Abstract
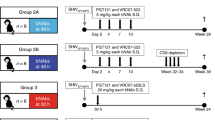
Prevention of mother-to-child transmission (MTCT) of HIV remains a major objective where antenatal care is not readily accessible. We tested HIV-1–specific human neutralizing monoclonal antibodies (NmAbs) as a post-exposure therapy in an infant macaque model for intrapartum MTCT. One-month-old rhesus macaques were inoculated orally with the simian-human immunodeficiency virus SHIVSF162P3. On days 1, 4, 7 and 10 after virus exposure, we injected animals subcutaneously with NmAbs and quantified systemic distribution of NmAbs in multiple tissues within 24 h after antibody administration. Replicating virus was found in multiple tissues by day 1 in animals that were not treated. All NmAb-treated macaques were free of virus in blood and tissues at 6 months after exposure. We detected no anti-SHIV T cell responses in blood or tissues at necropsy, and no virus emerged after CD8+ T cell depletion. These results suggest that early passive immunotherapy can eliminate early viral foci and thereby prevent the establishment of viral reservoirs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Mofenson, L.M. Prevention of mother-to-child HIV-1 transmission—why we still need a preventive HIV immunization strategy. J. Acquir. Immune Defic. Syndr. 58, 359–362 (2011).
Safrit, J.T. et al. Immunoprophylaxis to prevent mother-to-child transmission of HIV-1. J. Acquir. Immune Defic. Syndr. 35, 169–177 (2004).
Voronin, Y. et al. HIV monoclonal antibodies: a new opportunity to further reduce mother-to-child HIV transmission. PLoS Med. 11, e1001616 (2014).
Mofenson, L.M. & Watts, D.H. Safety of pediatric HIV elimination: the growing population of HIV- and antiretroviral-exposed, but uninfected, infants. PLoS Med. 11, e1001636 (2014).
Kumwenda, N.I. et al. Extended antiretroviral prophylaxis to reduce breast-milk HIV-1 transmission. N. Engl. J. Med. 359, 119–129 (2008).
Persaud, D. et al. Slower clearance of nevirapine-resistant virus in infants failing extended nevirapine prophylaxis for prevention of mother-to-child HIV transmission. AIDS Res. Hum. Retroviruses 27, 823–829 (2011).
Carias, A.M. et al. Defining the interaction of HIV-1 with the mucosal barriers of the female reproductive tract. J. Virol. 87, 11388–11400 (2013).
Demberg, T. & Robert-Guroff, M. Mucosal immunity and protection against HIV-SIV infection: strategies and challenges for vaccine design. Int. Rev. Immunol. 28, 20–48 (2009).
Haase, A.T. Early events in sexual transmission of HIV and SIV, and opportunities for interventions. Annu. Rev. Med. 62, 127–139 (2011).
Milush, J.M. et al. Rapid dissemination of SIV following oral inoculation. AIDS 18, 2371–2380 (2004).
Abel, K. et al. Rapid virus dissemination in infant macaques after oral simian immunodeficiency virus exposure in the presence of local innate immune responses. J. Virol. 80, 6357–6367 (2006).
Devito, C. et al. Mucosal and plasma IgA from HIV-1-exposed uninfected individuals inhibit HIV-1 transcytosis across human epithelial cells. J. Immunol. 165, 5170–5176 (2000).
Mascola, J.R. et al. Protection of macaques against vaginal transmission of a pathogenic HIV-1–SIV chimeric virus by passive infusion of neutralizing antibodies. Nat. Med. 6, 207–210 (2000).
Baba, T.W. et al. Human neutralizing monoclonal antibodies of the IgG1 subtype protect against mucosal simian-human immunodeficiency virus infection. Nat. Med. 6, 200–206 (2000).
Hessell, A.J. et al. Effective, low-titer antibody protection against low-dose repeated mucosal SHIV challenge in macaques. Nat. Med. 15, 951–954 (2009).
Hessell, A.J. et al. Broadly neutralizing human anti-HIV antibody 2G12 is effective in protection against mucosal SHIV challenge even at low serum neutralizing titers. PLoS Pathog. 5, e1000433 (2009).
Shingai, M. et al. Passive transfer of modest titers of potent and broadly neutralizing anti-HIV monoclonal antibodies block SHIV infection in macaques. J. Exp. Med. 211, 2061–2074 (2014).
Pegu, A. et al. Neutralizing antibodies to HIV-1 envelope protect more effectively in vivo than those to the CD4 receptor. Sci. Transl. Med. 6, 243ra88 (2014).
Frankel, S.S. et al. Neutralizing monoclonal antibodies block human immunodeficiency virus type 1 infection of dendritic cells and transmission to T cells. J. Virol. 72, 9788–9794 (1998).
Veazey, R.S. et al. Prevention of virus transmission to macaque monkeys by a vaginally applied monoclonal antibody to HIV-1 gp120. Nat. Med. 9, 343–346 (2003).
Bomsel, M. et al. Immunization with HIV-1 gp41 subunit virosomes induces mucosal antibodies protecting nonhuman primates against vaginal SHIV challenges. Immunity 34, 269–280 (2011).
