Abstract
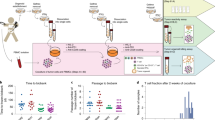
Human autologous tumour-specific cytotoxic T lymphocytes (CTL) were generated from peripheral blood on small formalin-fixed paraffin-embedded sections of a gastric cancer. The CTL killed live target cells at an effector/target ratio of 1 within 24 hours and showed the same target specificity as those induced on live cancer cells. The killing activity of the CTL lasted for more than four months in culture and was inhibited by antibodies against CD8 and MHC-class I. These results suggest that adoptive immunotherapy of tumours will be possible with CTL induced on a stable source of tumour antigen.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Kawakami, Y. et al. Identification of a human melanoma antigen recognized by tumor-infiltrating lymphocytes associated with in vivo tumor rejection. Proc. natn. Acad. Sci. U.S.A. 91, 6458–6462 (1994).
Slingluff, C.L. et al. Cytotoxic T-lymphocyte response to autologous human squamous cell cancer of the lung: Epitope reconstitution with peptides extracted from HLA-AW68. Cancer Res. 54, 2731–2737 (1994).
Tjoa, B.A. & Kranz, D.M. Generation of cytotoxic T-lymphocytes to a self-peptide/class I complex: A model for peptide-mediated tumor rejection. Cancer Res. 54, 204–208 (1994).
Ioannides, C.G. & Whiteside, T. T cell recognition of human tumors: Implications for molecular immunotherapy of cancer. Clin. Immunol. Immunopath. 66, 91–106 (1993).
Monaco, J.J. A molecular model of MHC class-I-restricted antigen processing. Immun. Today 13, 173–179 (1992).
Rosenberg, S.A. et al. A progress report on the treatment of 157 patients with advanced cancer using lymphokine-activated killer cells and interleukin-2 or high-dose interleukin-2 alone. New Engl. J. Med. 316, 889–897 (1987).
Fisher, R.I. et al. Metastatic renal cancer treated with interleukin-2 andlymphokine-activated killer cells-a phase II clinical trial. Annals intern Med. 108, 518–523 (1988).
Rosenberg, S.A. et al. Gene transfer into humans-immunotherapy of patients with advanced melanoma, using tumor-infiltrating lymphocytes modified by retroviral gene transduction. New Engl. J. Med. 323, 570–578 (1990).
Ikarashi, H. et al. Immunomodulation in patients with epithelial ovarian cancer after adoptive transfer of tumor-infiltrating lymphocytes. Cancer Res. 54, 190–196 (1994).
Wolfel, T. et al. Lysis of human pancreatic adenocarcinoma cells by autologous HLA-class I-restricted cytolytic T-lymphocyte (CTL) clones. Int. J. Cancer 54, 636–644 (1993).
Crowley, N.J., Darrow, T.L., Quinn-Allen, M.A. & Seigler, H.F. MHC-restricted recognition of autologous melanoma by tumor-specific cytotoxic T cells - evidence for restriction by a dominant HLA-A allele. J. Immun. 146, 1692–1699 (1991).
Hay, R.J., Park, J.-G., Gazar, A. (eds) Atlas of Human Tumor Cell Lines. (Academic, Los Angeles, California, 1994).
Holded, C.A. et al. The detection of endothelial cell antigens in cutaneous tissue using methacarn and periodate lysine paraformaldehyde fixation. J. immun. Methods 91, 45–52 (1986).
Pollard, K. & Holgate, C.S. Surface antigen preservation by PLP and formalin fixation and the effects of tissue processing. J. immun. Methods 96, 145 (1987).
Heo, D.S., Whiteside, T.L., Kanbour, A. & Herberman, R.B. Lymphocytes infiltrating human ovarian tumors: I. Role of Leu-19 (NKH1)-positive recombinant IL-2-activated cultures of lymphocytes infiltrating human ovarian tumors. J. Immun. 140, 4042–4049 (1988).
