Abstract
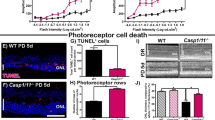
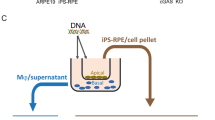
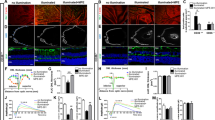
Geographic atrophy is a blinding form of age-related macular degeneration characterized by retinal pigmented epithelium (RPE) death; the RPE also exhibits DICER1 deficiency, resultant accumulation of endogenous Alu-retroelement RNA, and NLRP3-inflammasome activation. How the inflammasome is activated in this untreatable disease is largely unknown. Here we demonstrate that RPE degeneration in human-cell-culture and mouse models is driven by a noncanonical-inflammasome pathway that activates caspase-4 (caspase-11 in mice) and caspase-1, and requires cyclic GMP-AMP synthase (cGAS)-dependent interferon-β production and gasdermin D–dependent interleukin-18 secretion. Decreased DICER1 levels or Alu-RNA accumulation triggers cytosolic escape of mitochondrial DNA, which engages cGAS. Moreover, caspase-4, gasdermin D, interferon-β, and cGAS levels were elevated in the RPE in human eyes with geographic atrophy. Collectively, these data highlight an unexpected role of cGAS in responding to mobile-element transcripts, reveal cGAS-driven interferon signaling as a conduit for mitochondrial-damage-induced inflammasome activation, expand the immune-sensing repertoire of cGAS and caspase-4 to noninfectious human disease, and identify new potential targets for treatment of a major cause of blindness.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Change history
01 January 2018
In the version of this article initially published online, a micrograph in Figure 1d (WT, Vehicle) and a micrograph in Figure 2a (Gsdmd−/−, Alu RNA) are duplicates of two other micrographs and are incorrect. These errors do not affect the quantitative data reported or the conclusions of the article. These two micrographs have been replaced with the correct ones in the print, PDF and HTML versions of this article.
References
Wong, W.L. et al. Global prevalence of age-related macular degeneration and disease burden projection for 2020 and 2040: a systematic review and meta-analysis. Lancet Glob. Health 2, e106–e116 (2014).
Ambati, J., Ambati, B.K., Yoo, S.H., Ianchulev, S. & Adamis, A.P. Age-related macular degeneration: etiology, pathogenesis, and therapeutic strategies. Surv. Ophthalmol. 48, 257–293 (2003).
Ambati, J., Atkinson, J.P. & Gelfand, B.D. Immunology of age-related macular degeneration. Nat. Rev. Immunol. 13, 438–451 (2013).
Kaneko, H. et al. DICER1 deficit induces Alu RNA toxicity in age-related macular degeneration. Nature 471, 325–330 (2011).
Tarallo, V. et al. DICER1 loss and Alu RNA induce age-related macular degeneration via the NLRP3 inflammasome and MyD88. Cell 149, 847–859 (2012).
Anderson, O.A., Finkelstein, A. & Shima, D.T. A2E induces IL-1ß production in retinal pigment epithelial cells via the NLRP3 inflammasome. PLoS One 8, e67263 (2013).
Tseng, W.A. et al. NLRP3 inflammasome activation in retinal pigment epithelial cells by lysosomal destabilization: implications for age-related macular degeneration. Invest. Ophthalmol. Vis. Sci. 54, 110–120 (2013).
Gelfand, B.D. et al. Iron toxicity in the retina requires Alu RNA and the NLRP3 Inflammasome. Cell Rep. 11, 1686–1693 (2015).
Kayagaki, N. et al. Noncanonical inflammasome activation by intracellular LPS independent of TLR4. Science 341, 1246–1249 (2013).
Rathinam, V.A. et al. TRIF licenses caspase-11-dependent NLRP3 inflammasome activation by gram-negative bacteria. Cell 150, 606–619 (2012).
Zanoni, I. et al. An endogenous caspase-11 ligand elicits interleukin-1 release from living dendritic cells. Science 352, 1232–1236 (2016).
Kayagaki, N. et al. Non-canonical inflammasome activation targets caspase-11. Nature 479, 117–121 (2011).
Kleinman, M.E. et al. Sequence- and target-independent angiogenesis suppression by siRNA via TLR3. Nature 452, 591–597 (2008).
