Abstract
Infection with group A streptococci can result in acute and post-infectious pathology, including rheumatic fever and rheumatic heart disease. These diseases are associated with poverty and are increasing in incidence, particularly in developing countries and amongst indigenous populations, such as Australia's Aboriginal population, who suffer the highest incidence worldwide1. Immunity to group A streptococci is mediated by antibodies against the M protein, a coiled-coil alpha helical surface protein of the bacterium2. Vaccine development3,4,5 faces two substantial obstacles. Although opsonic antibodies directed against the N terminus of the protein are mostly responsible for serotypic immunity, more than 100 serotypes exist. Furthermore, whereas the pathogenesis of rheumatic fever is not well understood, increasing evidence indicates an autoimmune process6,7. To develop a suitable vaccine candidate, we first identified a minimum, helical, non-host-cross-reactive peptide from the conserved C-terminal half of the protein and displayed this within a non-M-protein peptide sequence designed to maintain helical folding and antigenicity, J14 (refs. 8,9). As this region of the M protein is identical in only 70% of group A streptococci isolates10, the optimal candidate might consist of the conserved determinant with common N-terminal sequences found in communities with endemic group A streptococci. We linked seven serotypic peptides with J14 using a new chemistry technique that enables the immunogen to display all the individual peptides pendant from an alkane backbone. This construct demonstrated excellent immunogenicity and protection in mice.
Similar content being viewed by others
Main
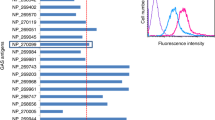
We defined common M proteins from group A streptococci (GAS) isolates from patients in Aboriginal communities and at the Royal Darwin Hospital (Northern Territory of Australia) by sequence typing of the N terminus11 and by Vir typing12. Seven sequence types made up 13% of the isolates in hospital and community GAS infections (Table 1). Given these epidemiological data, we synthesized seven peptides corresponding to the N-terminal M protein sequences from these isolates (Table 1). We chose N-terminal sequences because epitopes from the N terminus, rather than epitopes from the A and B repeat regions of the M proteins of these strains, were more effective in inducing protective responses (E.R.B. et al., unpublished data). Comparison of sera stratified by patient age (1–4 years old, 5–10 years old, 11–19 years old and 20 or more years old) showed that the antibody responses to most of these peptides increased with age (Fig. 1), in parallel with the acquisition of immunity to GAS (Fig. 1), which normally occurs by the third decade of life13. There were no age-related differences in antibodies to tetanus toxoid or diphtheria toxoid. This indicates that vaccine-induced antibody responses to these N-terminal peptides may contribute to immunity to GAS.
Short horizontal bars, mean anti-peptide IgG concentration (μg/ml sera). Each data point may represent more than one set of measurements. ↔, P<0.05 (comparison of means). The prevalence of NS51–19-specific antibodies in the 20+ age group shows one outlier. If this outlier is removed, there is no statistical difference between the age groups 1-4 years and 20+ years.