Liu, S.Q., Shiba, R., Kim, B.S., Saijo, K. & Ohno, T. Long-term serum/plasma-free culture of human cytotoxic T lymphocytes induced from peripheral blood. Cancer Immunol. Immunother. 39, 279–285 (1994).
Nakashima, M., Watanabe, T., Koprowski, H., Schuchter, L. & Steplewski, Z. In vitro expansion of tumor-specific, HLA-restricted human CD8+ cytolytic T lymphocytes. Cell. Immun. 155, 53–61 (1994).
Azuma, M., Cayabyab, M., Buck, D., Philips, J.H. & Lanier, L.L. Involvement of CD28 in MHC-unrestricted cytotoxicity mediated by a human natural killer leukemia cell line. J. Immun. 149, 1115–1123 (1992).
Takahashi, H., Nakada, T. & Puisieux, I. Inhibition of human colon cancer growth by antibody-directed human LAK cells in SCID mice. Science 259, 1460–1463 (1993).
Topalian, S.L., Solomon, D. & Rosenberg, S.A. Tumor-specific cytolysis by lymphocytes infiltrating human melanomas. J. Immunol. 142, 3714–3725 (1989).
Muul, L.M., Spiess, P.J., Director, E.P. & Rosenberg, S.A. Identification of specific cytolytic immune response against autologous tumor in humans bearing malignant melanoma. J. Immun. 138, 989–995 (1987).
Connors, M. et al. Pulmonary histopathology induced by respiratory syncytial virus (RSV) challenge of formalin-inactivated RSV-immunized Balb/c mice is abrogated by depletion of CD4+ T cells. J. Virol. 66, 7444–7451 (1992).
Rees, A.D. et al. Functional evidence for the recognition of endogenous peptides by autoreactive T cell clones. Int. Immun. 1, 624–630 (1989).
Lau, L.L., Jamieson, B.D., Somasundaram, T. & Ahmed, R., Cytotoxic T-cell memory without antigen. Nature 369, 648–652 (1994).
Mitra, A.B. et al. Allelotype analysis of cervical carcinoma. Cancer Res. 54, 4481–4487 (1994).
Fujino, T. et al. Allelotype of endometrial carcinoma. Cancer Res. 54, 4294–4298 (1994).
Perussia, B. et al. Preferential proliferation of natural killer cells among peripheral blood mononuclear cells cocultured with B lymphoblastoid cell lines. Nat. Immun. Cell Growth Regul. 6, 171–188 (1987).
Ucker, D.S. et al. Genome digestion is a dispensable consequence of physiological cell death mediated by cytotoxic T lymphocytes. Molec. Cell. Biol. 12, 3060–3069 (1992).
Mosmann, T. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J. immun. Methods 65, 55–63 (1983).
Saotome, K., Morita, H. & Umeda, M. Cytotoxicity test with simplified crystalviolet staining method using microtitre plates and its application to injection drugs. Toxicol. in Vitro 3, 317–321 (1989).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Liu, S., Saijo, K., Todoroki, T. et al. Induction of human autologous cytotoxic T lymphocytes on formalin-fixed and paraffin-embedded tumour sections. Nat Med 1, 267–271 (1995). https://doi.org/10.1038/nm0395-267
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm0395-267
This article is cited by
-
Long-lasting complete response status of advanced stage IV gall bladder cancer and colon cancer after combined treatment including autologous formalin-fixed tumor vaccine: two case reports
World Journal of Surgical Oncology (2017)
-
Ribonucleotide reductase represents a novel therapeutic target in primary effusion lymphoma
Oncogene (2017)
-
A gas sensor array for the simultaneous detection of multiple VOCs
Scientific Reports (2017)
-
Vaccine therapies for patients with glioblastoma
Journal of Neuro-Oncology (2014)
-
Radioresponsive tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) gene therapy for malignant brain tumors
Cancer Gene Therapy (2007)