Kleinman, M.E. et al. Short-interfering RNAs induce retinal degeneration via TLR3 and IRF3. Mol. Ther. 20, 101–108 (2012).
Kajiwara, Y. et al. A critical role for human caspase-4 in endotoxin sensitivity. J. Immunol. 193, 335–343 (2014).
Fowler, B.J. et al. Nucleoside reverse transcriptase inhibitors possess intrinsic anti-inflammatory activity. Science 346, 1000–1003 (2014).
Kerur, N. et al. TLR-independent and P2X7-dependent signaling mediate Alu RNA-induced NLRP3 inflammasome activation in geographic atrophy. Invest. Ophthalmol. Vis. Sci. 54, 7395–7401 (2013).
Shi, J. et al. Cleavage of GSDMD by inflammatory caspases determines pyroptotic cell death. Nature 526, 660–665 (2015).
Kayagaki, N. et al. Caspase-11 cleaves gasdermin D for non-canonical inflammasome signalling. Nature 526, 666–671 (2015).
Liu, X. et al. Inflammasome-activated gasdermin D causes pyroptosis by forming membrane pores. Nature 535, 153–158 (2016).
Aglietti, R.A. et al. GsdmD p30 elicited by caspase-11 during pyroptosis forms pores in membranes. Proc. Natl. Acad. Sci. USA 113, 7858–7863 (2016).
Kim, Y. et al. DICER1/Alu RNA dysmetabolism induces Caspase-8-mediated cell death in age-related macular degeneration. Proc. Natl. Acad. Sci. USA 111, 16082–16087 (2014).
Dunaief, J.L., Dentchev, T., Ying, G.S. & Milam, A.H. The role of apoptosis in age-related macular degeneration. Arch. Ophthalmol. 120, 1435–1442 (2002).
van Genderen, H. et al. In vitro measurement of cell death with the annexin A5 affinity assay. Nat. Protoc. 1, 363–367 (2006).
Fink, S.L., Bergsbaken, T. & Cookson, B.T. Anthrax lethal toxin and Salmonella elicit the common cell death pathway of caspase-1-dependent pyroptosis via distinct mechanisms. Proc. Natl. Acad. Sci. USA 105, 4312–4317 (2008).
Sun, L., Wu, J., Du, F., Chen, X. & Chen, Z.J. Cyclic GMP-AMP synthase is a cytosolic DNA sensor that activates the type I interferon pathway. Science 339, 786–791 (2013).
Schoggins, J.W. et al. Pan-viral specificity of IFN-induced genes reveals new roles for cGAS in innate immunity. Nature 505, 691–695 (2014).
Feher, J. et al. Mitochondrial alterations of retinal pigment epithelium in age-related macular degeneration. Neurobiol. Aging 27, 983–993 (2006).
Terluk, M.R. et al. Investigating mitochondria as a target for treating age-related macular degeneration. J. Neurosci. 35, 7304–7311 (2015).
West, A.P. et al. Mitochondrial DNA stress primes the antiviral innate immune response. Nature 520, 553–557 (2015).
Galluzzi, L., Blomgren, K. & Kroemer, G. Mitochondrial membrane permeabilization in neuronal injury. Nat. Rev. Neurosci. 10, 481–494 (2009).
Baines, C.P. et al. Loss of cyclophilin D reveals a critical role for mitochondrial permeability transition in cell death. Nature 434, 658–662 (2005).
McLeod, D.S. et al. Distribution and quantification of choroidal macrophages in human eyes with age-related macular degeneration. Invest. Ophthalmol. Vis. Sci. 57, 5843–5855 (2016).
Sakurai, E., Anand, A., Ambati, B.K., van Rooijen, N. & Ambati, J. Macrophage depletion inhibits experimental choroidal neovascularization. Invest. Ophthalmol. Vis. Sci. 44, 3578–3585 (2003).
Wang, X. et al. Requirement for microglia for the maintenance of synaptic function and integrity in the mature retina. J. Neurosci. 36, 2827–2842 (2016).
Karijolich, J., Abernathy, E. & Glaunsinger, B.A. Infection-induced retrotransposon-derived noncoding RNAs enhance herpesviral gene expression via the NF-κB pathway. PLoS Pathog. 11, e1005260 (2015).
Nunnari, J. & Suomalainen, A. Mitochondria: in sickness and in health. Cell 148, 1145–1159 (2012).