We then individually conjugated the N-terminal peptides 88/301–20, Y504S1–20, BSA101–28, NS271–19, NS11–19, NS51–19 and PL11–19 to tetanus toxoid and used these to immunize B10.BR mice to determine whether these antibodies could opsonize GAS. After one immunization of peptide in complete Freund's adjuvant and two subsequent boosts of peptide in phosphate-buffered saline (PBS), all mice produced high titers of antibodies against the immunizing peptide (titers greater than 12,800) (data not shown). None of the N-terminal peptide antisera cross-reacted with antigens or homologues involved in the pathogenesis of rheumatic fever (pig cardiac myosin, pig muscle tropomyosin, human epidermis keratin or mouse whole-heart extract), by a published western blot assay protocol14 (data not shown). We next used these sera in an indirect bactericidal assay to determine if they could opsonize the corresponding GAS isolate. Antisera against peptides 88/301–20, Y504S1–20, BSA101–28, NS271–19, NS11–19 and NS51–19 had a very high opsonic activity, with a reduction of more than 80% in colony forming units (CFU), and antibodies against PL11–19 resulted in a reduction in CFU of 55% of the PL1 GAS strain, further indicating that these epitopes may be useful vaccine candidates specific to these endemic isolates. We tested antisera for cross-reactivity to the other peptides by enzyme-linked immunosorbent assay (ELISA) and opsonization assay. Antisera against Y504S1–20 were opsonic against strain BSA10, even though antibodies specific for Y504S1–20 did not recognize the peptide BSA101–28 by ELISA, indicating that determinants that induce cross-opsonizing antibodies may cross-react with epitopes not located at the N terminus of the M protein. Several studies have reported that antibodies against peptide from the M protein can cross-react with several alpha-helical proteins including laminin, myosin and tropomyosin15. Thus, the cross-reactive antibodies against peptide (specific for Y504S1–20) might recognize and cross-opsonize other non-homologous α-helical M or M-like proteins (found in strain BSA10). However, antibodies specifically against BSA101–28 recognized Y504S1–20 (as well as NS141–19 and 88/301–20) by ELISA but did not opsonize these strains. This was unexpected; perhaps the structure of the opsonic determinant (rather than the ELISA determinant) is maintained on the Y504S bacterium, but not on the synthetic peptide. Factors other than sequence must be involved, and may include the presence of other proteins and the M protein itself. Other studies have also identified anti-peptide sera that cross-react with heterologous serotypes of M protein by ELISA, but none of these sera cross-opsonized the respective heterologous GAS (ref. 16).
In further experiments, we vaccinated B10.BR mice with J14. J14 was very immunogenic in inducing antibodies detectable by ELISA, but the amount of opsonic activity was low (average, 29.7% reduction in CFU (Table 2). However, in three separate experiments in which mice were primed with 30 μg J14 in complete Freund's adjuvant and boosted three times with 3 μg J14 in PBS, 16 of 19 mice were completely protected after challenge intraperitoneally with 3.6×105 organisms of a reference strain M6, known to express the J14 epitope9. In contrast, only 3 of 15 mice vaccinated with PBS survived. In other challenge experiments, we vaccinated mice with a very closely related conserved peptide, J8 (refs 8,9), linked to diptheria toxoid, and administered it with alum. After challenge, 8 of 10 mice survived.
Given these data, we chose seven N-terminal peptides (88/301–20, Y504S1–20, BSA101–28, NS271–19, NS11–19, NS51–19 and PL11–19) for further study. We designed a single immunogen containing these serotypic determinants and J14. To combine these epitopes into an immunogen highly accessible to the immune system, we used chemistry whereby purified peptides are assembled into polymers pendant from an alkane backbone17,18. We immunized outbred (Quackenbush) mice and boosted them twice with the construct, called a ‘heteropolymer’. Antibodies against each of the individual side chain peptides were induced at titers up to 1,638,400 by day 61 after vaccination (data not shown). We also determined the isotype response (Table 2). We next vaccinated BALB/c mice and athymic BALB/c nude (nu/nu) mice (which lack T cells). The heteropolymer construct was poorly immunogenic in nude mice (average titers in four nude mice, 125, compared to titers of 10,400 in normal BALB/c mice). These data, with the isotype data demonstrating an IgG antibody response to the heteropolymer in normal mice, strongly indicate that the vaccine is stimulating T cell-dependent response.
We determined the in vitro opsonic (bactericidal) activities of the mouse antisera against two different GAS strains, 88/30 and 2040 (serotype, M12), by the percentage reduction in CFU. These strains were chosen for this study because the N terminus (88/30) GAS M protein is present on the heteropolymer, whereas the N terminus of the 2040 GAS strain is not present on the heteropolymer. Both have C-terminal epitopes that are present on the heteropolymer (J14) (E.R.B. et al., unpublished data). The data showed ELISA titers and opsonic activity of up to 100% (Table 2). We also determined ELISA titers and opsonic activities of positive control antisera against diphtheria toxoid conjugates (88/301–20-DT and 20401–12-DT) (Table 2). Opsonic activity ranged from 62.7% to 100% and from 77.0% to 98.4%, respectively, for these conjugates. Negative control groups were mice immunized with PBS and mice immunized with the alkane backbone alone. Although the degree of opsonization shown by these studies is very encouraging, high titers of antibodies may not necessarily correlate with protection19.
