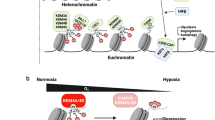
Abstract
Low oxygen tension influences tumor progression by enhancing angiogenesis; and histone deacetylases (HDAC) are implicated in alteration of chromatin assembly and tumorigenesis. Here we show induction of HDAC under hypoxia and elucidate a role for HDAC in the regulation of hypoxia-induced angiogenesis. Overexpressed wild-type HDAC1 downregulated expression of p53 and von Hippel–Lindau tumor suppressor genes and stimulated angiogenesis of human endothelial cells. A specific HDAC inhibitor, trichostatin A (TSA), upregulated p53 and von Hippel–Lindau expression and downregulated hypoxia-inducible factor-1α and vascular endothelial growth factor. TSA also blocked angiogenesis in vitro and in vivo. TSA specifically inhibited hypoxia-induced angiogenesis in the Lewis lung carcinoma model. These results indicate that hypoxia enhances HDAC function and that HDAC is closely involved in angiogenesis through suppression of hypoxia-responsive tumor suppressor genes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Grunstein, M. Histone acetylation in chromatin structure and transcription. Nature 389, 349–352 (1997).
Laherty, C.D. et al. Histone deacetylases associated with the mSin3 corepressor mediate Mad transcriptional repression. Cell 89, 349–356 (1997).
Nagy, L. et al. Nuclear receptor repression mediated by a complex containing SMRT, mSin3A and histone deacetylase. Cell 89, 373–380 (1997).
Kosugi, H. et al. Histone deacetylase inhibitors are the potent inducer/enhancer of differentiation in acute myeloid leukemia: a new approach to anti-leukemia therapy. Leukemia 13, 1316–1324 (1999).
Kouzarides, T. Histone acetylases and deacetylases in cell proliferation. Curr. Opin. Genet. Dev. 9, 40–48 (1999).
Guillemin, K. & Krasnow, M.A. The hypoxic response: huffing and HIFing. Cell 89, 9–12 (1997).
Ryan, H.E., Lo, J. & Johnson, R.S. HIF-1α is required for solid tumor formation and embryonic vascularization. EMBO J. 17, 3005–3015 (1998).
Kim, K.W. et al. Insulin-like growth factor II induced by hypoxia may contribute to angiogenesis of human hepatocellular carcinoma. Cancer Res. 58, 348–351 (1998).
Carmeliet, P. et al. Role of HIF-1α in hypoxia-mediated apoptosis, cell proliferation and tumour angiogenesis. Nature 394, 485–490 (1998).
Nor, J.E., Christensen, J., Mooney, D.J. & Polverini, P.J. Vascular endothelial growth factor (VEGF)-mediated angiogenesis is associated with enhanced endothelial cell survival and induction of Bcl-2 expression. Am. J. Pathol. 154, 375–384 (1999).
Gupta, K. et al. VEGF prevents apoptosis of human microvascular endothelial cells via opposing effects on MAPK/ERK and SAPK/JNK signaling. Exp. Cell Res. 247, 495–504 (1999).
Baek, J.H. et al. Hypoxia-induced VEGF enhances tumor survivability via suppression of serum deprivation-induced apoptosis. Oncogene 19, 4621–4631 (2000).
Gerber, H.P., Condorelli, F., Park, J. & Ferrara, N. Differential transcriptional regulation of the two vascular endothelial growth factor receptor genes. Flt-1, but not Flk-1/KDR, is upregulated by hypoxia. J. Biol. Chem. 272, 23659–23667 (1997).
Waltenberger, J., Mayr, U., Pentz, S. & Hombach, V. Functional upregulation of the vascular endothelial growth factor receptor KDR by hypoxia. Circulation 94, 1647–1654 (1996).
Kwon, H.J., Owa, T., Hassig, C.A., Shimada, J. & Schreiber, S.L. Depudecin induces morphological reversion of transformed fibroblasts via the inhibition of histone deacetylase. Proc. Natl. Acad. Sci. USA 95, 3356–3361 (1998).