West, A.P., Shadel, G.S. & Ghosh, S. Mitochondria in innate immune responses. Nat. Rev. Immunol. 11, 389–402 (2011).
Fang, C., Wei, X. & Wei, Y. Mitochondrial DNA in the regulation of innate immune responses. Protein Cell 7, 11–16 (2016).
Shimada, K. et al. Oxidized mitochondrial DNA activates the NLRP3 inflammasome during apoptosis. Immunity 36, 401–414 (2012).
Nakahira, K. et al. Autophagy proteins regulate innate immune responses by inhibiting the release of mitochondrial DNA mediated by the NALP3 inflammasome. Nat. Immunol. 12, 222–230 (2011).
Zhang, Q., Itagaki, K. & Hauser, C.J. Mitochondrial DNA is released by shock and activates neutrophils via p38 map kinase. Shock 34, 55–59 (2010).
Yan, Y. et al. Dicer expression exhibits a tissue-specific diurnal pattern that is lost during aging and in diabetes. PLoS One 8, e80029 (2013).
Hung, T. et al. The Ro60 autoantigen binds endogenous retroelements and regulates inflammatory gene expression. Science 350, 455–459 (2015).
Hitomi, J. et al. Involvement of caspase-4 in endoplasmic reticulum stress-induced apoptosis and Aβbeta-induced cell death. J. Cell Biol. 165, 347–356 (2004).
Katayama, T. et al. Presenilin-1 mutations downregulate the signalling pathway of the unfolded-protein response. Nat. Cell Biol. 1, 479–485 (1999).
Ozcan, U. et al. Endoplasmic reticulum stress links obesity, insulin action, and type 2 diabetes. Science 306, 457–461 (2004).
Yang, D., He, Y., Muñoz-Planillo, R., Liu, Q. & Núñez, G. Caspase-11 requires the Pannexin 1 channel and the purinergic P2X7 pore to mediate pyroptosis and endotoxic shock. Immunity 43, 923–932 (2015).
Shaw, P.X. et al. Complement factor H genotypes impact risk of age-related macular degeneration by interaction with oxidized phospholipids. Proc. Natl. Acad. Sci. USA 109, 13757–13762 (2012).
Suzuki, M. et al. Oxidized phospholipids in the macula increase with age and in eyes with age-related macular degeneration. Mol. Vis. 13, 772–778 (2007).
Brandstetter, C., Mohr, L.K., Latz, E., Holz, F.G. & Krohne, T.U. Light induces NLRP3 inflammasome activation in retinal pigment epithelial cells via lipofuscin-mediated photooxidative damage. J. Mol. Med. (Berl.) 93, 905–916 (2015).
Kauppinen, A. et al. Oxidative stress activates NLRP3 inflammasomes in ARPE-19 cells--implications for age-related macular degeneration (AMD). Immunol. Lett. 147, 29–33 (2012).
Marneros, A.G. NLRP3 inflammasome blockade inhibits VEGF-A-induced age-related macular degeneration. Cell Rep. 4, 945–958 (2013).
Doyle, S.L. et al. NLRP3 has a protective role in age-related macular degeneration through the induction of IL-18 by drusen components. Nat. Med. 18, 791–798 (2012).
Wallach, D., Kang, T.B., Dillon, C.P. & Green, D.R. Programmed necrosis in inflammation: toward identification of the effector molecules. Science 352, aaf2154 (2016).
Man, S.M. & Kanneganti, T.D. Converging roles of caspases in inflammasome activation, cell death and innate immunity. Nat. Rev. Immunol. 16, 7–21 (2016).
Hirano, Y. et al. IL-18 is not therapeutic for neovascular age-related macular degeneration. Nat. Med. 20, 1372–1375 (2014).
Ambati, J. & Fowler, B.J. Mechanisms of age-related macular degeneration. Neuron 75, 26–39 (2012).
Mariathasan, S. et al. Differential activation of the inflammasome by caspase-1 adaptors ASC and Ipaf. Nature 430, 213–218 (2004).
Kanneganti, T.D. et al. Bacterial RNA and small antiviral compounds activate caspase-1 through cryopyrin/Nalp3. Nature 440, 233–236 (2006).
Müller, U. et al. Functional role of type I and type II interferons in antiviral defense. Science 264, 1918–1921 (1994).
