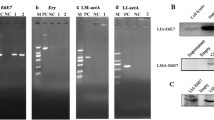
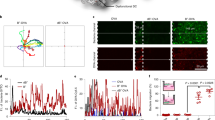
Abstract
Listeria monocytogenes is an intracellular organism that has the unusual ability to live in the cytoplasm of the cell. It is thus a good vector for targeting protein antigens to the cellular arm of the immune response. Here we use a model system, consisting of colon and renal carcinomas that express the influenza virus nucleoprotein and a recombinant L. monocytogenes that secretes this antigen, to test the potential of this organism as a cancer immunotherapeutic agent. We show that this recombinant organism can not only protect mice against lethal challenge with tumour cells that express the antigen, but can also cause regression of established macroscopic tumours in an antigen-specific T-cell-dependent manner.
Similar content being viewed by others
Article PDF
References
Kaufmann, S.H.E. Immunity to intracellular bacteria. Annu. Rev. Immun. 11, 129–163 (1993).
Cossart, P. et al. Listeriolysin O is essential for virulence of Listeria monocytogenes: Direct evidence obtained by gene complementation. Infect. Immun. 57, 3269–3636 (1989).
Tilney, L.G. & Portnoy, D.A. Actin filaments and the growth, movement, and spread of the intracellular bacterial parasite, Listeria monocytogenes . J. Cell Biol. 109, 1597–1608 (1989).
Berche, P., Gaillard, J. & Sansonetti, P.J. Intracellular growth of Listeria monocytogenes as a prerequisite for in vivo induction of T cell mediated immunity. J. Immun. 138, 2266–2271 (1987).
Hahn, H. & Kaufmann, S.H.E. The role of cell mediated immunity to bacterial infections. Rev. infect. Dis. 3, 1221–1250 (1991).
Kaufmann, S.H.E., Hug, E., Vath, U. & Muller, I. Effective protection against Listeria monocytogenes and delayed type hypersensitivity to Listeria antigens depend on cooperation between specific L3T4+ and Lyt2+ T Cells. Infect. Immun. 48, 263–266 (1985).
Schafer, R., Portnoy, D.A., Brassell, S.A. & Paterson, Y. Induction of a cellular immune response to a foreign antigen by a recombinant Listeria monocytogenes vaccine. J. Immun. 149, 53–59 (1992).
Ikonomidis, G., Paterson, Y., Kos, F.J. & Portnoy, D.A. Delivery of a viral antigen to the class I processing and presentation pathway of Listeria monocytogenes . J. exp. Med. 180, 2209–2218 (1994).
Ikonomidis, G., Frankel, F.R., Portnoy, D.A. & Paterson, Y. Listeria monocytogenes: A novel live vaccine vector. in Vaccines 95, (ed. Brown, F., Charnock, R., Ginsberg, H. & Norrby, E.) (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, New York) (in the press).
Boon, T., Cerottini, J.-C., Van den Eynde, B. & van der Bruggen, P. Tumour antigens recognized by T lymphocytes. Annu. Rev. Immun. 12, 337–365 (1994).
Coulie, P. et al. A new gene coding for a differentiation antigen recognized by autologous cytolytic T lymphocytes on HLA-A2 melanomas. J. exp. Med. 180, 35–42 (1994).
Cox, A.L. et al. Identification of a peptide recognized by five melanoma-specific human cytotoxic T cell lines. Science 264, 716–719 (1994).
Gaugler, B. et al. Human gene MAGE-3 codes for an antigen recognized on a melanoma by autologous cytolytic T lymphocytes. J. exp. Med. 179, 921–930 (1994).
Topalian, S.L. et al. Human CD4+ T cells specifically recognize a shared melanoma-associated antigen encoded by the tyrosinase gene. Proc. natn. Acad. Sci. U.S.A. 91, 9461–9465 (1994).
Kawakami, Y. et al. Cloning of the gene coding for a shared human melanoma antigen recognized by autologous T cells infiltrating into the tumor. Proc. natn. Acad. Sci. U.S.A. 91, 3515–3519 (1994).
Fearon, E.R., Itaya, T., Hunt, B., Vogelstein, B. & Frost, P. Induction in a murine tumor of immunogenic tumor variants by transfection with a foreign gene. Cancer Res. 38, 2975–2980 (1988).
Murphy, G.P. & Hushresky, W.J.J. A murine renal cell carcinoma. J. natn. Cancer Inst. 50, 1013–1025 (1973).
Schreiber, H. Tumor immunology. in Fundamental Immunology (ed. Paul, W.E.) chapt. 32 (Raven, New York, 1993).
Campbell, M.J., Esserman, L. & Levy, R. Immunotherapy of established murine B cell lymphoma: Combination of idiotype immunization and cyclophosphamide. J. Immun. 141, 3227–3233 (1988).