Ravi, R. et al. Regulation of tumor angiogenesis by p53-induced degradation of hypoxia-inducible factor 1α. Genes Dev. 14, 34–44 (2000).
Stratmann, R., Krieg, M., Haas, R. & Plate, K.H. Putative control of angiogenesis in hemangioblastomas by the von Hippel-Lindau tumor suppressor gene. J. Neuropathol. Exp. Neurol. 56, 1242–1252 (1997).
Pal, S., Claffey, K.P., Dvorak, H.F. & Mukhopadhyay, D. The von Hippel-Lindau gene product inhibits vascular permeability factor/vascular endothelial growth factor expression in renal cell carcinoma by blocking protein kinase C pathways. J. Biol. Chem. 272, 27509–27512 (1997).
Royds, J.A., Dower, S.K., Qwarnstrom, E.E. & Lewis, C.E. Response of tumour cells to hypoxia: role of p53 and NF-κB. Mol. Pathol. 51, 55–61 (1998).
Gnarra, J.R. et al. Post-transcriptional regulation of vascular endothelial growth factor mRNA by the product of the VHL tumor suppressor gene. Proc. Natl. Acad. Sci. USA 93, 10589–10594 (1996).
Schmaltz, C., Hardenbergh, P.H., Wells, A. & Fisher, D.E. Regulation of proliferation-survival decisions during tumor cell hypoxia. Mol. Cell. Biol. 18, 2845–2854 (1998).
Long, X. et al. p53 and the hypoxia-induced apoptosis of cultured neonatal rat cardiac myocytes. J. Clin. Invest. 99, 2635–2643 (1997).
Blagosklonny, M.V. et al. p53 inhibits hypoxia-inducible factor-stimulated transcription. J. Biol. Chem. 273, 11995–11998 (1998).
Krieg, M. et al. Up-regulation of hypoxia-inducible factors HIF-1α and HIF-2α under normoxic conditions in renal carcinoma cells by von Hoppel-Lindau tumor suppressor gene loss of function. Oncogene 19, 5435–5443 (2000).
Mukhopadhyay, D., Tsiokas, L. & Sukhatme, V.P. Wild-type p53 and v-Src exert opposing influences on human vascular endothelial growth factor gene expression. Cancer Res. 55, 6161–6165 (1995).
Mukhopadhyay, D., Knebelmann, B., Cohen, H.T., Ananth, S. & Sukhatme, V.P. The von Hippel-Lindau tumor suppressor gene product interacts with Sp1 to repress vascular endothelial growth factor promoter activity. Mol. Cell. Biol. 17, 5629–5639 (1997).
Krieg, M., Marti, H.H. & Plate, K.H. Coexpression of erythropoietin and vascular endothelial growth factor in nervous system tumors associated with von Hippel-Lindau tumor suppressor gene loss of function. Blood 92, 3388–3393 (1998).
Zhong, H. et al. Overexpression of hypoxia-inducible factor 1α in common human cancers and their metastases. Cancer Res. 59, 5830–5835 (1999).
Varia, M.A. et al. Pimonidazole: A novel hypoxia marker for complementary study of tumor hypoxia and cell proliferation in cervical carcinoma. Gynecol. Oncol. 71, 270–277 (1998).
Meininger, C.J., Schelling, M.E. & Granger, H.J. Adenosine and hypoxia stimulate proliferation and migration of endothelial cells. Am. J. Physiol. 255, H554–H562 (1988).
Conrad, K.P. & Benyo, D.F. Placental cytokines and the pathogenesis of preeclampsia. Am. J. Reprod. Immunol. 37, 240–249 (1997).
Hassig, C.A. et al. A role for histone deacetylase activity in HDAC1-mediated transcriptional repression. Proc. Natl. Acad. Sci. USA 95, 3519–3524 (1998).