We next challenged immunized mice and control mice with either the 88/30 or 2040 strain at a dose previously estimated to be lethal to 90% of mice. After challenge with 88/30, in two separate experiments, 19 of 20 heteropolymer-immunized mice were protected (10 of 10 in the first experiment and 9 of 10 in the second), and 9 of 9 heteropolymer-immunized mice were protected against challenge with 2040 (Table 2). Ten of twelve and seven of eight mice immunized with 88/301–20-DT and 20401–12-DT, respectively, were also protected whereas seven of forty-five negative control mice did not succumb to infection. Protection was evident very early after challenge, with most control mice succumbing after 2–3 days (Fig. 2) Protected mice showed no adverse responses to the vaccine and did not show obvious pathology or symptoms of infection after challenge.
To determine whether heteropolymer-immunized mice challenged with the GAS strain 88/30 were protected by antibodies against the conserved region (J14 peptide) or to the N-terminal 88/30 peptide on the polymer, we did a peptide inhibition bactericidal assay20. The addition of peptides 88/301–20, J14, or 88/301–20 plus J14 to heteropolymer antisera in repeat experiments led to inhibition of opsonization of 88/30 GAS, with ranges of 75–76%, 31–66% and 76–81%, respectively. The addition of a nonspecific peptide from schistosoma19 to the heteropolymer sera did not inhibit opsonization of 88/30 GAS. The addition of the 88/30 N-terminal peptide to 88/30-DT sera inhibited opsonization by up to 85%, whereas addition of J14 or the schistosoma peptide had no effect. In contrast, the N-terminal peptide from the 2040 isolate had no inhibitory effect on opsonization of 2040 GAS mediated by heteropolymer antisera, whereas J14 peptide completely inhibited opsonization, consistent with the fact that the heteropolymer does not contain the 2040 N-terminal peptide but does contain J14. In a further control experiment, 2040 N-terminal peptide inhibited opsonization mediated by 2040-DT antisera by 67%. These data strongly indicate that the heteropolymer can induce protection against challenge from GAS through antibodies specific for both the N terminus of the M protein (if that serotypic determinant is presented on the immunogen) and the C-terminal epitope, J14.
We also tested the heteropolymer construct for its ability to induce antibodies in rabbits. After a primary injection and two boosts, the antibody titers in two rabbits to the heteropolymer were 12,800 and 3,200. We also tested these antibodies for their ability to recognize myosin, tropomyosin and keratin (antigens that may be target antigens in rheumatic heart disease15). There was only slight recognition of one antigen, tropomyosin, with a titer of 200 in one rabbit.
We used new polymer technology here to deliver defined epitopes from conserved and serotypic regions of the M protein of GAS isolates from an endemic area. The advantages of a heteropolymer construct are that multiple epitopes, shown to be opsonic for GAS, without B- or T-cell cross-reactivity to host-tissue proteins such as myosin, tropomyosin or keratin, are combined into a single immunogen. We targeted very specific, highly effective N-terminal epitopes derived from GAS isolates common to a highly endemic region. Because a vaccine with only N-terminal epitopes would still be unable to target all GAS endemic to this region, we included a conserved region epitope, J14, to form the basis of a broad-spectrum vaccine. Such a vaccine might be widely effective, but within a high endemic area would be designed to deliver increased protection by targeting both serotypic and conserved determinants on the M protein. Not only did the heteropolymer induce strong antibody responses to all peptides in outbred mice, but also the induced antibodies substantially opsonized 88/30 and 2040 GAS in vitro. Moreover, a streptococcal multi-epitope vaccine protected mice against GAS challenge in vivo. Both conserved and variant epitopes were the targets of the protective immune responses. Given that the conserved peptide sequence is completely conserved in approximately 70% of GAS isolates10, it would seem wise to not rely solely on the J14 epitope alone in a vaccine. It is not known what effect the slight degree of sequence diversity within the J14 region would have on vaccine efficacy. BSA10 and PL1 strains, for example, have changes in three amino acids within the J14 region (E.R.B. et al., unpublished data). The approach described here could be broadened to include peptides (when defined) from other non-M-protein-derived vaccine candidates such as C5a peptidase21 and cysteine proteases22, as studies indicate they may induce non-serotypic immunity. New-generation multi-epitope vaccines may provide a safe and effective way to prevent endemic streptococcal infection and subsequent morbidity.
