Abstract
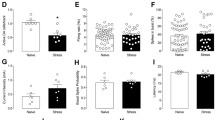
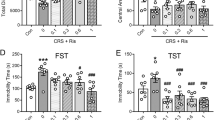
The lateral habenula (LHb) encodes aversive signals, and its aberrant activity contributes to depression-like symptoms. However, a limited understanding of the cellular mechanisms underlying LHb hyperactivity has precluded the development of pharmacological strategies to ameliorate depression-like phenotypes. Here we report that an aversive experience in mice, such as foot-shock exposure (FsE), induces LHb neuronal hyperactivity and depression-like symptoms. This occurs along with increased protein phosphatase 2A (PP2A) activity, a known regulator of GABAB receptor (GABABR) and G protein–gated inwardly rectifying potassium (GIRK) channel surface expression. Accordingly, FsE triggers GABAB1 and GIRK2 internalization, leading to rapid and persistent weakening of GABAB-activated GIRK-mediated (GABAB-GIRK) currents. Pharmacological inhibition of PP2A restores both GABAB-GIRK function and neuronal excitability. As a consequence, PP2A inhibition ameliorates depression-like symptoms after FsE and in a learned-helplessness model of depression. Thus, GABAB-GIRK plasticity in the LHb represents a cellular substrate for aversive experience. Furthermore, its reversal by PP2A inhibition may provide a novel therapeutic approach to alleviate symptoms of depression in disorders that are characterized by LHb hyperactivity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Accession codes
References
Knoll, A.T. & Carlezon, W.A.J. Jr. Dynorphin, stress and depression. Brain Res. 1314, 56–73 (2010).
Matsumoto, M. & Hikosaka, O. Lateral habenula as a source of negative reward signals in dopamine neurons. Nature 447, 1111–1115 (2007).
Morris, J.S., Smith, K.A., Cowen, P.J., Friston, K.J. & Dolan, R.J. Covariation of activity in habenula and dorsal raphé nuclei following tryptophan depletion. Neuroimage 10, 163–172 (1999).
Shumake, J., Edwards, E. & Gonzalez-Lima, F. Opposite metabolic changes in the habenula and ventral tegmental area of a genetic model of helpless behavior. Brain Res. 963, 274–281 (2003).
Chourbaji, S. et al. Learned helplessness: validity and reliability of depressive-like states in mice. Brain Res. Brain Res. Protoc. 16, 70–78 (2005).
Proulx, C.D., Hikosaka, O. & Malinow, R. Reward processing by the lateral habenula in normal and depressive behaviors. Nat. Neurosci. 17, 1146–1152 (2014).
Li, B. et al. Synaptic potentiation onto habenula neurons in the learned helplessness model of depression. Nature 470, 535–539 (2011).
Li, K. et al. β-CaMKII in lateral habenula mediates core symptoms of depression. Science 341, 1016–1020 (2013).
Brown, P.L. & Shepard, P.D. Lesions of the fasciculus retroflexus alter foot-shock–induced cFos expression in the mesopontine rostromedial tegmental area of rats. PLoS One 8, e60678 (2013).
Bagdy, G., Juhasz, G. & Gonda, X. A new clinical evidence-based gene-environment interaction model of depression. Neuropsychopharmacol. Hung. 14, 213–220 (2012).
Fatemi, S.H., Folsom, T.D. & Thuras, P.D. Deficits in GABAB receptor system in schizophrenia and mood disorders: a post-mortem study. Schizophr. Res. 128, 37–43 (2011).
Cryan, J.F. & Slattery, D.A. GABAB receptors and depression. Current status. Adv. Pharmacol. 58, 427–451 (2010).
Kawaura, K., Honda, S., Soeda, F., Shirasaki, T. & Takahama, K. Novel antidepressant-like action of drugs possessing GIRK channel–blocking action in rats. Yakugaku Zasshi 130, 699–705 (2010).
Llamosas, N., Bruzos-Cidón, C., Rodríguez, J.J., Ugedo, L. & Torrecilla, M. Deletion of GIRK2 subunit of GIRK channels alters the 5-HT1A receptor–mediated signaling and results in a depression-resistant behavior. Int. J. Neuropsychopharmacol. 18, pyv051 (2015).
Terunuma, M., Pangalos, M.N. & Moss, S.J. Functional modulation of GABAB receptors by protein kinases and receptor trafficking. Adv. Pharmacol. 58, 113–122 (2010).
Overmier, J.B. & Seligman, M.E. Effects of inescapable shock upon subsequent escape and avoidance responding. J. Comp. Physiol. Psychol. 63, 28–33 (1967).
Vollmayr, B. & Henn, F.A. Learned helplessness in the rat: improvements in validity and reliability. Brain Res. Brain Res. Protoc. 8, 1–7 (2001).
Lecca, S., Meye, F.J. & Mameli, M. The lateral habenula in addiction and depression: an anatomical, synaptic and behavioral overview. Eur. J. Neurosci. 39, 1170–1178 (2014).
Breier, A. et al. Controllable and uncontrollable stress in humans: alterations in mood and neuroendocrine and psychophysiological function. Am. J. Psychiatry 144, 1419–1425 (1987).
