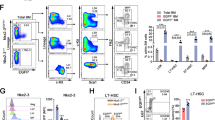
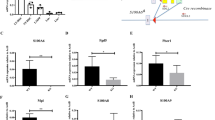
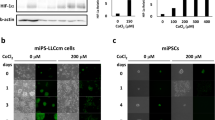
Abstract
Hematopoietic stem cells (HSCs) quiescently reside in bone marrow niches and have the capacity to self-renew or differentiate to form all of the blood cells throughout the lifespan of an animal1,2,3. Allogeneic HSC transplantation is a life-saving treatment for malignant and nonmalignant disorders4,5. HSCs isolated from umbilical cord blood (CB) are used for hematopoietic cell transplantation (HCT)6,7,8,9,10,11, but due to the limited numbers of HSCs in single units of umbilical CB, a number of methods have been proposed for ex vivo expansion of human HSCs7,8,12. We show here that antagonism of peroxisome proliferator-activated receptor (PPAR)-γ promotes ex vivo expansion of phenotypically and functionally defined subsets of human CB HSCs and hematopoietic progenitor cells (HSPCs). PPAR-γ antagonism in CB HSPCs strongly downregulated expression of several differentiation-associated genes, as well as fructose-bisphosphatase 1 (FBP1; which encodes a negative regulator of glycolysis), and enhanced glycolysis without compromising mitochondrial metabolism. The expansion of CB HSPCs by PPAR-γ antagonism was completely suppressed by removal of glucose or inhibition of glycolysis. Moreover, knockdown of FBP1 expression promoted glycolysis and ex vivo expansion of long-term repopulating CB HSPCs, whereas overexpression of FBP1 suppressed the expansion of CB HSPCs that was induced by PPAR-γ antagonism. Our study suggests the possibility for a new and simple means for metabolic reprogramming of CB HSPCs to improve the efficacy of HCT.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
References
Doulatov, S., Notta, F., Laurenti, E. & Dick, J.E. Hematopoiesis: a human perspective. Cell Stem Cell 10, 120–136 (2012).
Morrison, S.J. & Scadden, D.T. The bone marrow niche for hematopoietic stem cells. Nature 505, 327–334 (2014).
Eaves, C.J. Hematopoietic stem cells: concepts, definitions, and the new reality. Blood 125, 2605–2613 (2015).
Copelan, E.A. Hematopoietic stem cell transplantation. N. Engl. J. Med. 354, 1813–1826 (2006).
Singh, A.K. & McGuirk, J.P. Allogeneic stem cell transplantation: a historical and scientific overview. Cancer Res. 76, 6445–6451 (2016).
Broxmeyer, H.E. et al. Human umbilical cord blood as a potential source of transplantable hematopoietic stem/progenitor cells. Proc. Natl. Acad. Sci. USA 86, 3828–3832 (1989).
Ballen, K.K., Gluckman, E. & Broxmeyer, H.E. Umbilical cord blood transplantation: the first 25 years and beyond. Blood 122, 491–498 (2013).
Broxmeyer, H.E. & Farag, S. Background and future considerations for human cord blood hematopoietic cell transplantation, including economic concerns. Stem Cells Dev. 22 (Suppl. 1), 103–110 (2013).
Milano, F. et al. Cord blood transplantation in patients with minimal residual disease. N. Engl. J. Med. 375, 944–953 (2016).
Fares, I. et al. Cord blood expansion. Pyrimidoindole derivatives are agonists of human hematopoietic stem cell self-renewal. Science 345, 1509–1512 (2014).
Wagner, J.E. Jr. et al. Phase 1/2 trial of StemRegenin-1-expanded umbilical cord blood hematopoietic stem cells supports testing as a stand-alone graft. Cell Stem Cell 18, 144–155 (2016).
Nikiforow, S. & Ritz, J. Dramatic expansion of HSCs: new possibilities for HSC transplants? Cell Stem Cell 18, 10–12 (2016).