Parren, P.W. & Burton, D.R. The antiviral activity of antibodies in vitro and in vivo. Adv. Immunol. 77, 195–262 (2001).
Shingai, M. et al. Antibody-mediated immunotherapy of macaques chronically infected with SHIV suppresses viremia. Nature 503, 277–280 (2013).
Ferrantelli, F. et al. Post-exposure prophylaxis with human monoclonal antibodies prevented SHIV89.6P infection or disease in neonatal macaques. AIDS 17, 301–309 (2003).
Barouch, D.H. et al. Therapeutic efficacy of potent neutralizing HIV-1–specific monoclonal antibodies in SHIV-infected rhesus monkeys. Nature 503, 224–228 (2013).
Caskey, M. et al. Viremia suppressed in HIV-1–infected humans by broadly neutralizing antibody 3BNC117. Nature 522, 487–491 (2015).
Bolton, D.L. et al. Human immunodeficiency virus type 1 monoclonal antibodies suppress acute simian-human immunodeficiency virus viremia and limit seeding of cell-associated viral reservoirs. J. Virol. 90, 1321–1332 (2016).
Haigwood, N.L. et al. Passive immunotherapy in simian immunodeficiency virus–infected macaques accelerates the development of neutralizing antibodies. J. Virol. 78, 5983–5995 (2004).
Ng, C.T. et al. Passive neutralizing antibody controls SHIV viremia and enhances B cell responses in infant macaques. Nat. Med. 16, 1117–1119 (2010).
Jaworski, J.P. et al. Neutralizing polyclonal IgG present during acute infection prevents rapid disease onset in simian-human immunodeficiency virus SHIVSF162P3-infected infant rhesus macaques. J. Virol. 87, 10447–10459 (2013).
Kong, R. et al. Improving neutralization potency and breadth by combining broadly reactive HIV-1 antibodies targeting major neutralization epitopes. J. Virol. 89, 2659–2671 (2015).
Rudicell, R.S. et al. Enhanced potency of a broadly neutralizing HIV-1 antibody in vitro improves protection against lentiviral infection in vivo. J. Virol. 88, 12669–12682 (2014).
Walker, L.M. et al. Broad neutralization coverage of HIV by multiple highly potent antibodies. Nature 477, 466–470 (2011).
Julien, J.P. et al. Broadly neutralizing antibody PGT121 allosterically modulates CD4 binding via recognition of the HIV-1 gp120 V3 base and multiple surrounding glycans. PLoS Pathog. 9, e1003342 (2013).
Moldt, B. et al. Highly potent HIV-specific antibody neutralization in vitro translates into effective protection against mucosal SHIV challenge in vivo. Proc. Natl. Acad. Sci. USA 109, 18921–18925 (2012).
Jayaraman, P. et al. Evidence for persistent, occult infection in neonatal macaques following perinatal transmission of simian-human immunodeficiency virus SF162P3. J. Virol. 81, 822–834 (2007).
Hansen, S.G. et al. Profound early control of highly pathogenic SIV by an effector memory T cell vaccine. Nature 473, 523–527 (2011).
Okwundu, C.I., Uthman, O.A. & Okoromah, C.A. Antiretroviral pre-exposure prophylaxis (PrEP) for preventing HIV in high-risk individuals. Cochrane Database Syst. Rev. 7, CD007189 (2012).
Migueles, S.A. & Connors, M. Small molecules and big killers: the challenge of eliminating the latent HIV reservoir. Immunity 36, 320–321 (2012).
Allers, K. et al. Evidence for the cure of HIV infection by CCR5Δ32/Δ32 stem cell transplantation. Blood 117, 2791–2799 (2011).
Hansen, S.G. et al. Immune clearance of highly pathogenic SIV infection. Nature 502, 100–104 (2013).
Whitney, J.B. et al. Rapid seeding of the viral reservoir prior to SIV viremia in rhesus monkeys. Nature 512, 74–77 (2014).
Shah, S.K. et al. Research on very early ART in neonates at risk of HIV infection. Lancet Infect. Dis. 14, 797 (2014).
Persaud, D. et al. Absence of detectable HIV-1 viremia after treatment cessation in an infant. N. Engl. J. Med. 369, 1828–1835 (2013).
Miller, C.J. et al. Propagation and dissemination of infection after vaginal transmission of simian immunodeficiency virus. J. Virol. 79, 9217–9227 (2005).
Hessell, A.J. et al. Fc receptor but not complement binding is important in antibody protection against HIV. Nature 449, 101–104 (2007).
Ledgerwood, J.E. et al. Safety, pharmacokinetics and neutralization of the broadly neutralizing HIV-1 human monoclonal antibody VRC01 in healthy adults. Clin. Exp. Immunol. 182, 289–301 (2015).
Lynch, R.M. et al. The development of CD4–binding site antibodies during HIV-1 infection. J. Virol. 86, 7588–7595 (2012).
Committee for the Update of the Guide for the Care and Use of Laboratory Animals. Guide for the Care and Use of Laboratory Animals 8th edn. (The National Academies Press, Washington, D.C., 2011).