Maier, S.F. Learned helplessness and animal models of depression. Prog. Neuropsychopharmacol. Biol. Psychiatry 8, 435–446 (1984).
Stamatakis, A.M. & Stuber, G.D. Activation of lateral habenula inputs to the ventral midbrain promotes behavioral avoidance. Nat. Neurosci. 15, 1105–1107 (2012).
Lüthi, A. & Lüscher, C. Pathological circuit function underlying addiction and anxiety disorders. Nat. Neurosci. 17, 1635–1643 (2014).
Porsolt, R.D., Bertin, A. & Jalfre, M. Behavioral despair in mice: a primary screening test for antidepressants. Arch. Int. Pharmacodyn. Ther. 229, 327–336 (1977).
Meye, F.J. et al. Cocaine-evoked negative symptoms require AMPA receptor trafficking in the lateral habenula. Nat. Neurosci. 18, 376–378 (2015).
Shabel, S.J., Proulx, C.D., Piriz, J. & Malinow, R. Mood regulation. GABA and glutamate co-release controls habenula output and is modified by antidepressant treatment. Science 345, 1494–1498 (2014).
Lüscher, C. & Slesinger, P.A. Emerging roles for G protein–gated inwardly rectifying potassium (GIRK) channels in health and disease. Nat. Rev. Neurosci. 11, 301–315 (2010).
Logothetis, D.E., Kurachi, Y., Galper, J., Neer, E.J. & Clapham, D.E. The βγ subunits of GTP-binding proteins activate the muscarinic K+ channel in heart. Nature 325, 321–326 (1987).
Buynitsky, T. & Mostofsky, D.I. Restraint stress in biobehavioral research: recent developments. Neurosci. Biobehav. Rev. 33, 1089–1098 (2009).
Takahashi, L.K., Nakashima, B.R., Hong, H. & Watanabe, K. The smell of danger: a behavioral and neural analysis of predator odor–induced fear. Neurosci. Biobehav. Rev. 29, 1157–1167 (2005).
Zucker, R.S. & Regehr, W.G. Short-term synaptic plasticity. Annu. Rev. Physiol. 64, 355–405 (2002).
Hearing, M. et al. Repeated cocaine weakens GABAB-Girk signaling in layer 5/6 pyramidal neurons in the prelimbic cortex. Neuron 80, 159–170 (2013).
Padgett, C.L. et al. Methamphetamine-evoked depression of GABAB receptor signaling in GABA neurons of the VTA. Neuron 73, 978–989 (2012).
Guetg, N. et al. NMDA receptor–dependent GABAB receptor internalization via CaMKII phosphorylation of serine 867 in GABAB1 . Proc. Natl. Acad. Sci. USA 107, 13924–13929 (2010).
Morinobu, S. et al. Influence of immobilization stress on the expression and phosphatase activity of protein phosphatase 2A in the rat brain. Biol. Psychiatry 54, 1060–1066 (2003).
Mucic, G., Sase, S., Stork, O., Lubec, G. & Li, L. Networks of protein kinases and phosphatases in the individual phases of contextual fear conditioning in the C57BL/6J mouse. Behav. Brain Res. 280, 45–50 (2015).
Lu, J. et al. Inhibition of serine/threonine phosphatase PP2A enhances cancer chemotherapy by blocking DNA damage–induced defense mechanisms. Proc. Natl. Acad. Sci. USA 106, 11697–11702 (2009).
Jourdain, P., Fukunaga, K. & Muller, D. Calcium- and calmodulin-dependent protein kinase II contributes to activity-dependent filopodia growth and spine formation. J. Neurosci. 23, 10645–10649 (2003).
Lalive, A.L. et al. Firing modes of dopamine neurons drive bidirectional GIRK channel plasticity. J. Neurosci. 34, 5107–5114 (2014).
Wei, D. et al. Inhibition of protein phosphatase 2A radiosensitizes pancreatic cancers by modulating CDC25C and CDK1, and homologous recombination repair. Clin. Cancer Res. 19, 4422–4432 (2013).
Wang, M., Perova, Z., Arenkiel, B.R. & Li, B. Synaptic modifications in the medial prefrontal cortex in susceptibility and resilience to stress. J. Neurosci. 34, 7485–7492 (2014).
Strekalova, T., Spanagel, R., Bartsch, D., Henn, F.A. & Gass, P. Stress-induced anhedonia in mice is associated with deficits in forced swimming and exploration. Neuropsychopharmacology 29, 2007–2017 (2004).
Ménard, C., Hodes, G.E. & Russo, S.J. Pathogenesis of depression: insights from human and rodent studies. Neuroscience http://dx.doi.org/10.1016/j.neuroscience.2015.05.053 (2015).
González-Maeso, J., Wise, A., Green, A. & Koenig, J.A. Agonist-induced desensitization and endocytosis of heterodimeric GABAB receptors in CHO-K1 cells. Eur. J. Pharmacol. 481, 15–23 (2003).
Beaulieu, J.M. et al. An Akt–β-arrestin 2–PP2A signaling complex mediates dopaminergic neurotransmission and behavior. Cell 122, 261–273 (2005).
