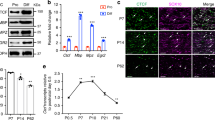
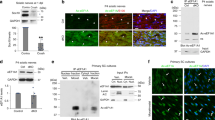
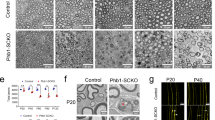
Abstract
Deficits in Schwann cell–mediated remyelination impair functional restoration after nerve damage, contributing to peripheral neuropathies. The mechanisms mediating block of remyelination remain elusive. Here, through small-molecule screening focusing on epigenetic modulators, we identified histone deacetylase 3 (HDAC3; a histone-modifying enzyme) as a potent inhibitor of peripheral myelinogenesis. Inhibition of HDAC3 enhanced myelin growth and regeneration and improved functional recovery after peripheral nerve injury in mice. HDAC3 antagonizes the myelinogenic neuregulin–PI3K–AKT signaling axis. Moreover, genome-wide profiling analyses revealed that HDAC3 represses promyelinating programs through epigenetic silencing while coordinating with p300 histone acetyltransferase to activate myelination-inhibitory programs that include the HIPPO signaling effector TEAD4 to inhibit myelin growth. Schwann cell–specific deletion of either Hdac3 or Tead4 in mice resulted in an elevation of myelin thickness in sciatic nerves. Thus, our findings identify the HDAC3–TEAD4 network as a dual-function switch of cell-intrinsic inhibitory machinery that counters myelinogenic signals and maintains peripheral myelin homeostasis, highlighting the therapeutic potential of transient HDAC3 inhibition for improving peripheral myelin repair.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Accession codes
References
Suter, U. & Scherer, S.S. Disease mechanisms in inherited neuropathies. Nat. Rev. Neurosci. 4, 714–726 (2003).
Scherer, S.S. & Wrabetz, L. Molecular mechanisms of inherited demyelinating neuropathies. Glia 56, 1578–1589 (2008).
Nave, K.A., Sereda, M.W. & Ehrenreich, H. Mechanisms of disease: inherited demyelinating neuropathies—from basic to clinical research. Nat. Clin. Pract. Neurol. 3, 453–464 (2007).
Zhou, Y. & Notterpek, L. Promoting peripheral myelin repair. Exp. Neurol. 283 (Pt. B), 573–580 (2016).
Chen, Z.L., Yu, W.M. & Strickland, S. Peripheral regeneration. Annu. Rev. Neurosci. 30, 209–233 (2007).
Nave, K.A. & Werner, H.B. Myelination of the nervous system: mechanisms and functions. Annu. Rev. Cell Dev. Biol. 30, 503–533 (2014).
Salzer, J.L. Schwann cell myelination. Cold Spring Harb. Perspect. Biol. 7, a020529 (2015).
Monk, K.R., Feltri, M.L. & Taveggia, C. New insights on Schwann cell development. Glia 63, 1376–1393 (2015).
Feltri, M.L., Poitelon, Y. & Previtali, S.C. How Schwann cells sort axons: new concepts. Neuroscientist 22, 252–265 (2016).
Stolt, C.C. & Wegner, M. Schwann cells and their transcriptional network: evolution of key regulators of peripheral myelination. Brain Res. 1641, 101–110 (2016).
Jessen, K.R., Mirsky, R. & Lloyd, A.C. Schwann cells: development and role in nerve repair. Cold Spring Harb. Perspect. Biol. 7, a020487 (2015).
Wu, L.M. et al. Zeb2 recruits HDAC-NuRD to inhibit Notch and controls Schwann cell differentiation and remyelination. Nat. Neurosci. 19, 1060–1072 (2016).
Quintes, S. et al. Zeb2 is essential for Schwann cell differentiation, myelination and nerve repair. Nat. Neurosci. 19, 1050–1059 (2016).
Poitelon, Y. et al. YAP and TAZ control peripheral myelination and the expression of laminin receptors in Schwann cells. Nat. Neurosci. 19, 879–887 (2016).
Deng, Y. et al. A reciprocal regulatory loop between TAZ/YAP and G-protein Gαs regulates Schwann cell proliferation and myelination. Nat. Commun. 8, 15161 (2017).
Grove, M. et al. YAP/TAZ initiate and maintain Schwann cell myelination. eLife 6, e20982 (2017).
Arthur-Farraj, P.J. et al. c-Jun reprograms Schwann cells of injured nerves to generate a repair cell essential for regeneration. Neuron 75, 633–647 (2012).
Emery, B. & Lu, Q.R. Transcriptional and epigenetic regulation of oligodendrocyte development and myelination in the central nervous system. Cold Spring Harb. Perspect. Biol. 7, a020461 (2015).
Pereira, J.A., Lebrun-Julien, F. & Suter, U. Molecular mechanisms regulating myelination in the peripheral nervous system. Trends Neurosci. 35, 123–134 (2012).