Methods
Patients and detection of human antibodies.
Sera were obtained from Australian Aboriginals from highly endemic areas of the Northern Territory and included 14 children 1–4 years old, 9 children 5–10 years old, 30 teenagers 11–19 years old and 76 adults (20 or more years old). ELISA was used to measure human serum antibodies to the peptides as described20. Standard curves of optical density versus known concentrations of human IgG were used to calculate antibody concentration, expressed as μg/ml serum20.
Syntheses of peptides and conjugation to a peptide carrier.
Synthetic peptides were produced as described23 and were purified by high-performance liquid chromatography. Peptides were conjugated through a C-terminal cysteine to either tetanus toxoid or diphtheria toxoid according to Coligan et al24. N-terminal peptide sequences are given in Table 1 (ref. 11). The sequence for J14 is KQAEDKVKASREAKKQVEKALEQLEDKVK (ref. 9). The sequence of the 2040 (M12) GAS N terminal peptide is DHSDLVAEKQRLC (ref. 11). The sequence of the non-specific schistosoma peptide is EGKVSTLPLDIQIIAATMSK (ref. 20).
Assembly of peptide-based polymers.
Peptide synthesis and polymerization were done as described17,18. Individual peptides representing portions of the M protein were assembled using fluorenylmethoxycarbonyl chemistry, and a residue of Lys (4-methyltrityl) was inserted at the C-terminus. All peptides were assembled as the carboxamide. After removal of the N-terminal fluorenylmethoxycarbonyl group, the exposed N-terminal amino group was acetylated and the 4-methyltrityl group of the C-terminal lysine was then removed in 1% trifluoroacetic acid in dichloromethane. The amino group exposed at the C-terminal lysine in this way was then derivitized with acryloyl chloride and polymerization of individual peptides was carried out. Polymers were dialyzed exhaustively against water; non-diffusable material eluted in the void volume of a Protein Pak 300 column (0.78×30 cm) (Waters, Australia, Sydney, Australia), indicating a molecular weight in excess of 400 kDa.
Immunization of mice.
Peptides (peptide alone, peptide conjugated to diphtheria toxoid or the heteropolymer construct) were administered subcutaneously at the tail base to B10.BR or Quackenbush (outbred) mice (Animal Resources Centre, Perth, Western Australia). Each mouse received a total of 30 μg immunogen emulsified in complete Freund's adjuvant (Difco Laboratories, Detroit, Michigan). Mice were boosted after 21 d with 30 μg immunogen in PBS and received two subsequent booster injections at 10-day intervals (except for J14-immunized mice, which were boosted with 3 μg peptide in PBS). Blood was obtained from mice through the tail artery at two weekly intervals after primary immunization.
Detection of murine antibodies.
ELISA was used to measure mouse serum antibodies and the antibody isotype to the peptides as described9,14. ‘Titer’ is defined as the highest dilution that produced absorbance more than three standard deviations (s.d.) above the mean absorbance measurement of control wells containing normal mouse sera.
GAS strains.
GAS isolates were obtained from the Menzies School of Health Research (Darwin, Australia)(Table 1). Isolates were collected in the Northern Territory of Australia. 2040 [M12] is a reference strain.
Bactericidal and peptide inhibition bactericidal assay.
Mouse sera against peptide were assayed for their ability to opsonize GAS in an indirect bactericidal assay essentially as described25. Bacteria were grown overnight at 37 °C in 5 ml Todd-Hewitt broth (THB), then 200 μl of overnight culture was subinoculated into 5 ml warm THB and grown for 2 hours at 37 °C. GAS was then serially diluted to 10−4 in saline. The bacteria dilution (50 μl) was mixed with fresh heat-inactivated sera (50 μl) and non-opsonic, heparinized human donor blood (400 μl). All donor blood was prescreened before being assayed to ensure that it could support the growth of the GAS strain by at least 32 times the inoculum in a 3-hour incubation at 37 °C (ref. 20). The mixture was incubated at 37 °C for 3 h with ‘end-to-end’ rotation, and 50 μl from each tube was plated out in duplicate on blood agar ‘pour plates’. After overnight incubation, the colonies on each plate were counted. Opsonic activity of the anti-peptide sera (% reduction in mean CFU) was calculated as [1–(CFU in the presence of anti-peptide sera)/(mean CFU in the presence of normal mouse sera)] ×100. Donor blood and bacteria controls were included with each experiment.
