Abstract
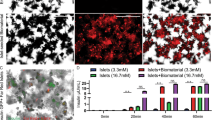
The transplantation of glucose-responsive, insulin-producing cells offers the potential for restoring glycemic control in individuals with diabetes1. Pancreas transplantation and the infusion of cadaveric islets are currently implemented clinically2, but these approaches are limited by the adverse effects of immunosuppressive therapy over the lifetime of the recipient and the limited supply of donor tissue3. The latter concern may be addressed by recently described glucose-responsive mature beta cells that are derived from human embryonic stem cells (referred to as SC-β cells), which may represent an unlimited source of human cells for pancreas replacement therapy4. Strategies to address the immunosuppression concerns include immunoisolation of insulin-producing cells with porous biomaterials that function as an immune barrier5,6. However, clinical implementation has been challenging because of host immune responses to the implant materials7. Here we report the first long-term glycemic correction of a diabetic, immunocompetent animal model using human SC-β cells. SC-β cells were encapsulated with alginate derivatives capable of mitigating foreign-body responses in vivo and implanted into the intraperitoneal space of C57BL/6J mice treated with streptozotocin, which is an animal model for chemically induced type 1 diabetes. These implants induced glycemic correction without any immunosuppression until their removal at 174 d after implantation. Human C-peptide concentrations and in vivo glucose responsiveness demonstrated therapeutically relevant glycemic control. Implants retrieved after 174 d contained viable insulin-producing cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Change history
18 February 2016
In the version of this article initially published online, the authors omitted acknowledgment recognizing the histology core of the Harvard Stem Cell Institute and several individuals for their assistance. The error has been corrected for the print, PDF and HTML versions of this article.
References
Robertson, R.P. Islet transplantation as a treatment for diabetes—a work in progress. N. Engl. J. Med. 350, 694–705 (2004).
Shapiro, A.M. et al. International trial of the Edmonton protocol for islet transplantation. N. Engl. J. Med. 355, 1318–1330 (2006).
Hirshberg, B. Lessons learned from the international trial of the Edmonton protocol for islet transplantation. Curr. Diab. Rep. 7, 301–303 (2007).
Pagliuca, F.W. et al. Generation of functional human pancreatic β-cells in vitro. Cell 159, 428–439 (2014).
Lim, F. & Sun, A.M. Microencapsulated islets as bioartificial endocrine pancreas. Science 210, 908–910 (1980).
Soon-Shiong, P. et al. Insulin independence in a type 1 diabetic patient after encapsulated islet transplantation. Lancet 343, 950–951 (1994).
Dolgin, E. Encapsulate this. Nat. Med. 20, 9–11 (2014).
Shaw, J.E., Sicree, R.A. & Zimmet, P.Z. Global estimates of the prevalence of diabetes for 2010 and 2030. Diabetes Res. Clin. Pract. 87, 4–14 (2010).
Pickup, J.C. Insulin-pump therapy for type 1 diabetes mellitus. N. Engl. J. Med. 366, 1616–1624 (2012).
Veiseh, O., Tang, B.C., Whitehead, K.A., Anderson, D.G. & Langer, R. Managing diabetes with nanomedicine: challenges and opportunities. Nat. Rev. Drug Discov. 14, 45–57 (2015).
Shapiro, A.M. et al. Islet transplantation in seven patients with type 1 diabetes mellitus using a glucocorticoid-free immunosuppressive regimen. N. Engl. J. Med. 343, 230–238 (2000).
Qi, M. et al. Five-year follow-up of patients with type 1 diabetes transplanted with allogeneic islets: the UIC experience. Acta Diabetol. 51, 833–843 (2014).
Shapiro, A.M. Islet transplantation in type 1 diabetes: ongoing challenges, refined procedures and long-term outcome. Rev. Diabet. Stud. 9, 385–406 (2012).
Vogel, G. Stem cell recipe offers diabetes hope. Science 346, 148 (2014).