Chan, S.F. & Sucher, N.J. An NMDA receptor signaling complex with protein phosphatase 2A. J. Neurosci. 21, 7985–7992 (2001).
Lavine, N. et al. G protein–coupled receptors form stable complexes with inwardly rectifying potassium channels and adenylyl cyclase. J. Biol. Chem. 277, 46010–46019 (2002).
Nobles, M., Benians, A. & Tinker, A. Heterotrimeric G proteins precouple with G protein–coupled receptors in living cells. Proc. Natl. Acad. Sci. USA 102, 18706–18711 (2005).
Riven, I., Iwanir, S. & Reuveny, E. GIRK channel activation involves a local rearrangement of a preformed G protein channel complex. Neuron 51, 561–573 (2006).
Clancy, S.M., Boyer, S.B. & Slesinger, P.A. Co-regulation of natively expressed pertussis toxin–sensitive muscarinic receptors with G protein–activated potassium channels. J. Neurosci. 27, 6388–6399 (2007).
Luján, R., Marron Fernandez de Velasco, E., Aguado, C. & Wickman, K. New insights into the therapeutic potential of Girk channels. Trends Neurosci. 37, 20–29 (2014).
Lammel, S. et al. Input-specific control of reward and aversion in the ventral tegmental area. Nature 491, 212–217 (2012).
Jhou, T.C., Fields, H.L., Baxter, M.G., Saper, C.B. & Holland, P.C. The rostromedial tegmental nucleus (RMTg), a GABAergic afferent to midbrain dopamine neurons, encodes aversive stimuli and inhibits motor responses. Neuron 61, 786–800 (2009).
Tye, K.M. et al. Dopamine neurons modulate neural encoding and expression of depression-related behaviour. Nature 493, 537–541 (2013).
Nemeroff, C.B. The neurobiology of depression. Sci. Am. 278, 42–49 (1998).
Henn, F.A. Pharmacogenetic studies of depression. Biol. Psychiatry 63, 1101–1102 (2008).
Jhou, T.C. et al. Cocaine drives aversive conditioning via delayed activation of dopamine-responsive habenular and midbrain pathways. J. Neurosci. 33, 7501–7512 (2013).
Mameli, M., Balland, B., Luján, R. & Lüscher, C. Rapid synthesis and synaptic insertion of GluR2 for mGluR-LTD in the ventral tegmental area. Science 317, 530–533 (2007).
Fanselow, M.S. & Bolles, R.C. Naloxone- and shock-elicited freezing in the rat. J. Comp. Physiol. Psychol. 93, 736–744 (1979).
Can, A. et al. The mouse forced-swim test. J. Vis. Exp. 59, e3638 (2012).
Acknowledgements
We thank C. Bellone, M. Carta, E. Schwartz, C. Lüscher, F.J. Meye and members of the Mameli laboratory for discussions and comments on the manuscript. This work was supported by funds from the INSERM Atip-Avenir program (M.M.), a European Research Council starting grant (grant no. SalienSy 335333; M.M.), the Paris School of Neuroscience Network (ENP) Chair of Excellence (M.M.) and the Junta de Comunidades de Castilla–La Mancha (grant no. PPII-2014-005-P; R.L.). The Mameli laboratory is part of the Initiative of Excellence Labex BioPsy network. We thank J. Kovach and Lixte Biotechnology Holdings, Inc. for the gift of LB-100. The monoclonal antibody specific to GABAB1 (clone N93A/49) was developed by the University of California, Davis–US National Institutes of Health NeuroMab facility (Department of Neurobiology, Physiology and Behavior, College of Biological Sciences, University of California, Davis).
Author information
Authors and Affiliations
Contributions
S.L. and M.M. performed and analyzed all of the in vitro electrophysiological recordings and behavioral experiments with the help of A.T.; A.P. and D.H. designed and performed biochemical assays; I.M. performed immunohistochemistry and designed the viral vector; R.L. performed electron microscopy and analyzed the data; and S.L. and M.M. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–9 (PDF 6081 kb)
Supplementary Data
Supplementary Source Data (XLSX 45 kb)
Rights and permissions
About this article
Cite this article
Lecca, S., Pelosi, A., Tchenio, A. et al. Rescue of GABAB and GIRK function in the lateral habenula by protein phosphatase 2A inhibition ameliorates depression-like phenotypes in mice. Nat Med 22, 254–261 (2016). https://doi.org/10.1038/nm.4037
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.4037
This article is cited by
-
Potentiation of the lateral habenula-ventral tegmental area pathway underlines the susceptibility to depression in mice with chronic pain
Science China Life Sciences (2024)
-
Involvement of cortical input to the rostromedial tegmental nucleus in aversion to foot shock
Neuropsychopharmacology (2023)
-
Effects of early life stress and subsequent re-exposure to stress on neuronal activity in the lateral habenula
Neuropsychopharmacology (2023)
-
Sustained antidepressant effect of ketamine through NMDAR trapping in the LHb
Nature (2023)
-
Plasticity of synapses and reward circuit function in the genesis and treatment of depression
Neuropsychopharmacology (2023)