The peptide inhibition bactericidal assay was done as described above except that 100 μg peptide was added to 50 μl fresh heat-inactivated sera and incubated at room temperature for 30 min before the addition of bacteria and non-opsonic heparinized donor blood20.
GAS challenge procedure.
GAS strains were passaged through mice by the intraperitoneal route to enhance their virulence. Each strain was cultured overnight in THB with 1% neopeptone, washed twice in THB with 1% neopeptone and resuspended in 25% of the original volume. The inoculum dose was determined by absorbance at 600 nm and by mean colony counts of ‘pour plates’ prepared with 50-μl samples of 10-fold dilutions of the bacterial suspensions added to 20 ml melted THB agar with 2.5% horse blood. After overnight incubation at 37 °C, colonies were counted. Immunized and control mice were challenged intraperitoneally with a 90% lethal dose of GAS, 10 d after the last immunization. According to Institutional Ethics Committee requirements, mice that were found to be moribund were killed.
Statistical analysis.
Geometric means were calculated and comparisons between groups of patients were made by Student's t-test. The χ2 test was used to calculate significance in GAS challenge experiments. A P value of <0.05 was considered statistically significant for all analysis.
References
Carapetis, J.R., Wolff, D.R. & Currie, B.J. Acute rheumatic fever and rheumatic heart disease in the top end of Australia's Northern Territory. Med. J. Aust. 164, 146–149 (1996).
Beachey, E.H., Seyer, J.M., Dale, J.B., Simpson, W.A. & Kang, A.H. Type-specific protective immunity evoked by synthetic peptide of Streptococcus pyogenes M protein. Nature 292, 457–459 (1981).
Bessen, D. & Fischetti, V.A. Influence of intra nasal immunisation with synthetic peptides corresponding to conserved epitopes of M protein on mucosal colonisation by group A streptococci. Infect. Immun. 56, 2666–2672 (1988).
Pruksakorn, S. et al. Towards a vaccine for rheumatic fever: identification of a conserved target epitope on M protein of group A streptococci. Lancet 344, 639–642 (1994).
Dale, J.B., Simmons, M., Xhiang, E.C. & Chiang, E.Y. Recombinant, octavalent group A streptococcal M protein vaccine. Vaccine 14, 944–948 (1996).
Kotb, M., Courtney, H.S., Dale, J.B. & Beachey, E.H. Cellular and biochemical responses of human T lymphocytes stimulated with streptococcal M proteins. J. Immunol. 142, 966–979 (1989).
Pruksakorn, S. et al. Identification of T-cell autoepitopes that cross-react with the carboxyterminal segment of the M protein of Group A streptococci. Int. Immunol.. 6, 1235–1244 (1994).
Relf, W.A. et al. Mapping a conserved conformational epitope from the M protein of group A streptococci. Peptide Res. 9, 12–20 (1996).
Hayman, W.A. et al. Mapping the minimal T and B cell epitope within a peptide vaccine candidate from the conserved region of the M protein of group A streptococci. Int. Immunol. 9, 1723–1733 (1997).
Brandt, E.R., Hayman, W.A., Currie, B., Pruksakorn, S. & Good, M.F. Human antibodies to the conserved region of the M protein: opsonisation of heterologous strains of group A streptococci. Vaccine 16, 1805–1812 (1997).
Relf, W.A., Martin, D.R. & Sriprakash, K.S Identification of sequence types among the m-nontypeable group A streptococci. J. Clin. Microbiol. 30, 3190–3194 (1992).
Gardiner, D. et al. Vir typing: A long-PCR typing method for group A streptococci. PCR Meth. Applic. 4, 288–293 (1995).