Pardoll, D.M. Cancer vaccines. Trends pharmacol. Sci. 14, 202–208 (1993).
Asher, A. et al. Murine tumor cells transduced with the gene for tumor necrosis factor-α. J. Immun. 146, 3227–3234 (1991).
Blankenstein, T. et al. Tumor suppression after tumor cell-targeted tumor necrosis factor gene transfer. J. exp. Med. 173, 1047–1052 (1991).
Porgador, A. et al. Interleukin 6 gene transfection into Lewis lung carcinoma tumor cells suppresses the malignant phenotype and confers immunotherapeutic competence against parental metastatic cells. Cancer Res. 52, 3679–3686 (1992).
Baskar, S. et al. Constitutive expression of B7 restores the immunogenicity of tumor cells expressing truncated MHC class II molecules. Proc. natn. Acad. Sci. U.S.A. 90, 5687–5690 (1993).
Guo, Y. et al. Effective tumor vaccine generated by fusion of hepatoma cells with activated B cells. Science 263, 518–520 (1994).
Hsieh, C.-S. et al. Development of TH1 CD4+ T cells through IL-12 produced by Listeria-induced macrophages. Science 260, 547–549 (1993).
Restifo, N.P. Identification of human cancers deficient in antigen processing. J. exp. Med. 177, 265–272 (1993).
Hui, K., Grosveld, F. & Festenstein, H. Rejection of transplantable AKR leukaemia cells following MHC DNA-mediated cell transformation. Nature 311, 750–752 (1984).
Wallich, R. et al. Abrogation of metastatic properties of tumour cells by de novo expression of H-2k antigens following H-2 gene transfection. Nature 315, 301–305 (1985).
Doyle, A. et al. Markedly decreased expression of class I histocompatibility antigens, protein and mRNA on human small-cell lung cancer. J. exp. Med. 161, 1135–1151 (1985).
Lassam, N. & Jay, G. Suppression of MHC class I RNA in highly oncogenic cells occurs at the level of transcription initiation. J. Immun. 143, 3792–3797 (1989).
Slingluff, C.L., Hunt, D.F. & Engelhard, V.H. Direct analysis of tumour-associated peptide antigens. Curr. Opin. Immun. 6, 733–740 (1994).
van der Bruggen, P.C. et al. A gene encoding an antigen recognized by cytolytic T lymphocytes on a human melanoma. Science 254, 1643–1650 (1991).
Cason, J., Khan, S.A. & Best, J.M. Towards vaccines against human papillomavirus type-16 genital infections. Vaccine 11, 603–611 (1993).
Wu, T.-C. Immunology of the human papilloma virus in relation to cancer. Curr. Opin. Immun. 6, 746–754 (1994).
Fetten, J.H., Roy, N. & Gilboa, E. A frameshift mutation at the NH2 terminus of the nucleoprotein gene does not affect generation of cytotoxic T lymphocyte epitopes. J. Immun. 147, 2697–2705 (1991).
Huang, A.T.C. et al. Role of bone marrow-derived cells in presenting MHC class I-restricted tumor antigens. Science 264, 961–965 (1994).
Geoffroy, C. et al. Purification and characterization of an extracellular 29-kilodalton phospholipase C from Listeria monocytogenes . Infect. Immun. 59, 2382–2388 (1991).
Kruisbeek, A. In vivo depletion of CD4 and CD8 specific T cells. in Current Protocols in Immunology (eds Coligan, J.E., Kruisbeek, A.M., Margulies, D.H., Shevach, E.M., Strober, W.) V. 1, 4.1.1–4.1.2 (Wiley, New York, 1994).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pan, ZK., Ikonomidis, G., Lazenby, A. et al. A recombinant Listeria monocytogenes vaccine expressing a model tumour antigen protects mice against lethal tumour cell challenge and causes regression of established tumours. Nat Med 1, 471–477 (1995). https://doi.org/10.1038/nm0595-471
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm0595-471
This article is cited by
-
Listeria monocytogenes-infected human monocytic derived dendritic cells activate Vγ9Vδ2 T cells independently of HMBPP production
Scientific Reports (2021)
-
Therapeutic options for treatment of human papillomavirus-associated cancers - novel immunologic vaccines: ADXS11–001
Gynecologic Oncology Research and Practice (2017)
-
Blockade of CTLA-4 promotes the development of effector CD8+ T lymphocytes and the therapeutic effect of vaccination with an attenuated protozoan expressing NY-ESO-1
Cancer Immunology, Immunotherapy (2015)
-
Whole tumor antigen vaccination using dendritic cells: Comparison of RNA electroporation and pulsing with UV-irradiated tumor cells
Journal of Translational Medicine (2008)
-
Cancer immunotherapy using Listeria monocytogenes and listerial virulence factors
Immunologic Research (2008)