Jacobs-Tulleneers-Thevissen, D. et al. Sustained function of alginate-encapsulated human islet cell implants in the peritoneal cavity of mice leading to a pilot study in a type 1 diabetic patient. Diabetologia 56, 1605–1614 (2013).
Scharp, D.W. & Marchetti, P. Encapsulated islets for diabetes therapy: history, current progress and critical issues requiring solution. Adv. Drug Deliv. Rev. 67–68, 35–73 (2014).
Lum, Z.P. et al. Prolonged reversal of diabetic state in NOD mice by xenografts of microencapsulated rat islets. Diabetes 40, 1511–1516 (1991).
Schneider, S. et al. Long-term graft function of adult rat and human islets encapsulated in novel alginate-based microcapsules after transplantation in immunocompetent diabetic mice. Diabetes 54, 687–693 (2005).
Peppas, N.A., Hilt, J.Z., Khademhosseini, A. & Langer, R. Hydrogels in biology and medicine: from molecular principles to bionanotechnology. Adv. Mater. 18, 1345–1360 (2006).
Basta, G. et al. Long-term metabolic and immunological follow-up of non-immunosuppressed patients with type 1 diabetes treated with microencapsulated islet allografts: four cases. Diabetes Care 34, 2406–2409 (2011).
Calafiore, R. et al. Microencapsulated pancreatic islet allografts into non-immunosuppressed patients with type 1 diabetes: first two cases. Diabetes Care 29, 137–138 (2006).
de Groot, M., Schuurs, T.A. & van Schilfgaarde, R. Causes of limited survival of microencapsulated pancreatic islet grafts. J. Surg. Res. 121, 141–150 (2004).
Tuch, B.E. et al. Safety and viability of microencapsulated human islets transplanted into diabetic humans. Diabetes Care 32, 1887–1889 (2009).
Elliott, R.B. et al. Intraperitoneal alginate-encapsulated neonatal porcine islets in a placebo-controlled study with 16 diabetic cynomolgus primates. Transplant. Proc. 37, 3505–3508 (2005).
Omer, A. et al. Survival and maturation of microencapsulated porcine neonatal pancreatic cell clusters transplanted into immunocompetent diabetic mice. Diabetes 52, 69–75 (2003).
Veiseh, O. et al. Size- and shape-dependent foreign-body immune response to materials implanted in rodents and nonhuman primates. Nat. Mater. 14, 643–651 (2015).
Vegas, A.J. et al. Combinatorial hydrogel library enables identification of materials that mitigate the foreign-body response in primates. Nat. Biotechnol. doi:10.1038/nbt.3462 (25 January 2016).
Kolb, M. et al. Differences in the fibrogenic response after transfer of active transforming growth factor–β1 gene to lungs of 'fibrosis-prone' and 'fibrosis-resistant' mouse strains. Am. J. Respir. Cell Mol. Biol. 27, 141–150 (2002).
Dang, T.T. et al. Spatiotemporal effects of a controlled-release anti-inflammatory drug on the cellular dynamics of host response. Biomaterials 32, 4464–4470 (2011).
King, A., Sandler, S. & Andersson, A. The effect of host factors and capsule composition on the cellular overgrowth on implanted alginate capsules. J. Biomed. Mater. Res. 57, 374–383 (2001).
Pepper, A.R. et al. A prevascularized subcutaneous device-less site for islet and cellular transplantation. Nat. Biotechnol. 33, 518–523 (2015).
Lee, S.H. et al. Human beta cell precursors mature into functional insulin-producing cells in an immunoisolation device: implications for diabetes cell therapies. Transplantation 87, 983–991 (2009).
Rezania, A. et al. Maturation of human embryonic stem cell–derived pancreatic progenitors into functional islets capable of treating pre-existing diabetes in mice. Diabetes 61, 2016–2029 (2012).