Quinn, R.W., Zwaag, R.W. & Lowry, P.N. Aquisition of group A streptococcal M protein antibodies. Ped. Infect. Dis. J. 4, 374–378 (1985).
Pruksakorn, S., Galbraith, A., Houghten, R.A. & Good, M.F. Conserved T and B cell epitopes on the M protein of group A streptococci: Induction of bactericidal antibodies. J. Immunol. 149, 2729–2735 (1992).
Robinson, J.H. & Kehoe, M.A. Group A streptococcal M proteins: virulence factors and protective antigens. Immunol. Today 13, 362–367 (1992).
Beachey, E.H. & Seyer, J.M. Protective and nonprotective epitopes of chemically synthesized peptides of the NH2-terminal region of type 6 streptococcal M protein. J. Immunol. 136, 2287–2292 (1986).
O'Brien-Simpson, N.M., Ede, N.J., Brown, L.E., Swan, J. & Jackson, D.C. Polymerization of unprotected synthetic peptides: a view towards synthetic peptide vaccines. J. Am. Chem. Soc. 119, 1183–1188 (1997).
Jackson, D.C., O'Brien-Simpson, N., Ede, N.J. & Brown, L.E. Free radical induced polymerization of synthetic peptides into polymeric immunogens. Vaccine 15, 1697–1705 (1997).
Salvadori, L.G., Blake, M.S., McCarty, M., Tai, J.Y. & Zabriskie, J.B. Group A streptococcus-liposome ELISA antibody titers to group A polysaccharide and opsonophagocytic capabilities of the antibodies. J. Infect. Dis. 171, 593–600 (1995).
Brandt, E.R. et al. Opsonic human antibodies from an endemic population specific for a conserved epitope on the M protein of group A streptococci. Immunology 89, 331–337 (1986).
Ji, Y., Carlson. B., Kondagunta, A. & Cleary, P.P. Intranasal immunisation with C5a peptidase prevents nasopharyngeal colonisation of mice by group A streptococcus. Infect. Immun. 65, 2080–2087 (1997).
Kapur, V. et al. Vaccination with streptococcal cysteine protease protects mice against challenge with heterologous group A streptococci. Microb. Pathog. 16, 443–450 (1994).
Houghten, R.A. General method for the rapid solid phase synthesis of large numbers of peptides: specificity of antigen- antibody interaction at the level of individual amino acids. Proc. Natl. Acad. Sci. USA 82, 5231–5235 (1985).
Coligan, J.E., Kruisbeek, A.M., Margulie, D.H., Shevach, E.M. & Strober, W. in Current Protocols in Immunology Vol. 2 (John Wiley, New York, 1992).
De Malmanche, S.A. & Martin, D.R. Protective immunity to the group A streptococcus may only be strain specific. Med. Microbiol. Immunol. 183, 299–306 (1996).
Acknowledgements
We thank A. Kelso, C. Olive, D. Pombo and L. Powell for reviewing the manuscript. This work was supported by the National Health and Medical Research Council (Australia), the National Heart Foundation of Australia, The Prince Charles Hospital Foundation and the Cooperative Research Centre for Vaccine Technology.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Brandt, E., Sriprakash, K., Hobb, R. et al. New multi-determinant strategy for a group A streptococcal vaccine designed for the Australian Aboriginal population. Nat Med 6, 455–459 (2000). https://doi.org/10.1038/74719
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/74719
This article is cited by
-
Atlas of group A streptococcal vaccine candidates compiled using large-scale comparative genomics
Nature Genetics (2019)
-
Physicochemical characterisation, immunogenicity and protective efficacy of a lead streptococcal vaccine: progress towards Phase I trial
Scientific Reports (2017)
-
Cutting Edge Issues in Rheumatic Fever
Clinical Reviews in Allergy & Immunology (2012)
-
Current status of multiple antigen-presenting peptide vaccine systems: Application of organic and inorganic nanoparticles
Chemistry Central Journal (2011)
-
Novel strategies for controlling Streptococcus pyogenes infection and associated diseases: from potential peptide vaccines to antibody immunotherapy
Immunology & Cell Biology (2009)