Dangond, F. & Gullans, S.R. Differential expression of human histone deacetylase mRNAs in response to immune cell apoptosis induction by trichostatin A and butyrate. Biochem. Biophys. Res. Commun. 247, 833–837 (1998).
Bartl, S. et al. Identification of mouse histone deacetylase 1 as a growth factor-inducible gene. Mol. Cell. Biol. 17, 5033–5043 (1997).
Nan, X. et al. Transcriptional repression by the methyl-CpG-binding protein MeCP2 involves a histone deacetylase complex. Nature 393, 386–389 (1998).
Schroeder, M. & Mass, M.J. CpG methylation inactivates the transcriptional activity of the promoter of the human p53 tumor suppressor gene. Biochem. Biophys. Res. Commun. 235, 403–406 (1997).
Prowse, A.H. et al. Somatic inactivation of the VHL gene in Von Hippel-Lindau disease tumors. Am. J. Hum. Genet. 60, 765–771 (1997).
Herman, J.G. et al. Silencing of the VHL tumor-suppressor gene by DNA methylation in renal carcinoma. Proc. Natl. Acad. Sci. USA 91, 9700–9704 (1994).
Cameron, E.E., Bachman, K.E., Myohanen, S., Herman, J.G. & Baylin, S.B. Synergy of demethylation and histone deacetylase inhibition in the re-expression of genes silenced in cancer. Nature Genet. 21,103–107 (1999).
Selker, E.U. Trichostatin A causes selective loss of DNA methylation in Neurospora. Proc. Natl. Acad. Sci. USA 95, 9430–9435 (1998).
Morwenna, S. & Ratcliffe, W.P. Mammalian oxygen sensing and hypoxia inducible factor-1. Int. J. Biochem. Cell Biol. 29, 1419–1432 (1997).
Kim, M.S. et al. Anti-angiogenic and Anti-invasive Activity of Torilin, a Sesquiterpene isolated from Torilis japonica. Int. J. Cancer 87, 269–275 (2000).
Yamamoto, H., Itoh, F., Hinoda, Y. & Imai, K. Inverse association of cell adhesion regulator messenger RNA expression with metastasis in human colorectal cancer. Cancer Res. 56, 3605–3609 (1996).
Passaniti, A. et al. A simple, quantitative method for assessing angiogenesis and antiangiogenic agents using reconstituted basement membrane, heparin, and fibroblast growth factor. Lab. Invest. 67, 519–528 (1992).
Lee, Y.M. et al. Determination of hypoxic region by hypoxia marker in developing mouse embryos in vivo: A possible signal for vessel development. Dev. Dyn. 220, 175–186 (2001).
Acknowledgements
We thank S.L. Schreiber for the gift of pBJ5-wt-HDAC1–expressing vector; J.A. Raleigh for hypoxia marker, pimonidazole and its associated antibody; and B.P. Yu for critical reading of the manuscript. Financial support was from the National Research Laboratory Fund (2000-N-NL-01-C-015), the Ministry of Science and Technology, Korea (to K-W.K.) and the Korea Science and Engineering Foundation (to H.J.K.).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kim, M., Kwon, H., Lee, Y. et al. Histone deacetylases induce angiogenesis by negative regulation of tumor suppressor genes. Nat Med 7, 437–443 (2001). https://doi.org/10.1038/86507
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/86507
This article is cited by
-
Gene regulation by histone-modifying enzymes under hypoxic conditions: a focus on histone methylation and acetylation
Experimental & Molecular Medicine (2022)
-
Epigenetics: key to improve delayed wound healing in type 2 diabetes
Molecular and Cellular Biochemistry (2022)
-
Differential molecular mechanistic behavior of HDACs in cancer progression
Medical Oncology (2022)
-
Targeting hypoxia-inducible factor 1 (HIF-1) signaling with natural products toward cancer chemotherapy
The Journal of Antibiotics (2021)
-
Anti-angiogenic agents — overcoming tumour endothelial cell anergy and improving immunotherapy outcomes
Nature Reviews Clinical Oncology (2021)