Mantel, C.R. et al. Enhancing hematopoietic stem cell transplantation efficacy by mitigating oxygen shock. Cell 161, 1553–1565 (2015).
Csaszar, E. et al. Rapid expansion of human hematopoietic stem cells by automated control of inhibitory feedback signaling. Cell Stem Cell 10, 218–229 (2012).
Boitano, A.E. et al. Aryl hydrocarbon receptor antagonists promote the expansion of human hematopoietic stem cells. Science 329, 1345–1348 (2010).
Chaurasia, P., Gajzer, D.C., Schaniel, C., D'Souza, S. & Hoffman, R. Epigenetic reprogramming induces the expansion of cord blood stem cells. J. Clin. Invest. 124, 2378–2395 (2014).
Notta, F. et al. Isolation of single human hematopoietic stem cells capable of long-term multilineage engraftment. Science 333, 218–221 (2011).
Guo, B., Huang, X., Cooper, S. & Broxmeyer, H.E. Glucocorticoid-hormone-induced chromatin remodeling enhances human hematopoietic stem cell homing and engraftment. Nat. Med. 23, 424–428 (2017).
Sun, J. et al. Clonal dynamics of native haematopoiesis. Nature 514, 322–327 (2014).
Shubinsky, G. & Schlesinger, M. The CD38 lymphocyte differentiation marker: new insight into its ectoenzymatic activity and its role as a signal transducer. Immunity 7, 315–324 (1997).
Wei, D.G. et al. Expansion and long-range differentiation of the NKT cell lineage in mice expressing CD1d exclusively on cortical thymocytes. J. Exp. Med. 202, 239–248 (2005).
Lee, J.H., Ulrich, B., Cho, J., Park, J. & Kim, C.H. Progesterone promotes differentiation of human cord blood fetal T cells into T regulatory cells but suppresses their differentiation into TH17 cells. J. Immunol. 187, 1778–1787 (2011).
Wang, X. et al. Inactivation of a novel FGF23 regulator, FAM20C, leads to hypophosphatemic rickets in mice. PLoS Genet. 8, e1002708 (2012).
Su, H. et al. PRMT1-mediated methylation of DUSP4 determines megakaryocyte–erythroid lineage choice by regulating p38 signaling. Blood 126, 2387 (2015).
Rönn, R.E. et al. Retinoic acid regulates hematopoietic development from human pluripotent stem cells. Stem Cell Rep. 4, 269–281 (2015).
Dong, C. et al. Loss of FBP1 by Snail-mediated repression provides metabolic advantages in basal-like breast cancer. Cancer Cell 23, 316–331 (2013).
Li, B. et al. Fructose-1,6-bisphosphatase opposes renal carcinoma progression. Nature 513, 251–255 (2014).
Wang, Y.H. et al. Cell-state-specific metabolic dependency in hematopoiesis and leukemogenesis. Cell 158, 1309–1323 (2014).
Ito, K. et al. A PML–PPAR-δ pathway for fatty acid oxidation regulates hematopoietic stem cell maintenance. Nat. Med. 18, 1350–1358 (2012).
Brown, J. Effects of 2-deoxyglucose on carbohydrate metablism: review of the literature and studies in the rat. Metabolism 11, 1098–1112 (1962).
Erion, M.D. et al. MB06322 (CS-917): A potent and selective inhibitor of fructose 1,6-bisphosphatase for controlling gluconeogenesis in type 2 diabetes. Proc. Natl. Acad. Sci. USA 102, 7970–7975 (2005).
Huang, X. et al. Activation of OCT4 enhances ex vivo expansion of human cord blood hematopoietic stem and progenitor cells by regulating HOXB4 expression. Leukemia 30, 144–153 (2016).
Yang, X.O. et al. T helper 17 lineage differentiation is programmed by orphan nuclear receptors ROR-α and ROR-γ. Immunity 28, 29–39 (2008).