Sato, M. et al. Distinct and essential roles of transcription factors IRF-3 and IRF-7 in response to viruses for IFN-alpha/beta gene induction. Immunity 13, 539–548 (2000).
Suschak, J.J., Wang, S., Fitzgerald, K.A. & Lu, S. A cGAS-independent STING/IRF7 pathway mediates the immunogenicity of DNA vaccines. J. Immunol. 196, 310–316 (2016).
Jin, L. et al. MPYS is required for IFN response factor 3 activation and type I IFN production in the response of cultured phagocytes to bacterial second messengers cyclic-di-AMP and cyclic-di-GMP. J. Immunol. 187, 2595–2601 (2011).
Bennett, E.A. et al. Active Alu retrotransposons in the human genome. Genome Res. 18, 1875–1883 (2008).
Shaikh, T.H., Roy, A.M., Kim, J., Batzer, M.A. & Deininger, P.L. cDNAs derived from primary and small cytoplasmic Alu (scAlu) transcripts. J. Mol. Biol. 271, 222–234 (1997).
Nagai, H. et al. Gene transfer of secreted-type modified interleukin-18 gene to B16F10 melanoma cells suppresses in vivo tumor growth through inhibition of tumor vessel formation. J. Invest. Dermatol. 119, 541–548 (2002).
Huang, J. et al. Comparison of noncontact specular and confocal microscopy for evaluation of corneal endothelium. Eye Contact Lens https://doi.org/10.1097/ICL.0000000000000362 (2017).
Laing, R.A., Sanstrom, M.M., Berrospi, A.R. & Leibowitz, H.M. Changes in the corneal endothelium as a function of age. Exp. Eye Res. 22, 587–594 (1976).
Doughty, M.J., Müller, A. & Zaman, M.L. Assessment of the reliability of human corneal endothelial cell-density estimates using a noncontact specular microscope. Cornea 19, 148–158 (2000).
Inaba, M., Matsuda, M., Shiozaki, Y. & Kosaki, H. Regional specular microscopy of endothelial cell loss after intracapsular cataract extraction: a preliminary report. Acta Ophthalmol. (Copenh.) 63, 232–235 (1985).
Laing, R.A., Sandstrom, M.M. & Leibowitz, H.M. In vivo photomicrography of the corneal endothelium. Arch. Ophthalmol. 93, 143–145 (1975).
Ach, T. et al. Lipofuscin redistribution and loss accompanied by cytoskeletal stress in retinal pigment epithelium of eyes with age-related macular degeneration. Invest. Ophthalmol. Vis. Sci. 56, 3242–3252 (2015).
Dridi, S. et al. ERK1/2 activation is a therapeutic target in age-related macular degeneration. Proc. Natl. Acad. Sci. USA 109, 13781–13786 (2012).
Grossniklaus, H.E., Nickerson, J.M., Edelhauser, H.F., Bergman, L.A. & Berglin, L. Anatomic alterations in aging and age-related diseases of the eye. Invest. Ophthalmol. Vis. Sci. 54, ORSF23–ORSF7 (2013).
Bannwarth, S., Procaccio, V. & Paquis-Flucklinger, V. Rapid identification of unknown heteroplasmic mutations across the entire human mitochondrial genome with mismatch-specific Surveyor Nuclease. Nat. Protoc. 1, 2037–2047 (2006).
Yang, P., Tyrrell, J., Han, I. & Jaffe, G.J. Expression and modulation of RPE cell membrane complement regulatory proteins. Invest. Ophthalmol. Vis. Sci. 50, 3473–3481 (2009).
Miceli, M.V. & Jazwinski, S.M. Nuclear gene expression changes due to mitochondrial dysfunction in ARPE-19 cells: implications for age-related macular degeneration. Invest. Ophthalmol. Vis. Sci. 46, 1765–1773 (2005).
Kagan, V.E. et al. Oxidized arachidonic and adrenic PEs navigate cells to ferroptosis. Nat. Chem. Biol. 13, 81–90 (2017).
Wang, X. et al. Polydatin, a natural polyphenol, protects arterial smooth muscle cells against mitochondrial dysfunction and lysosomal destabilization following hemorrhagic shock. Am. J. Physiol. Regul. Integr. Comp. Physiol. 302, R805–R814 (2012).