Rezania, A. et al. Reversal of diabetes with insulin-producing cells derived in vitro from human pluripotent stem cells. Nat. Biotechnol. 32, 1121–1133 (2014).
Wang, T. et al. An encapsulation system for the immunoisolation of pancreatic islets. Nat. Biotechnol. 15, 358–362 (1997).
Acknowledgements
This work was supported jointly by the JDRF and the Leona M. and Harry B. Helmsley Charitable Trust (grant no. 3-SRA-2014-285-M-R (R.L. and D.G.A.)), the US National Institutes of Health (grants EB000244 (R.L.), EB000351 (R.L.), DE013023 (R.L.), CA151884 (R.L.) and UC4DK104218 (D.L.G.)), and through a generous gift from the Tayebati Family Foundation (D.G.A. and R.L.). O.V. was supported by JDRF and Department of Defense Congressionally Directed Medical Research Program (DOD/CDMRP) postdoctoral fellowships (grants 3-2013-178 and W81XWH-13-1-0215, respectively). J.R.M. was supported by a fellowship from the Harvard Stem Cell Institute. J.O. is supported by the Chicago Diabetes Project. The authors acknowledge R. Bogorad for useful discussions and assistance and the Koch Institute Swanson Biotechnology Center for technical support, specifically for the use of the Hope Babette Tang Histology, Microscopy, Flow Cytometry and Animal Imaging and preclinical testing core facilities. We acknowledge the use of imaging resources at the Harvard University Center for Nanoscale Systems, the W.M. Keck Biological Imaging Facility (Whitehead Institute) and the histology core of the Harvard Stem Cell Institute. We would like to thank A. Graham, W. Salmon, C. MacGillivray and J. Wyckoff for their assistance.
Author information
Authors and Affiliations
Contributions
A.J.V., O.V. and D.G.A. designed experiments, analyzed data and wrote the manuscript. M.G., J.R.M., F.W.P. and D.A.M. provided SC-β cells. A.J.V., O.V., M.G., J.R.M., F.W.P., A.R.B., J.C.D., J.L., M.C., K.O., S.J., E.L., S.A.-D., S.G., J.J.M., M.A.B. and J.H.-L. performed experiments. H.H.T. performed statistical analyses of data sets and aided in the preparation of displays communicating data sets. J.O., D.L.G., G.C.W., D.A.M. and R.L. provided conceptual advice and technical support. R.L. and D.G.A. supervised the study. All of the authors discussed the results and assisted in the preparation of the manuscript.
Corresponding author
Ethics declarations
Competing interests
F.W.P., J.R.M., M.G. and D.A.M. declare a financial interest via a patent filed by Harvard University and HHMI on the production of stem cell–derived β-cells. A.J.V., O.V., J.C.D., R.L. and D.G.A. declare financial interests via patents filed by MIT on the material and hydrogel capsule technology.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7 (PDF 1541 kb)
Rights and permissions
About this article
Cite this article
Vegas, A., Veiseh, O., Gürtler, M. et al. Long-term glycemic control using polymer-encapsulated human stem cell–derived beta cells in immune-competent mice. Nat Med 22, 306–311 (2016). https://doi.org/10.1038/nm.4030
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.4030
This article is cited by
-
Hypoimmune induced pluripotent stem cells survive long term in fully immunocompetent, allogeneic rhesus macaques
Nature Biotechnology (2024)
-
Electrocatalytic on-site oxygenation for transplanted cell-based-therapies
Nature Communications (2023)
-
Screening hydrogels for antifibrotic properties by implanting cellularly barcoded alginates in mice and a non-human primate
Nature Biomedical Engineering (2023)
-
Encapsulated stem cell–derived β cells exert glucose control in patients with type 1 diabetes
Nature Biotechnology (2023)
-
Advances in alginate encapsulation of pancreatic islets for immunoprotection in type 1 diabetes
Journal of Pharmaceutical Investigation (2023)