Accelerated Alzheimer-type phenotype in trasgenic mice carrying both mutant amyloid precursor protein and presenilin 1 transgene. Nature Med. 4, 97–100 ( 1998).
Staufenbiel, M. et al. Neuron loss in APP transgenic mice. Nature 395,755–756 (1998).
Haass, C. et al. Proteolytic processing of Alzheimer's disease associated proteins. J. Neural Transm. Suppl. 53:159–167 (1998).
Zhang, Z. et al. Destabilization of β-catenin by mutations in presenilin-1 potentiates neuronal apoptosis. Nature 395,698–702 (1998).
Geula, C. et al. Aging renders the brain vulnerable to amyloid β-protein neurotoxicity. Nature Med. 4, 827– 831 (1998).
Crook, R. et al. A variant of Alzheimer's disease with spastic paraparesis and unusual plaques due to deletion of exon 9 of presenilin 1. Nature Med. 4, 452–459 ( 1998).
Mizoguchi, K. et al. Stress induces neuronal death in the hippocampus of castrated rats. Neurosci. Lett. 138, 157– 160 (1992).
Chui, D.H. et al. Both N-terminal and C-terminal fragments of presenilin 1 colocalize with neurofibrillary tangles in neurons and dystrophic neurites of senile plaques in Alzheimer's disease. J. Neurosci. Res. 53 , 99–106 (1998).
Liu, X.Z. et al. Neuronal and glial apoptosis after traumatic spinal cord injury. J. Neurosci. 17,5395–5406 (1997).
Chui, D.H. et al. Immunohistochemical analyses of the leprosy brain show low β-amyloid but high abnormal tau deposition. Am. J. Pathol. 145 , 771–775 (1994).
Barelli, H. et al. Characterization of new polyclonal antibodies specific for 40 and 42 amino acid-long amyloid β peptides: Their use to examine the cell biology of presenilins and the immunohistochemistry of sporadic Alzheimer's disease and cerebral amyloid angiopathy cases. Mol. Med. 3, 695–707 (1997).
Koike, F. et al. Immunohistochemical study of Alzheimer's disease using antibodies to synthetic amyloid and fibronectin. J. Neurol. Sci. 85, 9–15 (1988).
Acknowledgements
The authors thank S. Naito at Chugai Pharmaceutical for his effort in coordinating this project, and J. Galvin (Allegheny University Hospital) and R.P. Friedland (Case Western Reserve University) for reading this manuscript. This study was partially supported by grants from the Ministry of Health and Welfare (Brain Science), the Science and Technology Agency (COE and Japan-Hungary), and the Human Health Science Foundation in Japan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chui, DH., Tanahashi, H., Ozawa, K. et al. Transgenic mice with Alzheimer presenilin 1 mutations show accelerated neurodegeneration without amyloid plaque formation. Nat Med 5, 560–564 (1999). https://doi.org/10.1038/8438
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/8438
This article is cited by
-
Senescence-related impairment of autophagy induces toxic intraneuronal amyloid-β accumulation in a mouse model of amyloid pathology
Acta Neuropathologica Communications (2023)
-
Fe3+ Facilitates Endocytic Internalization of Extracellular Aβ1–42 and Enhances Aβ1–42-Induced Caspase-3/Caspase-4 Activation and Neuronal Cell Death
Molecular Neurobiology (2019)
-
Evidence of intraneuronal Aβ accumulation preceding tau pathology in the entorhinal cortex
Acta Neuropathologica (2018)
-
Aβ42 oligomers modulate β-secretase through an XBP-1s-dependent pathway involving HRD1
Scientific Reports (2016)
-
Shengmai Formula Ameliorates Pathological Characteristics in AD C. elegans
Cellular and Molecular Neurobiology (2016)