Bonora, M. et al. Comprehensive analysis of mitochondrial permeability transition pore activity in living cells using fluorescence-imaging-based techniques. Nat. Protoc. 11, 1067–1080 (2016).
Acknowledgements
We thank Z. Chen, V. Tarallo, and P. Pinton for valuable discussions. We thank H. Virgin (Washington University in St. Louis), G. Shadel (Yale University), W. T. Wong (NIH), L. Zhao (NIH), and V. Dixit (Genentech) for reagents. We thank G. Pattison, E. Ghias, K. Langberg, D. Robertson, E. Doswell, X. Zhou, K. Atwood, R. Makin, O. Kirillina, A. Bobrov, E. Dinning, L. Pandya, C. Payne, G. Botzet, N. Bell, R. King, L. Xu, L. Toll, and A. Uittenbogaard for technical assistance, and the University of Kentucky Viral Core (COBRE) for providing lentiviral vectors. J.A. was supported by NIH grants (DP1GM114862, R01EY018350, R01EY018836, R01EY020672, R01EY022238, and R01EY024068), a Doris Duke Distinguished Clinical Scientist Award, a Burroughs Wellcome Fund Clinical Scientist Award in Translational Research, an Ellison Medical Foundation Senior Scholar in Aging Award, and the John Templeton Foundation; as the Dr. E. Vernon Smith and Eloise C. Smith Macular Degeneration Endowed Chair; and through a DuPont Guerry, III Professorship. N.K. was supported by NIH grants K99EY024336 and R00EY024336, and the Beckman Initiative for Macular Research (BIMR). S.F. was supported by a Research Grant from the Japan Eye Bank Association and Society for the Promotion of Science (JSPS) Overseas Research Fellowship. B.J.F. was supported by NIH grants T32HL091812 and UL1RR033173. R.Y. was supported by an Association for Research in Vision and Ophthalmology (ARVO)/Alcon Early Career Clinician-Scientist Research Award. T.Y. was supported by a Fight for Sight postdoctoral award. A.B.-C. was supported by the Programme for Advanced Medical Education (sponsored by Fundação Calouste Gulbenkian, Fundação Champalimaud, Ministério da Saúde and Fundação para a Ciência e Tecnologia, Portugal). D.R.H. was supported by NIH R01EY001545 and an unrestricted departmental grant from Research to Prevent Blindness. J.C.C. was supported by NIH R21AI099346. S.F. was supported by a Research Grant from the Japan Eye Bank Association. S.M.J. was supported by NIH R37AG006168. B.D.G. was supported by the American Heart Association and by the National Center for Research Resources and the National Center for Advancing Translational Sciences, National Institutes of Health, through grant UL1TR000117. V.S. was supported by NIH grants T32GM007055 and F31DK108553. N.L. was supported by NIH grants R01DK096076 and P01 HL120840. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
Author information
Authors and Affiliations
Contributions
N.K., S.F., D.B., Y. Kim, D.F., I.A., A.V., R.Y., B.J.F., E.B., K.M.M., X.H., T.Y., Y.H., V.S., M.A., V.L.A., N.L., K.A., S.H., A.B.-C., and B.D.G. performed experiments or analyzed data. Y. Kajiwara, D.R.H., J.C.C., J.D.B., A.P.W., S.M.J., M.C.K., K.A.F., K.B.K., Y.O., H.T., H.N., I.H., and T.O. contributed mice, tissues or reagents. J.A. and N.K. conceived and directed the project, and wrote the paper with assistance from B.J.F., B.D.G., N.L., K.A., and S.R.S. All authors had the opportunity to discuss the results and comment on the manuscript.
Corresponding authors
Ethics declarations
Competing interests
J.A. is a cofounder of iVeena Holdings, iVeena Pharmaceuticals, iVeena Delivery Systems, and Inflammasome Therapeutics, and has been a consultant for Allergan, Biogen, and Olix Pharmaceuticals in a capacity unrelated to this work. J.A., N.K., B.J.F., and K.A. are named as inventors on patent applications on macular degeneration filed by the University of Kentucky or the University of Virginia.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–16 (PDF 6052 kb)
Rights and permissions
About this article
Cite this article
Kerur, N., Fukuda, S., Banerjee, D. et al. cGAS drives noncanonical-inflammasome activation in age-related macular degeneration. Nat Med 24, 50–61 (2018). https://doi.org/10.1038/nm.4450
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.4450