Ma, K.H. & Svaren, J. Epigenomic reprogramming in peripheral nerve injury. Neural Regen. Res. 11, 1930–1931 (2016).
Malvaez, M. et al. HDAC3-selective inhibitor enhances extinction of cocaine-seeking behavior in a persistent manner. Proc. Natl. Acad. Sci. USA 110, 2647–2652 (2013).
Jiang, Y. & Hsieh, J. HDAC3 controls gap 2/mitosis progression in adult neural stem/progenitor cells by regulating CDK1 levels. Proc. Natl. Acad. Sci. USA 111, 13541–13546 (2014).
Jacob, C. et al. HDAC1 and HDAC2 control the transcriptional program of myelination and the survival of Schwann cells. Nat. Neurosci. 14, 429–436 (2011).
Chen, Y. et al. HDAC-mediated deacetylation of NF-κB is critical for Schwann cell myelination. Nat. Neurosci. 14, 437–441 (2011).
Bei, F. et al. Restoration of visual function by enhancing conduction in regenerated axons. Cell 164, 219–232 (2016).
Rai, M. et al. HDAC inhibitors correct frataxin deficiency in a Friedreich ataxia mouse model. PLoS One 3, e1958 (2008).
Jia, H. et al. The effects of pharmacological inhibition of histone deacetylase 3 (HDAC3) in Huntington's disease mice. PLoS One 11, e0152498 (2016).
Parrinello, S. et al. EphB signaling directs peripheral nerve regeneration through Sox2-dependent Schwann cell sorting. Cell 143, 145–155 (2010).
Painter, M.W. et al. Diminished Schwann cell repair responses underlie age-associated impaired axonal regeneration. Neuron 83, 331–343 (2014).
Watkins, T.A., Emery, B., Mulinyawe, S. & Barres, B.A. Distinct stages of myelination regulated by γ-secretase and astrocytes in a rapidly myelinating CNS coculture system. Neuron 60, 555–569 (2008).
Montgomery, R.L. et al. Maintenance of cardiac energy metabolism by histone deacetylase 3 in mice. J. Clin. Invest. 118, 3588–3597 (2008).
Lappe-Siefke, C. et al. Disruption of Cnp1 uncouples oligodendroglial functions in axonal support and myelination. Nat. Genet. 33, 366–374 (2003).
Jaegle, M. et al. The POU proteins Brn-2 and Oct-6 share important functions in Schwann cell development. Genes Dev. 17, 1380–1391 (2003).
Doerflinger, N.H., Macklin, W.B. & Popko, B. Inducible site-specific recombination in myelinating cells. Genesis 35, 63–72 (2003).
Michailov, G.V. et al. Axonal neuregulin-1 regulates myelin sheath thickness. Science 304, 700–703 (2004).
Sheean, M.E. et al. Activation of MAPK overrides the termination of myelin growth and replaces Nrg1/ErbB3 signals during Schwann cell development and myelination. Genes Dev. 28, 290–303 (2014).
Cotter, L. et al. Dlg1–PTEN interaction regulates myelin thickness to prevent damaging peripheral nerve overmyelination. Science 328, 1415–1418 (2010).
Norrmén, C. & Suter, U. Akt/mTOR signalling in myelination. Biochem. Soc. Trans. 41, 944–950 (2013).
Okumura, K. et al. PCAF modulates PTEN activity. J. Biol. Chem. 281, 26562–26568 (2006).
Erjala, K. et al. Signaling via ErbB2 and ErbB3 associates with resistance and epidermal growth factor receptor (EGFR) amplification with sensitivity to EGFR inhibitor gefitinib in head and neck squamous cell carcinoma cells. Clin. Cancer Res. 12, 4103–4111 (2006).
Langfelder, P. & Horvath, S. WGCNA: an R package for weighted correlation network analysis. BMC Bioinformatics 9, 559 (2008).
Zhang, L. et al. Hdac3 interaction with p300 histone acetyltransferase regulates the oligodendrocyte and astrocyte lineage fate switch. Dev. Cell 36, 316–330 (2016).
Wang, Z. et al. Genome-wide mapping of HATs and HDACs reveals distinct functions in active and inactive genes. Cell 138, 1019–1031 (2009).
Creyghton, M.P. et al. Histone H3K27ac separates active from poised enhancers and predicts developmental state. Proc. Natl. Acad. Sci. USA 107, 21931–21936 (2010).
Yagi, R. et al. Transcription factor TEAD4 specifies the trophectoderm lineage at the beginning of mammalian development. Development 134, 3827–3836 (2007).
Lundborg, G. A 25-year perspective of peripheral nerve surgery: evolving neuroscientific concepts and clinical significance. J. Hand Surg. Am. 25, 391–414 (2000).
Brosius Lutz, A. & Barres, B.A. Contrasting the glial response to axon injury in the central and peripheral nervous systems. Dev. Cell 28, 7–17 (2014).