Wu, L. et al. Inhibition of PPAR-γ in myeloid-lineage cells induces systemic inflammation, immunosuppression, and tumorigenesis. Blood 119, 115–126 (2012).
Evans, R.M. & Mangelsdorf, D.J. Nuclear receptors, RXR, and the Big Bang. Cell 157, 255–266 (2014).
Jeong, Y. & Mangelsdorf, D.J. Nuclear receptor regulation of stemness and stem cell differentiation. Exp. Mol. Med. 41, 525–537 (2009).
Purton, L.E., Bernstein, I.D. & Collins, S.J. All-trans retinoic acid enhances the long-term repopulating activity of cultured hematopoietic stem cells. Blood 95, 470–477 (2000).
Purton, L.E. et al. RAR-γ is critical for maintaining a balance between hematopoietic stem cell self-renewal and differentiation. J. Exp. Med. 203, 1283–1293 (2006).
Chute, J.P. et al. Inhibition of aldehyde dehydrogenase and retinoid signaling induces the expansion of human hematopoietic stem cells. Proc. Natl. Acad. Sci. USA 103, 11707–11712 (2006).
Ghiaur, G. et al. Regulation of human hematopoietic stem cell self-renewal by the microenvironment's control of retinoic acid signaling. Proc. Natl. Acad. Sci. USA 110, 16121–16126 (2013).
Acknowledgements
We thank S. Messina-Graham for technical support with the Seahorse assay, other members of the Broxmeyer laboratory for helpful discussions and assistance, and A.L. Sinn (In vivo Therapeutics Core, Indiana University School of Medicine) for assistance with the transplantation experiments. This work was supported by US Public Health Service Grants from the NIH (R01 HL112669, R01 HL056416 and U54 DK106846; all to H.E.B.) and by the National Research Foundation, which is funded by the Korean government (NRF-2017M3A9C6033069 to M.R.L.).
Author information
Authors and Affiliations
Contributions
B.G. conceived the research, designed and performed experiments, interpreted data and wrote the manuscript; X.H. designed and performed the Seahorse extracellular flux assays and transplantation, and interpreted data; M.R.L. and S.A.L. designed the teratoma formation experiment and analyzed the results; H.E.B. supervised the study, designed and performed experiments, interpreted data and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
H.E.B. is a member of the Medical Scientific Advisory Board of CordUse, a cord blood banking company based in Orlando, Florida.
Supplementary information
Supplementary Figures & Tables
Supplementary Figures 1–13 & Supplementary Tables 2–5,7–9 (PDF 6111 kb)
Supplementary Table 1
GW9662 treatment promotes ex vivo expansion of CB phenotypic HSPCs (XLS 17 kb)
Supplementary Table 6
MB05032 treatment promotes ex vivo expansion of CB phenotypic HSPCs (XLS 17 kb)
Rights and permissions
About this article
Cite this article
Guo, B., Huang, X., Lee, M. et al. Antagonism of PPAR-γ signaling expands human hematopoietic stem and progenitor cells by enhancing glycolysis. Nat Med 24, 360–367 (2018). https://doi.org/10.1038/nm.4477
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.4477
This article is cited by
-
Targeted knockdown of PGAM5 in synovial macrophages efficiently alleviates osteoarthritis
Bone Research (2024)
-
Nuclear pore protein POM121 regulates subcellular localization and transcriptional activity of PPARγ
Cell Death & Disease (2024)
-
Transient regulation of RNA methylation in human hematopoietic stem cells promotes their homing and engraftment
Leukemia (2023)
-
Low doses of IFN-γ maintain self-renewal of leukemia stem cells in acute myeloid leukemia
Oncogene (2023)
-
Melatonin inhibits bladder tumorigenesis by suppressing PPARγ/ENO1-mediated glycolysis
Cell Death & Disease (2023)