Abstract
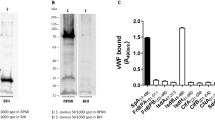
Fever, hypotension and bleeding disorders are common symptoms of sepsis and septic shock. The activation of the contact-phase system is thought to contribute to the development of these severe disease states by triggering proinflammatory and procoagulatory cascades; however, the underlying molecular mechanisms are obscure. Here we report that the components of the contact-phase system are assembled on the surface of Escherichia coli and Salmonella through their specific interactions with fibrous bacterial surface proteins, curli and fimbriae. As a consequence, the proinflammatory pathway is activated through the release of bradykinin, a potent inducer of fever, pain and hypotension. Absorption of contact-phase proteins and fibrinogen by bacterial surface proteins depletes relevant coagulation factors and causes a hypocoagulatory state. Thus, the complex interplay of microbe surface proteins and host contact-phase factors may contribute to the symptoms of sepsis and septic shock.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Risberg, B., Andreasson, S. & Eriksson, E. Disseminated intravascular coagulation. Acta Anaesthesiol. Scand. Suppl. 95, 60–71 (1991).
Müller-Esterl, W.,Kinnogens, kinins and kinships. Thromb. Haemost. 61, 2–6 (1989).
Colman, R.W. & Schmaier, A.H. The contact activation system: Biochemistry and interactions of these surface-mediated defence reactions. Crit. Rev. Oncol. Hematol. 5, 57–85 (1986).
Kaplan, A.P. & Silverberg, M. The coagulation-kinin pathway of human plasma. Blood 70, 1–15 (1987).
Hall, J.M. Bradykinin receptors: Pharmacological prpperties biological roles. Pharmacol. Ther. 56, 131–190 (1992).
Olsén, A., Jonsson, A. & Normark, S. Fibronectin binding mediated by a novel class of surface organelles on Escherichia coli. Nature 338, 652–655 (1989).
Collinson, S.K., Emody, L., Müller, K.H., Trust, T.J. Kay, W.W. Purification and characterization of thin, aggregative fimbriae from Salmonella enteritidis. J. Bacteriol. 173, 4773–4781 (1991).
Olsén, A., Arnqvist, A., Hammar, M., Sukupolvi, S. & Normark, S., The RpoS sigma factor relieves H-NS-mediated transcriptional repression of csgA, the subunit gene of fibronectin-binding curli in Escherichia coli. Mol. Microbiol. 7, 523–536 (1993).
Collinson, S.K. et al. Thin, aggregative fimbriae mediate binding of Salmonella enteritidis to fibronectin. J. Bacteriol. 175, 12–18 (1993).
Sjöbring, U., Pohl, C. & Olsén, A., Plasminogen, absorbed by Escherichia coli expressing curli or by Salmonella enteritidis expressing thin aggregative fimbriae, can be activated by simultaneously captured tissue-type plasminogen activator (t-PA). Mol. Microbiol. 14, 443–452 (1994).
Ben Nasr, A.B., Olsén, A., Sjöbring, U., Muller-Esterl, W. & Björck, L. Assembly of human contact phase factors and release of bradykinin at the surface of curli-ex-pressing Escherichia coli. Mol. Microbiol. 20, 927–935 (1996).
Sukupolvi, S., Edelstein, A., Rhen, M., Normark, S.J. & Pfeifer, J.D. Development of a murine model of chronic Salmonella infection. Infect. Immun. 65, 838–842 (1997).
Furie, B. & Furie, B.C. The molecular basis of blood coagulation. Cell 53, 505–518 (1988).
Müller-Esterl, W., Johnson, D., Salvesen, G. & Barrett, A.A. Human kininogens. Methods Enzymol. 163, 240–256 (1988).
Ponjee, G.A., Vader, H.L. de Wild, P.J., Janssen, G.W. & van der Graaf, F. One-step chromogenic equivalent of activated partial thromboplastin time evaluated for clinical application. Clin. Chem. 37, 1235–1244 (1991).
Scott, C.F., Whitaker, E.J., Hammond, B.F. & Colman, R.W. Purification and characterization of a potent 70-kDa thiol lysyl-proteinase (Lys-gingivain) from Porphyromonas gingivalis that cleaves kininogens and fibrinogen. J. Biol. Chem. 268, 7935–7942 (1993).
Herwald, H., Collin, M., Müller-Esterl, W. & Bjorck, L. Streptococcal cysteine proteinase releases kinins: A novel virulence mechanism. J. Exp. Med. 184, 665–673 (1996).
Ubben, D. & Schmitt, R. Tn1721 derivatives for transposon mutagenesis, restriction mapping and nucleotide sequence analysis. Gene 41, 145–152 (1986).
Evans, D.G., Evans, D.J.J. & Tjoa, W. Hemagglutination of human group A erythro-cytes by enterotoxigenic Escherichia coli isolated from adults with diarrhea: Correlation with colonization factor. Infect. Immun. 18, 330–337 (1977).
Hasan, A.A., Cines, D.B., Zhang, J. & Schmaier, A.H. The carboxyl terminus of bradykinin and amino terminus of the light chain of kininogens comprise an endothelial cell binding domain. J. Biol. Chem. 269, 31822–31830 (1994).
Hock, J., Vogel, R., Linke, R.P. & Müller-Esterl, W. High molecular weight kininogen-binding site of prekallikrein probed by monoclonal antibodies. J. Biol. Chem. 265, 12005–12011 (1990).
Fraker, P.J. & Speck, S.C.J. Protein and cell membrane iodinations with a sparingly soluble chloroamide, 1,3,4,6-tetrachloro-3a,6a-diphrenylglycoluril. Biochem. Biophys. Res. Commun. 80, 849–857 (1978).
Laemmli, U.K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227, 680–685 (1970).
Egberg, N. & Heedman, P.A. Simplified performance of amidolytic factor X assay. Thromb. Res. 25, 437–440 (1982).
Becker, U., Bartl, K. & Wahlefeld, A.W. A functional photometric assay for plasma fibrinogen. Thromb. Res. 35, 475–484 (1984).
Odegard, O.R., Lie, M. & Abildgaard, U. Heparin cofactor activity measured with an amidolytic method. Thromb. Res. 6, 287–294 (1975).
Nilsson, I.M. Haemorrhagic and Thrombotic Diseases. 217–218 (Wiley and Sons, London, UK, 1974).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Herwald, H., Mörgelin, M., Olsén, A. et al. Activation of the contact-phase system on bacterial surfaces—a clue to serious complications in infectious diseases. Nat Med 4, 298–302 (1998). https://doi.org/10.1038/nm0398-298
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nm0398-298
This article is cited by
-
Adhesive behavior and detachment mechanisms of bacterial amyloid nanofibers
npj Computational Materials (2019)
-
NETosis, complement, and coagulation: a triangular relationship
Cellular & Molecular Immunology (2019)
-
The initiation and effects of plasma contact activation: an overview
International Journal of Hematology (2017)
-
Comprehensive study into the activation of the plasma enzyme systems during attacks of hereditary angioedema due to C1-inhibitor deficiency
Orphanet Journal of Rare Diseases (2015)
-
Structural and mechanistic insights into the bacterial amyloid secretion channel CsgG
Nature (2014)