Cattin, A.L. et al. Macrophage-induced blood vessels guide Schwann cell–mediated regeneration of peripheral nerves. Cell 162, 1127–1139 (2015).
Kang, H. & Lichtman, J.W. Motor axon regeneration and muscle reinnervation in young adult and aged animals. J. Neurosci. 33, 19480–19491 (2013).
Suzuki, M. Peripheral neuropathy in the elderly. Handb. Clin. Neurol. 115, 803–813 (2013).
Cho, Y., Sloutsky, R., Naegle, K.M. & Cavalli, V. Injury-induced HDAC5 nuclear export is essential for axon regeneration. Cell 155, 894–908 (2013).
Brügger, V. et al. Delaying histone deacetylase response to injury accelerates conversion into repair Schwann cells and nerve regeneration. Nat. Commun. 8, 14272 (2017).
Haberland, M., Montgomery, R.L. & Olson, E.N. The many roles of histone deacetylases in development and physiology: implications for disease and therapy. Nat. Rev. Genet. 10, 32–42 (2009).
Sadoul, K., Boyault, C., Pabion, M. & Khochbin, S. Regulation of protein turnover by acetyltransferases and deacetylases. Biochimie 90, 306–312 (2008).
Noseda, R. et al. DDIT4/REDD1/RTP801 is a novel negative regulator of Schwann cell myelination. J. Neurosci. 33, 15295–15305 (2013).
Huang, L.L., Liu, Z.Y., Huang, J.H. & Luo, Z.J. Expression pattern of neuregulin-1 type III during the development of the peripheral nervous system. Neural Regen. Res. 10, 65–70 (2015).
Fernando, R.N. et al. Optimal myelin elongation relies on YAP activation by axonal growth and inhibition by Crb3/Hippo pathway. Nat. Commun. 7, 12186 (2016).
Koontz, L.M. et al. The Hippo effector Yorkie controls normal tissue growth by antagonizing scalloped-mediated default repression. Dev. Cell 25, 388–401 (2013).
Mottamal, M., Zheng, S., Huang, T.L. & Wang, G. Histone deacetylase inhibitors in clinical studies as templates for new anticancer agents. Molecules 20, 3898–3941 (2015).
Li, Y. & Seto, E. HDACs and HDAC inhibitors in cancer development and therapy. Cold Spring Harb. Perspect. Med. 6, a026831 (2016).
Acknowledgements
We thank M. Wegner, J. Wells, and E. Hurlock for critical reading of the manuscript. We are grateful to E. Olson (University of Texas Southwestern Medical Center), D. Meijer (University of Edinburgh), and M. Wegner (Friedrich-Alexander-Universität Erlangen-Nürnberg (FAU)) for Hdac3-floxed mice, Dhh-Cre mice, and antibodies against EGR2 and KROX20, respectively, and to N. Wu and L. Xu for technical support. This study was funded in part by the US National Institutes of Health (NIH; grant no. R37NS096359 and R01NS075243 to Q.R.L.; R35NS097303 to B.D.T.; and R01AR064551-01A1 to M.P.J.) and the National Multiple Sclerosis Society (grant no. NMSS-RG1507 to Q.R.L.).
Author information
Authors and Affiliations
Contributions
X.H., L.Z., and Q.R.L. designed the experiments, analyzed the data, and wrote the manuscript with input from all authors. L.F.Q. and M.P.J. carried out CMAP analysis. X.H., L.Z., X.L., A.L., G.K., and X.D. performed the in vitro, in vivo, gene profiling, ChIP–seq, and in silico analyses. R.R.W., W.Z., S.-O.Y., J.B.R., M.X., K.-A.N., and B.D.T. provided resources and inputs. A.B. and K.N. provided floxed Tead4 and Cnp-Cre animals, respectively. Q.R.L. supervised the project.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Figures
Supplementary Figures 1–27 (PDF 3467 kb)
Supplementary Table 1
Transcription Factor loci with chromatin co-occupancy of HDAC3 and p300 (XLSX 13 kb)
Rights and permissions
About this article
Cite this article
He, X., Zhang, L., Queme, L. et al. A histone deacetylase 3–dependent pathway delimits peripheral myelin growth and functional regeneration. Nat Med 24, 338–351 (2018). https://doi.org/10.1038/nm.4483
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.4483
This article is cited by
-
Genes in Axonal Regeneration
Molecular Neurobiology (2024)
-
HDAC3 Inhibition Stimulates Myelination in a CMT1A Mouse Model
Molecular Neurobiology (2022)
-
Decellularized extracellular matrix mediates tissue construction and regeneration
Frontiers of Medicine (2022)
-
The role of histone deacetylase 3 in breast cancer
Medical Oncology (2022)
-
Sodium phenylbutyrate inhibits Schwann cell inflammation via HDAC and NFκB to promote axonal regeneration and remyelination
Journal of Neuroinflammation (2021)