Leary, S. et al. AVMA Guidelines for the Euthanasia of Animals: 2013 Edition (American Veterinary Medical Association, Schaumburg, Illinois, USA, 2013).
Wu, X. et al. Rational design of envelope identifies broadly neutralizing human monoclonal antibodies to HIV-1. Science 329, 856–861 (2010).
Harouse, J.M. et al. Mucosal transmission and induction of simian AIDS by CCR5-specific simian-human immunodeficiency virus SHIVSF162P3 . J. Virol. 75, 1990–1995 (2001).
Cline, A.N., Bess, J.W., Piatak, M. Jr. & Lifson, J.D. Highly sensitive SIV plasma viral load assay: practical considerations, realistic performance expectations and application to reverse-engineering of vaccines for AIDS. J. Med. Primatol. 34, 303–312 (2005).
Malouli, D. et al. Cytomegalovirus pp65 limits dissemination but is dispensable for persistence. J. Clin. Invest. 124, 1928–1944 (2014).
Kozlowski, P.A., Cu-Uvin, S., Neutra, M.R. & Flanigan, T.P. Mucosal vaccination strategies for women. J. Infect. Dis. 179 (suppl. 3), S493–S498 (1999).
Malherbe, D.C. et al. Sequential immunization with a subtype B HIV-1 envelope quasispecies partially mimics the in vivo development of neutralizing antibodies. J. Virol. 85, 5262–5274 (2011).
Hirsch, V.M. et al. Induction of AIDS by simian immunodeficiency virus from an African green monkey: species-specific variation in pathogenicity correlates with the extent of in vivo replication. J. Virol. 69, 955–967 (1995).
Zhou, T. et al. Transplanting supersites of HIV-1 vulnerability. PLoS One 9, e99881 (2014).
Malherbe, D.C. et al. Envelope variants circulating as initial neutralization breadth developed in two HIV-infected subjects stimulate multiclade neutralizing antibodies in rabbits. J. Virol. 88, 12949–12967 (2014).
Acknowledgements
We thank D. Malherbe for helpful discussions and C. Corbacci for graphic design work. We appreciate the expertise, dedication and thoughtful care provided to the infant macaques by H. Sidener and the ONPRC nursery animal care technicians. Titrated stocks of SHIVSF162P3 (passage 3) virus were obtained through the AIDS Research and Reference Reagent Program, Division of AIDS, National Institute of Allergy and Infectious Diseases NIAID), NIH (catalog number 6526; contributors J. Harouse, C. Cheng-Mayer and R. Pal) for each study. The SIVsmE660 stock was kindly provided by V.M. Hirsch. The linearized pBSII-SIVgag was kindly gifted by Michael Piatak, National Cancer Institute at the NIH. Funding was provided by US Public Health Service grants from the NIH (grant no. R01-HD080459 (N.L.H.), P51-OD011092 (J. Robertson), P51-OD011092 pilot funding (E.E.)), the Elizabeth Glaser Pediatric AIDS Foundation (N.L.H.) and the American Foundation for AIDS Research (amfAR) (grant no. 108823-55-RGRL; N.L.H.). This work was also funded, in part, by the intramural research program of the Vaccine Research Center (B.S.G. and J.R.M.) and the Laboratory of Molecular Microbiology, NIAID, NIH, Division of Health and Human Services, US Public Health Service (V.M.H.).
Author information
Authors and Affiliations
Contributions
Studies were designed and planned by N.L.H. and A.J.H.; experimental work was done by A.J.H., J.P.J., E.E., K.M., S.P., J.R., W.F.S., K.B.H., T.A.C., P.T.B., A.W.L., S.P., X.C., K.W., D.S., D.B.; pathology was described by A.W.L.; veterinary care was provided by J.J.S.; A.J.H., N.L.H., J.B.S., J.R.M. and B.S.G. wrote the manuscript; A.J.H., J.P.J., E.E., C.K., M.K.A., V.M.H., A.P., J.B.S. and N.L.H. analyzed the data; B.S.P. performed statistical analyses.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6 and Supplementary Tables 1–2 (PDF 4926 kb)
Rights and permissions
About this article
Cite this article
Hessell, A., Jaworski, J., Epson, E. et al. Early short-term treatment with neutralizing human monoclonal antibodies halts SHIV infection in infant macaques. Nat Med 22, 362–368 (2016). https://doi.org/10.1038/nm.4063
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.4063
This article is cited by
-
More than the Infinite Monkey Theorem: NHP Models in the Development of a Pediatric HIV Cure
Current HIV/AIDS Reports (2024)
-
Research priorities for an HIV cure: International AIDS Society Global Scientific Strategy 2021
Nature Medicine (2021)
-
Single-dose bNAb cocktail or abbreviated ART post-exposure regimens achieve tight SHIV control without adaptive immunity
Nature Communications (2020)
-
Antibody-mediated prevention and treatment of HIV-1 infection
Retrovirology (2018)
-
A single injection of crystallizable fragment domain–modified antibodies elicits durable protection from SHIV infection
Nature Medicine (2018)